- Search
Abstract
Purpose
Leptin is encoded by the ob gene and is involved in the control of food intake and energy expenditure. Recent studies have implicated leptin expression to be an indicator of tumor features and prognosis. The purpose of this study was to investigate the association of tissue expression of leptin with the clinicopathological characteristics and clinical outcomes in colorectal cancer patients.
Methods
Patients who had undergone a curative surgical resection for a colorectal adenocarcinoma from 2000 to 2004 were included in the study. Immunohistochemical analyses of leptin expression were performed, and clinicopathological parameters were evaluated.
Results
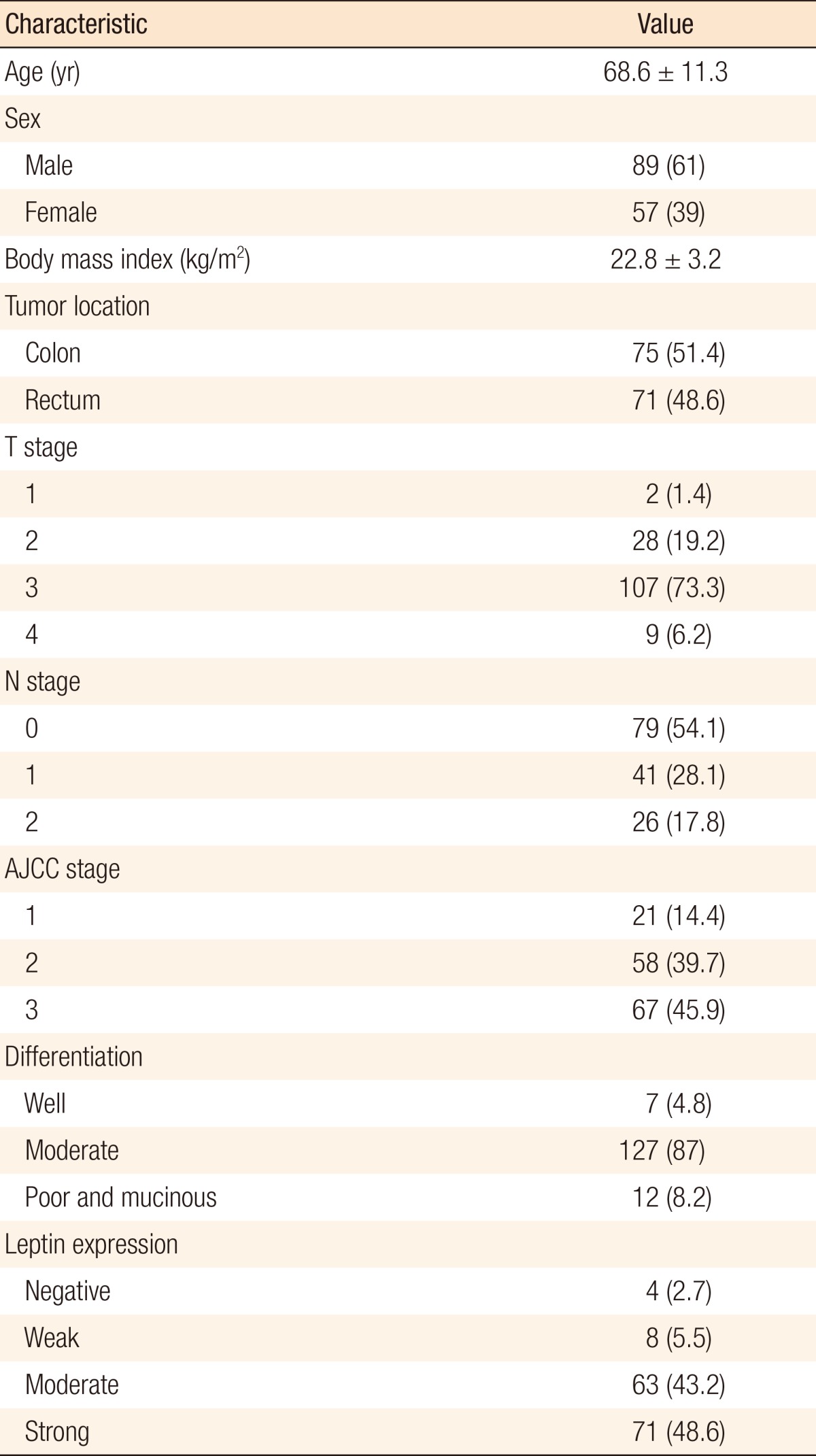
Clinical data and tumor tissues of 146 patients were evaluated. The mean age was 68.6 ± 11.3 years, and 61.0% were men. Immunohistochemically, the rates of negative, weak, moderate, and strong leptin expression were 2.7% (4 of 146), 5.5% (8 of 146), 43.2% (63 of 146), and 48.6% (71 of 146), respectively. We compared the negative, weak, and moderate expression group (group A) with the strong expression group (group B). Leptin expression was inversely associated with nodal stage (P = 0.007) between the two groups. Leptin expression was not significantly associated with differentiation (P = 0.37), T stage (P = 0.16), and American Joint Committee on Cancer stage (P = 0.49), and no significant differences in the disease-free and the overall survivals (P = 0.78 and P = 0.61) were observed.
A generally accepted idea is that obesity and/or endocrine dysfunction of adipose tissue is related to the development of and the prognosis for colorectal cancer. Leptin is encoded by the ob gene, which is secreted primarily by adipocytes [1]. This 167-amino-acid cytokine-like peptide regulates food intake and energy expenditure via interactions with specific receptors in the hypothalamus [2,3]. In obese individuals, increased serum leptin levels are observed [4].
A suggestion has been made that leptin expression can be used as an indicator of tumor features and prognosis as studies have shown that leptin can stimulate the proliferation and the migration of normal intestinal epithelial cells and colorectal cancer cells in vitro [5,6]. These studies strongly suggest the potential of leptin in colorectal carcinogenesis. There has, however, been some controversy regarding the relationship between the serum leptin level and colorectal cancer development [7,8,9]. Studies on the relationship of leptin expression levels in the tissues of a normal healthy colon, adenomas, and adenocarcinomas have shown somewhat inconsistent results [10,11]. While studies have investigated the correlation between tissue leptin expression and survival in colorectal carcinoma patients [10,11,12], sufficient data are not available to reach an informed conclusion. This study, therefore, aimed to determine the association of leptin tissue expression with the clinicopathological characteristics, as well as the clinical outcomes, in colorectal adenocarcinoma patients.
The medical records of colorectal adenocarcinoma patients who had consecutively undergone a curative surgical resection from 2000 to 2004 were reviewed retrospectively. We also obtained survival data for the patients from the Division of Cancer Registration & Surveillance, National Cancer Control Institute, Korea. Patients having a history of preoperative chemotherapy or radiotherapy, having distant metastases and having ages less than 20 years were excluded. A total of 146 patients were enrolled in this study, and 146 tumor tissue samples were collected from the patients. All tissue samples were formalin-fixed and paraffin-embedded. Hematoxylin and eosin slides and all medical records were reviewed. This study protocol was reviewed and approved by the Institutional Review Board, Keimyung University Donsan Medical Center (IRB No.: 2014-11-046). Informed consent was waived due to the retrospective design of the study.
Paraffin blocks were used for the reconstruction of the tissue microarray (TMA). All tissues were fixed in 10% neutral-buffered formalin and were embedded in paraffin. A representative area without necrosis was selected by using a light microscope from each tumor paraffin block to construct the TMA. A pair of 3-mm-diameter tissue cores was taken from the donor paraffin blocks and was transferred to the recipient paraffin blocks by using a Quick-Ray Manual Tissue Microarrayer (Unitma, Seoul, Korea).
Immunohistochemistry was performed by using the automated Benchmark platform (Ventana Medical Systems, Tucson, AZ, USA) according to the manufacturer's recommendations. From each TMA block, 4-µm-thick sections were obtained and immunostained for leptin by using an iView universal DAB detection kit (Ventana Medical Systems). Antigen retrieval by using a cell-conditioning solution (Ventana Medical Systems) was applied for all cases. The primary antibody used was a rabbit polyclonal antileptin antibody (Y-20, Santa Cruz Biotechnology, Santa Cruz, CA, USA) diluted 1:100. All slides were counterstained with hematoxylin (Fig. 1). For the negative control staining, the primary antibody was omitted. In case of positive control staining, a formalin-fixed paraffin-embedded normal human placenta-tissue section was used.
An expert pathologist examined the tumor tissues. Leptin expression was assessed by two pathologists who were blinded to the pathologic information on the TMA, the original diagnosis, and the patient outcomes at the time of assessment. The scoring systems for the staining intensity and for the extent of leptin expression were derived from a previous study [11]. The staining intensity for leptin was given a score of 0 (negative), 1 (weak), 2 (moderate), or 3 (strong) (Fig. 2). The staining extent for leptin was given a score of 0 (0%), 1 (1%-25%), 2 (26%-50%), or 3 (51%-100%), according to the percentage of cells stained positively for leptin. Leptin expression was categorized into four groups after multiplying above scores: negative (0), weak (1-3), moderate (4-6), and strong (9).
For statistical analyses, Pearson chi-square test, Fisher exact test, Student t-test, or one-way analysis of variance was used depending on the nature of data. The Kaplan-Meier method was used to analyze survival. A two-tailed P < 0.05 was considered statistically significant.
The characteristics of and the clinical data for the enrolled patients are presented in Table 1. The mean age of the patients was 68.6 years (range, 39-100 years), and 61% were men. The mean body mass index (BMI) was 22.8 kg/m2 (range, 16.9-31.6 kg/m2); 75 of the patients (51.4%) had colon cancer, and 71 (48.6%) had rectal cancer.
Immunohistochemically, the rates of negative, weak, moderate, and strong leptin expression were 2.7% (4 of 146), 5.5% (8 of 146), 43.2% (63 of 146), and 48.6% (71 of 146), respectively. Almost half the patients showed strong leptin expression. The patients were divided into two groups: negative, weak, and moderate expression group (group A) and strong expression group (group B). In all, 75 patients (51.4%) were included in group A, and 71 patients (48.6%) in group B.
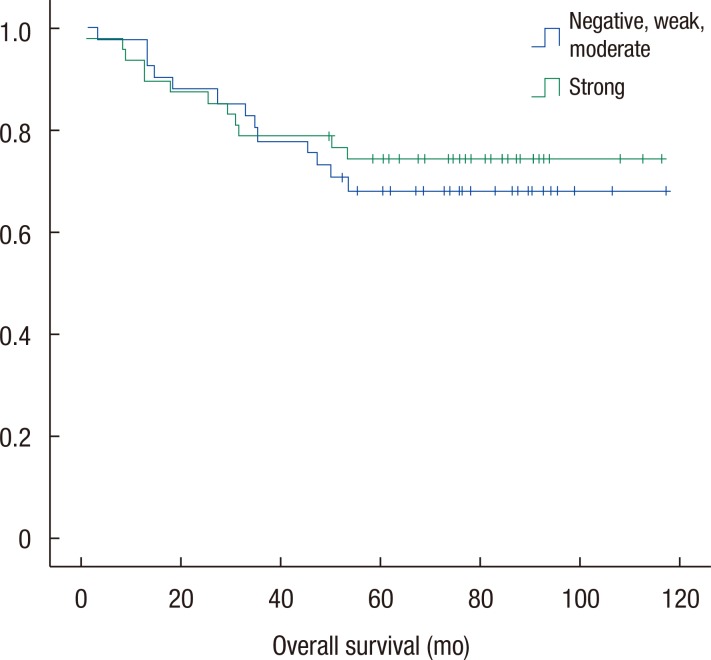
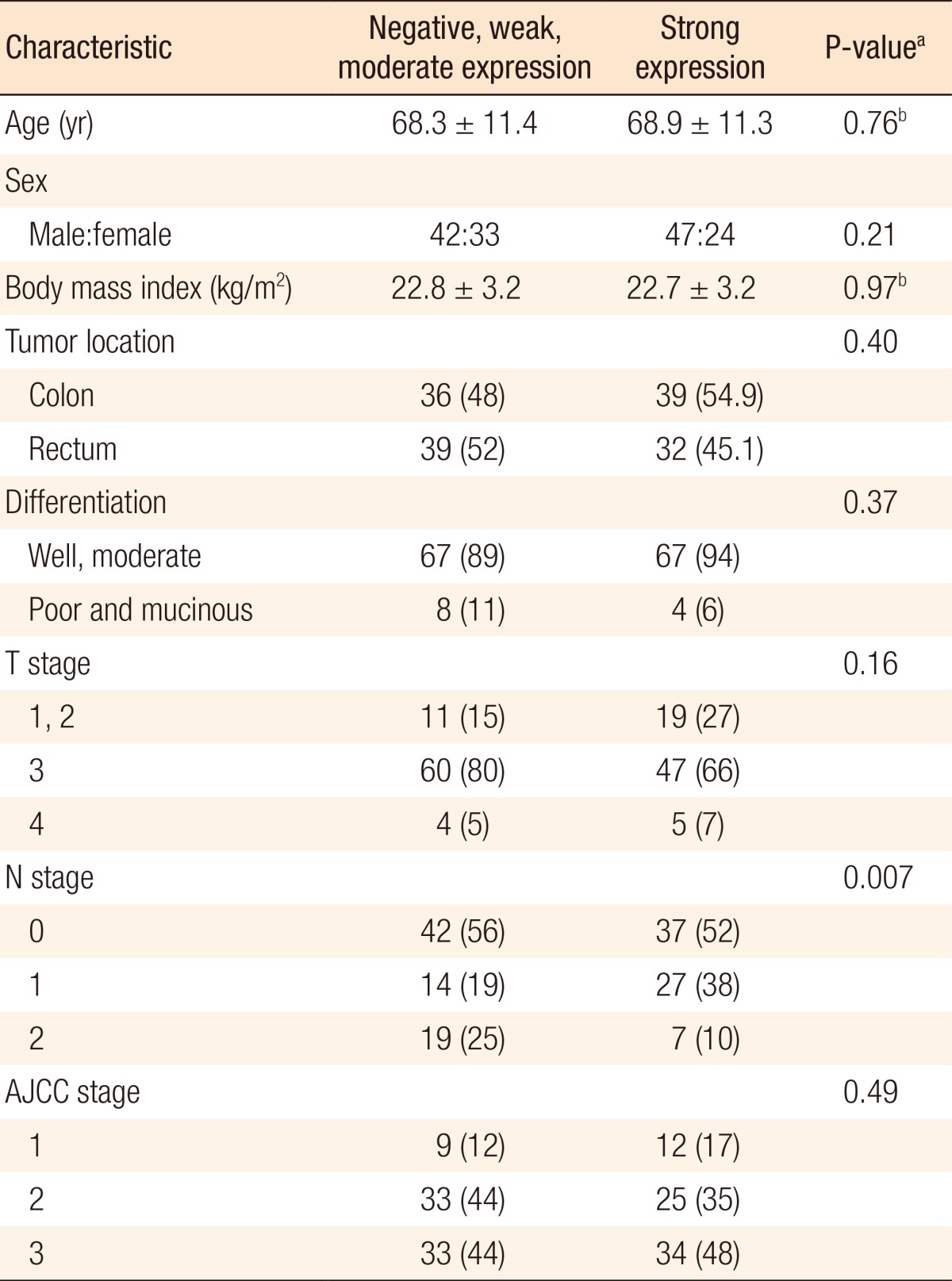
Groups A and B were compared with respect to demographics and oncologic outcomes. Disease-free and overall survival rates were also compared. The two groups were matched for age, gender, BMI, and tumor location (Table 2). Tumor differentiation grade (P = 0.37), T stage (P = 0.16), and American Joint Committee on Cancer (AJCC) stage (P = 0.49) were not significantly different between the two groups. N stage alone showed a significant difference (P = 0.007). Leptin expression was inversely associated with nodal stage between the 2 groups. No significant difference in the disease-free (Fig. 3) and the overall survival (Fig. 4) rates were observed (P = 0.78 and P = 0.61, respectively).
Much controversy has existed regarding the relationship between serum leptin levels and colorectal cancer development. For example, a study by Stattin et al. [7] found a significant increase in colon cancer risk with increasing levels of serum leptin while another study observed no increases in serum leptin concentrations in colorectal cancer patients [8], and a third investigation reported that serum leptin concentrations in patients with advanced gastrointestinal cancer were lower than those in controls [9].
Studies have also investigated and compared the levels of tissue leptin expression in a normal colon, an adenoma, and an adenocarcinoma. Although the results were somewhat inconsistent, studies have indicated that high tissue leptin expression may be correlated with colorectal carcinogenesis. Koda et al. [5] found that the leptin expression levels in both adenoma and adenocarcinoma tissues were higher than those in normal tissues while the percentages of positive leptin expression in adenoma and adenocarcinoma tissues were similar. However, Paik et al. [11] demonstrated a gradient of increasing tissue leptin expression in normal, adenoma, and adenocarcinoma tissues (normal - adenoma - adenocarcinoma), which may be an indication of the key involvement of leptin in multistep colorectal carcinogenesis.
Recently, experimental studies regarding the effects of leptin on colorectal cancer have been published. Leptin has been shown to have mitogenic and anti-apoptotic effects on colon cancer [5,6,13,14]. In vitro studies have demonstrated that leptin affects processes related to colon cancer initiation and progression [15], influences the growth and survival of colorectal cancer stem cell, and enhances adhesion and invasion of colorectal cancer [16]. In an animal study, leptin seemed to stimulate the proliferation of colon cancer cells [17].
The relationship between leptin expression and oncologic outcomes has not been elucidated sufficiently. Although studies have investigated the survival of patients with a colorectal adenocarcinoma according to leptin expression, data are insufficient to draw any conclusions. In the current study, the strong leptin expression group (group B) showed less lymph node metastasis than the negative, weak, and moderate leptin expression group (group A). No significant differences were observed between the two groups when the N0 and the N1+N2 stages were compared (56% and 44% vs. 52% and 48%, respectively; P = 0.638). The percentages of N0 stage between the two groups were similar (42 of 75, 56% vs. 37 of 71, 52%). Fewer patients in group A had N1 stage (14 of 75, 19% vs. 27 of 71, 38%); however, the percentage of N2 stage was higher in group A (19 of 75, 25% vs. 7 of 71, 10%). Group A had more patients with more advanced N stage (N2) than group B. Our data suggest that leptin expression is inversely associated with lymph node involvement. We postulate that a higher leptin expression level might predict a better oncologic outcome.
These results are consistent with those of a study by Paik et al. [11], in which low leptin expression correlated frequently with lymph node metastasis. Koda et al. [5], however, reported no significant relationship between leptin expression and lymph node metastasis. In our study, no significant associations of leptin expression with depth of invasion (T stage) and AJCC stage were observed, which is in line with the results reported in previous studies by Koda et al. [5]. In contrast, Paik et al. [11] reported that leptin expression correlated with tumor features, such as the depth of invasion and the AJCC and Duke's stages. Another prognostic factor evaluated in our study was tumor differentiation, and we found no significant differences in leptin expressions for the different tumor differentiation grades between the two groups in this study, although well and moderately differentiated tumors were more prominent in group B (89% vs. 94%, respectively). Previous investigations have found lower levels of leptin expression in poorly differentiated tumors than in better differentiated tumors [5,11]. Furthermore, Paik et al. [11] reported that the high leptin expression group showed better survival than the low or the negative leptin expression group. In the current study, however, we found no significant differences in the disease-free and the overall survival rates between groups A and B.
In the current study, the relationship between leptin expression in cancer tissue and oncologic outcome was not fully consistent with that reported in previous studies. In those studies, high leptin expression did not show unfavorable oncologic outcome, although the prognostic factors showing favorable outcome were inconsistent between studies. In the study by Koda et al. [5], tumors with better differentiation showed high leptin expression while in the study by Paik et al. [11], high leptin expression correlated with favorable outcomes with regard to the depth of invasion, lymph node metastasis, AJCC stage, tumor differentiation, and lymphatic invasion. In our study, high leptin expression correlated with lower nodal stage. Based on these results, high leptin expression in colorectal cancer tissue might be a prognostic factor, although further evaluation is required to determine the role of leptin in the prognosis for colorectal cancer patients.
The current study has some limitations. First, this study was not a randomized controlled study; thus, it could be affected by potential selection bias. Second, the sample size (146 patients) of this study was not estimated because tumor tissues were retrospectively recruited, so a sample-size estimate could not be performed. Finally, the sample size was relatively small. The number of patients enrolled in the study of Paik et al. [11] was notably higher than those in the study of Koda et al. [5] and our study (437, 166, and 146, respectively).
In conclusion, in this study, high leptin expression level was found to be inversely associated with nodal stage. In addition, the results showed that a higher leptin expression level might also be a predictor of a better oncologic outcome. However, further studies are warranted to identify the exact role of leptin expression in colorectal cancer.
References
1. Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature 1994;372:425–432. PMID: 7984236.


2. Zhao X, Huang K, Zhu Z, Chen S, Hu R. Correlation between expression of leptin and clinicopathological features and prognosis in patients with gastric cancer. J Gastroenterol Hepatol 2007;22:1317–1321. PMID: 17559372.


3. Attele AS, Shi ZQ, Yuan CS. Leptin, gut, and food intake. Biochem Pharmacol 2002;63:1579–1583. PMID: 12007560.


4. Considine RV, Sinha MK, Heiman ML, Kriauciunas A, Stephens TW, Nyce MR, et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N Engl J Med 1996;334:292–295. PMID: 8532024.


5. Koda M, Sulkowska M, Kanczuga-Koda L, Surmacz E, Sulkowski S. Overexpression of the obesity hormone leptin in human colorectal cancer. J Clin Pathol 2007;60:902–906. PMID: 17660334.



6. Hardwick JC, Van Den Brink GR, Offerhaus GJ, Van Deventer SJ, Peppelenbosch MP. Leptin is a growth factor for colonic epithelial cells. Gastroenterology 2001;121:79–90. PMID: 11438496.


7. Stattin P, Lukanova A, Biessy C, Soderberg S, Palmqvist R, Kaaks R, et al. Obesity and colon cancer: does leptin provide a link? Int J Cancer 2004;109:149–152. PMID: 14735482.


8. Tessitore L, Vizio B, Jenkins O, De Stefano I, Ritossa C, Argiles JM, et al. Leptin expression in colorectal and breast cancer patients. Int J Mol Med 2000;5:421–426. PMID: 10719061.


9. Bolukbas FF, Kilic H, Bolukbas C, Gumus M, Horoz M, Turhal NS, et al. Serum leptin concentration and advanced gastrointestinal cancers: a case controlled study. BMC Cancer 2004;4:29PMID: 15217519.



10. Koda M, Sulkowska M, Kanczuga-Koda L, Cascio S, Colucci G, Russo A, et al. Expression of the obesity hormone leptin and its receptor correlates with hypoxia-inducible factor-1 alpha in human colorectal cancer. Ann Oncol 2007;18(Suppl 6): vi116–vi119. PMID: 17591803.


11. Paik SS, Jang SM, Jang KS, Lee KH, Choi D, Jang SJ. Leptin expression correlates with favorable clinicopathologic phenotype and better prognosis in colorectal adenocarcinoma. Ann Surg Oncol 2009;16:297–303. PMID: 19050975.

12. Uddin S, Bavi PP, Hussain AR, Alsbeih G, Al-Sanea N, Abduljabbar A, et al. Leptin receptor expression in Middle Eastern colorectal cancer and its potential clinical implication. Carcinogenesis 2009;30:1832–1840. PMID: 19520793.


13. Ratke J, Entschladen F, Niggemann B, Zanker KS, Lang K. Leptin stimulates the migration of colon carcinoma cells by multiple signaling pathways. Endocr Relat Cancer 2010;17:179–189. PMID: 19952122.


14. Huang XF, Chen JZ. Obesity, the PI3K/Akt signal pathway and colon cancer. Obes Rev 2009;10:610–616. PMID: 19527447.


15. Jaffe T, Schwartz B. Leptin promotes motility and invasiveness in human colon cancer cells by activating multiple signal-transduction pathways. Int J Cancer 2008;123:2543–2556. PMID: 18767036.


16. Yoon KW, Park SY, Kim JY, Lee SM, Park CH, Cho SB, et al. Leptin-induced adhesion and invasion in colorectal cancer cell lines. Oncol Rep 2014;31:2493–2498. PMID: 24700392.


17. Endo H, Hosono K, Uchiyama T, Sakai E, Sugiyama M, Takahashi H, et al. Leptin acts as a growth factor for colorectal tumours at stages subsequent to tumour initiation in murine colon carcinogenesis. Gut 2011;60:1363–1371. PMID: 21406387.


Fig. 1
Sections of normal colonic mucosal (A) and cancer (B) tissue immunostained for leptin (×200). Diffuse and mild intensity with a cytoplasmic membranous staining pattern is seen in the normal colonic mucosal cells (A, filled arrow). Focal and mild intensity with a cytoplasmic membranous staining pattern in colon cancer cells (B, unfilled arrow).
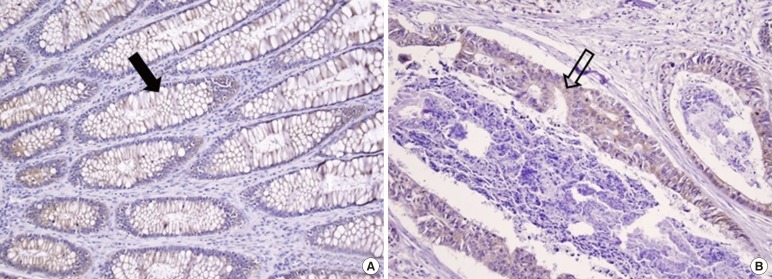
Fig. 2
Sections of colorectal cancer tissue immunostained for leptin (×200): (A) negative, (B) weak, (C) moderate, and (D) strong expression.
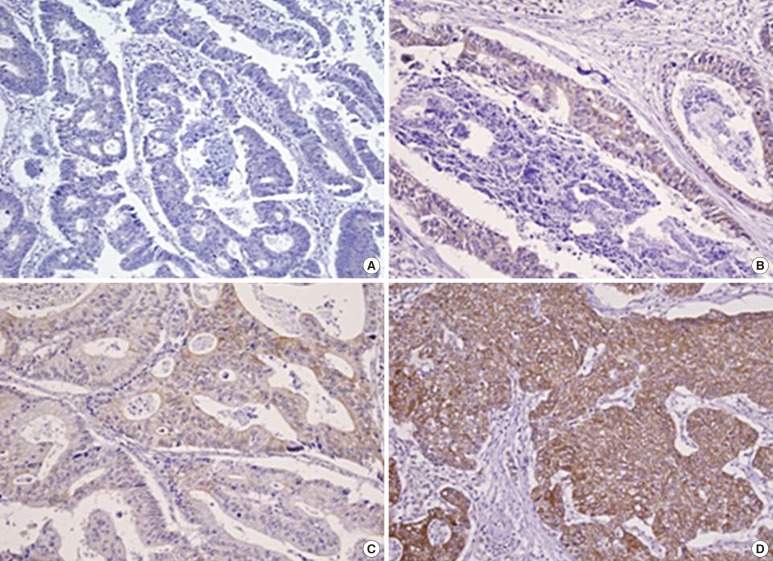
Fig. 3
Comparison of disease-free survival rates. No significant difference in the disease-free rates was observed between the two groups (P = 0.78).
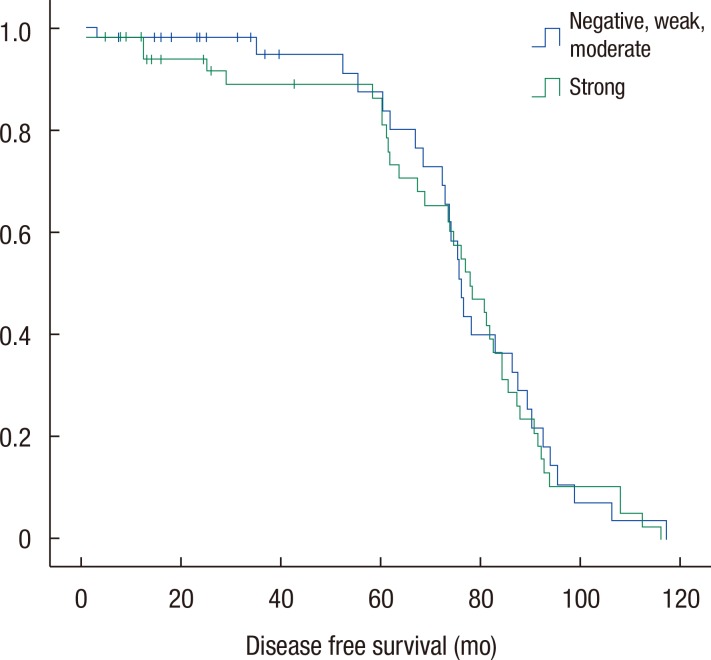
Fig. 4
Comparison of overall survival rates. No significant difference in the overall survival rate was observed between the two groups (P = 0.61).