- Search
Abstract
Purpose
The association between stomach cancer and colorectal cancer is controversial. The purpose of this study was to determine the synchronous prevalence of colorectal neoplasms in patients with stomach cancer.
Methods
A total of 123 patients with stomach cancer (86 male) and 246 consecutive, age- and sex-matched persons without stomach cancer were analyzed from July 2005 to June 2010. All of them underwent colonoscopy within 6 months after undergoing gastroscopy.
Results
The prevalence of colorectal neoplasms was significantly higher in the stomach cancer group (35.8%) than in the control group (17.9%) (P < 0.001). Colorectal neoplasms were more prevalent in the patients with stomach cancer (odds ratio [OR], 3.10; 95% confidence interval [CI], 1.71 to 5.63). In particular, the difference in the prevalence of colorectal neoplasms was more prominent in the patients above 50 years old (OR, 3.54; 95% CI, 1.80 to 6.98).
Conclusion
The results showed that the synchronous prevalence of colorectal neoplasms was higher in patients with stomach cancer than in those without stomach cancer. Therefore, patients with stomach cancer should be regarded as a high-risk group for colorectal neoplasms, and colonoscopy should be recommended for screening.
With the increasing aging population, the development of diagnostic techniques for cancer and the increase of environmental exposure to carcinogens, the prevalence of multiple primary cancers has been increasing [1]. In East Asia, stomach cancer with a relatively higher incidence rate compared to cancer in other organs shows a high rate of multiple primary cancers, and it has been reported to be associated with colorectal cancer in many cases [2]. Studies in South Korea also have reported that the incidence of multiple primary cancers was the highest in stomach cancer [3, 4], and a current research on the incidence of multiple primary cancers in stomach cancer patients surgically treated in South Korea found that colorectal cancer occurred most frequently [5]. However, there have been only a few studies on the synchronous prevalence of stomach cancer and colorectal cancer, so more studies on its causes, mechanisms and differences by region are needed.
According to statistics of the Korean Ministry of Health and Welfare in 2007, stomach cancer was the most common in the country, and colorectal cancer ranked third and fourth in males and females, respectively [6]. Thus, the prevalence of stomach cancer and colorectal cancer is high in South Korea, and the country manages the types of cancer through the national cancer screening program. For colorectal cancer, the guideline for early detection of colorectal cancer enacted in 2001 recommends colonoscopy screening test from the age of 50 years for adults with no symptoms [7]. However, there is no criterion for colonoscopy in stomach cancer patients.
For the synchronous prevalence of stomach cancer and colorectal cancer, while the prevalence of stomach cancer is known to be lower or similar in patients with colorectal cancer in the West [8-10], a higher prevalence of colorectal cancer in stomach cancer patients has been continuously reported in the East [11-13]. Thus, studies on the prevalence of stomach cancer and colorectal cancer in the East and the West have shown conflicting results, which is closely related with genetic and environmental factors, but there have been nearly no studies on the synchronous prevalence of stomach cancer and colorectal cancer and its screening test. Therefore, this study aims to investigate the association of colorectal neoplasms in stomach cancer patients compared to normal people by comparing the prevalence of colorectal neoplasms between persons with and without stomach cancer and to determine the usefulness of colonoscopy as a screening test to detect colorectal neoplasms in stomach cancer patients.
In this study, 123 patients, who were diagnosed as having stomach cancer through upper gastrointestinal endoscopy at Gyeongsang National University Hospital from July 2005 to June 2010 and who underwent colonoscopy within six months from the diagnosis, were included as the stomach cancer group. Among persons without stomach cancer in the upper gastrointestinal endoscopy for the same period and taking colonoscopy within six months, 246 persons matching the gender and the age distributions of the stomach cancer group were selected as the control group. In the stomach cancer and the control groups, persons with history of stomach or colorectal surgery, history of colonoscopy within the last five years or colonoscopic polypectomy, family history of colorectal cancer, infectious intestinal disease, or colon metastasis from stomach cancer and persons whose whole large intestine could not be observed in colonoscopy were excluded in this study.
With the subjects, their gender, age, diabetes, smoking, drinking, body mass index (BMI), administration of aspirin or non-steroid anti-inflammatory drugs (NSAIDs), findings of upper gastrointestinal endoscopy and pathological results, findings of colonoscopy and pathological results, and stomach cancer stage were retrospectively investigated with their medical record. Around 77.0 to 93.8% of the data could be obtained from the medical records.
Because gender, age, diabetes, smoking, drinking, BMI and the administration of aspirin or NASIDs can affect the incidence of stomach cancer and colorectal cancer, their correlations with stomach cancer and colorectal neoplasms were analyzed through a multiple regression analysis after revising them statistically in the stomach cancer and the control groups. The staging of stomach cancer was based on the American Joint Committee on Cancer (AJCC) cancer staging manual, sixth edition [14], and only histological stomach adenocarcinomas were investigated in this study.
Colorectal adenomas were divided into villous, tubulovillous and villous adenomas based on the results of the pathological examination and were classified into less than 5 mm, 5-9 mm and over 10 mm based on size. For the location of the colorectal neoplasm, the right colon, the left colon and the rectum were defined as from the caecum to the spleen flexure, from the descending colon to the sigmoid colon, and lower than the rectosigmoid junction, respectively. The number of colorectal neoplasms was divided into 0-3, 4-6, and 7 or more than 7.
All statistical analyses were conducted with SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA). The statistical significances of continuous and categorical variables were analyzed with the independent t-test and the chi-square test, respectively. To calculate the odds ratio of advanced colorectal neoplasms, a multiple regression analysis was utilized by revising drinking, smoking, BMI, diabetes and the administration of aspirin or NSAIDs with a 95% confidence interval. Statistical significance was determined as a P value of less than 0.05.
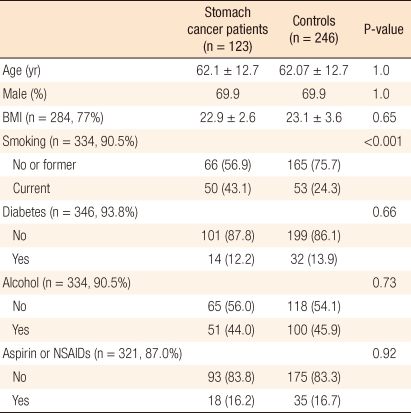
The mean age of the subjects was 62.1 ± 12.7 years, and males accounted for 69.9%. Because the subjects in the control group were selected by matching their gender and age to those of the stomach cancer group, there were no differences in gender and age between the two groups. The rate of smokers was significantly higher in the stomach cancer group than in the control group, being 43.1% (50/123) and 24.3% (53/246), respectively (P < 0.001). BMI, drinking, diabetes and the administration of aspirin or NSAIDs did not show any significant differences between the two groups (Table 1).
The prevalences of colorectal neoplasms were 35.8% (44/123) and 17.9% (44/246) in the stomach cancer and the control groups, respectively, and the prevalence in the stomach cancer group was significantly higher (Table 2). When a multiple regression analysis was performed by revising BMI, diabetes, smoking, drinking and the administration of aspirin or NSAIDs in the two groups, the prevalence of colorectal neoplasms was higher in the stomach cancer group than in the control group (odds ratio [OR], 3.10; 95% confidence interval [CI], 1.71 to 5.63). In particular, the odds ratio of the stomach cancer group to the control group was 3.54 in subjects aged over 50 years, so their prevalence of colorectal neoplasms was higher compared to that in subjects aged less than 50 years.
Comparison of the location of the colorectal neoplasm between the two groups found that the rate in the right colon (45.5%) was higher than that in the left colon (36.4%) in the stomach cancer group while the rates in both the right colon and the left colon were the same, 43.2%, in the control group.
For the size of a colorectal neoplasm, neoplasms with sizes of less than 5 mm, 5-9 mm, and 10 or more than 10 mm accounted for 47.7%, 27.3%, and 25.0%, respectively, in the stomach cancer group and 52.3%, 27.3%, and 20.5%, respectively, in the control group, but this difference was not statistically significant. The distribution of the number of colorectal neoplasms also was not significantly different as the rates of 0-3, 4-6, and 7 or more than 7 were 84.1%, 13.6%, and 52.3%, respectively, in the stomach cancer group and 90.9%, 6.8%, and 2.3%, respectively, in the control group. The pathological results for the colorectal neoplasms were villous, tubulovillous and villous adenoma and colorectal cancer in 79.5%, 2.3%, 11.4%, and 6.8% of the stomach cancer group, respectively, and in 86.4%, 6.8%, 6.8%, and 0.0% of the control group, but the difference between the two groups was not statistically significant.
The stage of stomach cancer was found to be stage 1, 2, 3, and 4 in 59 (51.8%), 21 (18.4%), 12 (10.5%), and 22 (19.3%) patients, respectively, and the prevalence of colorectal neoplasms in stage 1, 2, 3, and 4 showed no significant difference, being 35.6% (21/59), 23.8% (5/21), 25% (3/12), and 50% (11/22), respectively (Table 3).
The reported correlations between stomach cancer and colorectal cancer are inconsistent [15]. According to many studies in the West, the prevalence of stomach cancer was lower or similar in colorectal cancer patients than in the general population [8-10]. However, current research in Asian has reported that the prevalence of colorectal cancer is higher in stomach-cancer patients and studies conducted in South Korea have also found that the prevalence of colorectal cancer is higher in patients with stomach cancer [11, 12]. Therefore, the epidemiological correlation of the synchronous prevalence of colorectal cancer in stomach-cancer patients has not been determined clearly, and more studies are clearly necessary.
One hypothesis to explain the correlation between stomach cancer and colorectal cancer is their genetic correlation. APC, p53 and K-ras are already known to show the same genetic changes in stomach cancer and colorectal cancer [16-18]. Genes of hMSH2 and hMLH1 play a critical role in repairing the base pair mismatch found during DNA replication, and the changes of these genes, which are observed in 50% of hereditary nonpolyposis colorectal cancer patients, are important in the incidence of hereditary nonpolyposis colorectal cancer. In addition, as similar changes are shown in 10-15% of sporadic stomach cancer and colorectal cancer, the possibility of genetic mutation of hMSH2 and hMLH1 can be pointed to as a common genetic change in stomach cancer and colorectal cancer [19]. Microsatellite instability is caused by abnormality of the DNA mismatch repair pathway, and it is observed in 90% of hereditary nonpolyposis colorectal cancer patients, playing an important role in its incidence [20]. Moreover, microsatellite instability is shown in 18% of stomach cancer patients, 13-17% of colorectal cancer patients and 50% of multiple primary gastrointestinal cancer patients, so it is known to be closely related with the incidence of multiple primary gastrointestinal cancers [12, 19].
Another hypothesis is related with environmental factors, which means that factors of infectious disease, such as hyperglycemia, lipid foods, lack of fiber, obesity, smoking and JC virus (JCV) are at work in both stomach cancer and colorectal cancer patients [21-27]. Smoking was reported to be significantly related with malignant colorectal tumors with p53 expression and colorectal tumors with APC, K-ras and p53 transverse mutations [28]. In addition, p53 gene mutation was reported to be observed significantly more in smokers compared to non-smokers [29]. For obesity, the relative risks of colorectal cancer were high, being 1.5-2.0 and 1.2-1.5 in males and females, respectively, and a similar relation was found in colorectal adenomas. Another study said that obesity was related with an adenocarcinoma of the gastric cardia [30]. Obesity provokes hyperinsulinemia, and insulin is known to work as a growth factor in the large intestine so that hyperglycemia plays a role as a risk factor of colorectal cancer through this mechanism [30]. This study analyzed these environmental factors by reflecting them in the clinical characteristics of the two groups. While BMI, diabetes, drinking and the administration of aspirin or NSAIDs were not significantly different between the two groups, the rate of smokers was significantly higher in the stomach cancer group than in the control group. However, when the prevalences of colorectal neoplasms in the two groups were analyzed by revising these variables through a logistic regression analysis, they were found not to influence the results of this study. As a finding that viral infectious disease caused by JCV or one of the polyoma viruses could lead to stomach cancer and colorectal cancer has been reported, the possibility for an infectious disease like one caused by JCV to contribute to both stomach cancer and colorectal cancer is also suggested [25-27].
A colorectal adenoma is a pre-stage lesion of cancer. A study of Chung et al. [31] in South Korea reported that the prevalence of colorectal adenomas increased at older ages, being 10%, 22%, and 33% in the subjects in their 30s, 40s and 50s, respectively, and the overall incidence of colorectal adenomas is currently increasing. In this study, the prevalences of colorectal neoplasms were 35.8% and 17.9% in the stomach cancer and the control groups, respectively, and the prevalence for all subjects was not much different from that in previous studies (23.8%, 88/369) (Table 2). When colonoscopy was conducted within six months in stomach cancer patients, the prevalence of colorectal neoplasms was significantly higher compared to persons without stomach cancer, and the risk of colorectal neoplasms was higher in subjects aged over 50 years with an odds ratio of 3.54 (95% CI, 1.80 to 6.98) than in those aged less than 50 years. This finding emphasizes the necessity of colonoscopy screening test in persons over 50 years of age. In addition, the prevalence of villous adenomas, which is more likely to progress to colorectal cancer compared to villous adenomas, was higher in the stomach cancer group than in the control group, and the prevalence of colorectal cancer also tended to be higher in the stomach cancer group. That supports the necessity of screening through colonoscopy in stomach cancer patients.
In comparison with previous studies on the synchronous prevalence of stomach neoplasms and colorectal neoplasms performed in Korea [11, 12, 32], the prevalences of overall colorectal adenomas were similar, and the finding that the prevalence of colorectal adenomas was higher in the stomach neoplasm group was consistent except for some regions. However, the subjects of this study were different from those of other studies as most of the subjects in this study were from rural areas mainly in Western Gyeongnam. Their mean age was comparatively old, 62.1 ± 12.7 years, the rate of smokers in the stomach cancer group (43.1%) was much higher, and the prevalence of diabetes was higher (stomach cancer group: 12.2% vs. control group: 13.9%). Moreover, more fiber intake due to the diet, which following the rural geographical characteristics around Jiri mountain in western Gyeongnam, is also thought to have affected the incidence of colorectal neoplasms. However, genetic factors have not been studied until now, and this study did not investigate them either, so future studies on them are necessary. Although this study included more elderly population and showed a higher prevalence of diabetes and a higher rate of smokers compared to other studies, its results were not largely different from those of previous studies in South Korea because other factors, including fiber intake, exposure to carcinogens and genetic predispositions, work together in a complex way. The analysis under the hypothesis that the prevalence of colorectal neoplasms would go up at a higher stomach cancer stage, did not find any significance, which needs to be confirmed through more studies.
As limitations of this study, it was a retrospective study using medical records, and its subjects were limited to western Gyeongnam. Prospective studies in various regions with more cases are necessary.
Stomach cancer and colorectal cancer are common in South Korea, and screening tests for them have been performed as a part of the national cancer screening program. Although studies on the correlation between stomach cancer and colorectal cancer have been reported continuously and the awareness of the necessity of colonoscopy screening test in stomach cancer patients has increased, clear guidelines on it have not been suggested. This study found that the prevalence of colorectal neoplasms was higher in stomach cancer patients than in the control group, and this finding suggests that colonoscopy as a screening test for colorectal neoplasm is needed in patients diagnosed as having stomach cancer.
The results of studies on the synchronous prevalence of colorectal neoplasms in stomach cancer patients are conflicting and their correlation is controversial. Therefore, we conducted this study to determine that correlation and found that the prevalence of colorectal neoplasms was significantly higher in the stomach cancer group than in the control group. Thus, colonoscopy as a screening test for colorectal neoplasms is certainly considered to be necessary in stomach cancer patients.
References
1. Moertel CG, Dockerty MB, Baggenstoss AH. Multiple primary malignant neoplasms: I. Introduction and presentation of data. Cancer 1961;14:221–230. PMID: 13771652.


2. Okamoto N, Morio S, Inoue R, Akiyama K. The risk of a second primary cancer occurring in five-year survivors of an initial cancer. Jpn J Clin Oncol 1987;17:205–213. PMID: 3669364.

3. Park EK, Kwon JH, Lee JH, Lee SJ. Clinical study of multiple primary malignant tumor. Korean J Med 2005;69:410–418.
5. Hur YH, Ryu SY, Kim DY, Kim YJ, Kim SK. Study of combined multiple primary cancer in gastric cancer patients. J Korean Surg Soc 2003;64:296–301.
6. National Cancer Center, Ministry of Health and Welfare. Annual report of the Korea Central Cancer Registry 2007. 2008.Goyang: National Cancer Center.
7. Jung SY. Screening guidelines for the early detection of colorectal cancer: panel discussion. Korean J Gastrointest Endosc 2002;24:317–320.
8. Evans HS, Moller H, Robinson D, Lewis CM, Bell CM, Hodgson SV. The risk of subsequent primary cancers after colorectal cancer in southeast England. Gut 2002;50:647–652. PMID: 11950810.



9. Levi F, Randimbison L, Te VC, Rolland-Portal I, Franceschi S, La Vecchia C. Multiple primary cancers in the Vaud Cancer Registry, Switzerland, 1974-89. Br J Cancer 1993;67:391–395. PMID: 8431373.




10. Buiatti E, Crocetti E, Acciai S, Gafa L, Falcini F, Milandri C, et al. Incidence of second primary cancers in three Italian population-based cancer registries. Eur J Cancer 1997;33:1829–1834. PMID: 9470841.


11. Park DI, Park SH, Yoo TW, Kim HS, Yang SK, Byeon JS, et al. The prevalence of colorectal neoplasia in patients with gastric cancer: a Korean Association for the Study of Intestinal Disease (KASID) Study. J Clin Gastroenterol 2010;44:102–105. PMID: 19561531.


12. Oh SY, Park DI, Yoo TW, Kang MS, Kim SH, Park JH, et al. Is gastric cancer a new indication for surveillance colonoscopy? Colon cancer is increased in gastric cancer patients. Korean J Gastroenterol 2006;47:191–197. PMID: 16554672.

13. Watanabe S, Kodama T, Shimosato Y, Arimoto H, Suemasu K. Second primary cancers in patients with gastrointestinal cancers. Jpn J Clin Oncol 1985;15(Suppl 1): 171–182. PMID: 4009980.

14. Greene FL. American Joint Committee on Cancer. American Cancer Society. AJCC cancer staging manual. 2002.6th ed. New York: Springer-Verlag.
15. Engeland A, Bjorge T, Haldorsen T, Tretli S. Use of multiple primary cancers to indicate associations between smoking and cancer incidence: an analysis of 500,000 cancer cases diagnosed in Norway during 1953-93. Int J Cancer 1997;70:401–407. PMID: 9033646.


16. Uchino S, Tsuda H, Noguchi M, Yokota J, Terada M, Saito T, et al. Frequent loss of heterozygosity at the DCC locus in gastric cancer. Cancer Res 1992;52:3099–3102. PMID: 1591722.

17. Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M, et al. Genetic alterations during colorectal-tumor development. N Engl J Med 1988;319:525–532. PMID: 2841597.


18. Ohtani H, Yashiro M, Onoda N, Nishioka N, Kato Y, Yamamoto S, et al. Synchronous multiple primary gastrointestinal cancer exhibits frequent microsatellite instability. Int J Cancer 2000;86:678–683. PMID: 10797290.


19. Chung DC, Rustgi AK. DNA mismatch repair and cancer. Gastroenterology 1995;109:1685–1699. PMID: 7557155.


20. Schulmann K, Reiser M, Schmiegel W. Colonic cancer and polyps. Best Pract Res Clin Gastroenterol 2002;16:91–114. PMID: 11977931.


21. Cannon-Albright LA, Skolnick MH, Bishop DT, Lee RG, Burt RW. Common inheritance of susceptibility to colonic adenomatous polyps and associated colorectal cancers. N Engl J Med 1988;319:533–537. PMID: 2841598.


22. Grady WM. Genetic testing for high-risk colon cancer patients. Gastroenterology 2003;124:1574–1594. PMID: 12761718.


24. Chung YW, Han DS, Park YK, Son BK, Paik CH, Lee HL, et al. Association of obesity, serum glucose and lipids with the risk of advanced colorectal adenoma and cancer: a case-control study in Korea. Dig Liver Dis 2006;38:668–672. PMID: 16790371.


25. Shin SK, Li MS, Fuerst F, Hotchkiss E, Meyer R, Kim IT, et al. Oncogenic T-antigen of JC virus is present frequently in human gastric cancers. Cancer 2006;107:481–488. PMID: 16795066.


26. Jung WT, Li MS, Goel A, Boland CR. JC virus T-antigen expression in sporadic adenomatous polyps of the colon. Cancer 2008;112:1028–1036. PMID: 18205186.



27. Goel A, Li MS, Nagasaka T, Shin SK, Fuerst F, Ricciardiello L, et al. Association of JC virus T-antigen expression with the methylator phenotype in sporadic colorectal cancers. Gastroenterology 2006;130:1950–1961. PMID: 16762618.


28. Diergaarde B, Vrieling A, van Kraats AA, van Muijen GN, Kok FJ, Kampman E. Cigarette smoking and genetic alterations in sporadic colon carcinomas. Carcinogenesis 2003;24:565–571. PMID: 12663519.



29. Miyaki M, Iijima T, Ishii R, Kita Y, Koike M, Kuroki T, et al. Increased frequency of p53 mutation in sporadic colorectal cancer from cigarette smokers. Jpn J Clin Oncol 2002;32:196–201. PMID: 12110635.



30. Schoen RE, Tangen CM, Kuller LH, Burke GL, Cushman M, Tracy RP, et al. Increased blood glucose and insulin, body size, and incident colorectal cancer. J Natl Cancer Inst 1999;91:1147–1154. PMID: 10393723.



31. Chung SJ, Kim YS, Yang SY, Song JH, Park MJ, Kim JS, et al. Prevalence and risk of colorectal adenoma in asymptomatic Koreans aged 40-49 years undergoing screening colonoscopy. J Gastroenterol Hepatol 2010;25:519–525. PMID: 20370730.


32. Park SY, Kim HS, Yoon KW, Cho SB, Lee WS, Park CH, et al. Prevalence of colorectal adenoma is increased in patients with gastric adenoma. Korean J Gastroenterol 2009;54:220–226. PMID: 19844141.


- TOOLS
-
METRICS

- Related articles in ACP
-
The Synchronous Prevalence of Colorectal Neoplasms in Patients with Stomach Cancer2011 October;27(5)