- Search
| Ann Coloproctol > Volume 36(2); 2020 > Article |
|
Abstract
Nasal metastases from colorectal cancer is rare. The presentation of nasal metastases is often very similar to primary nasal sinus adenocarcinoma. A high index of suspicion is required, especially in patients who have had a previous history of colorectal carcinoma. Histology is ultimately required for diagnosis. We describe 2 cases of nasal metastases from colorectal carcinoma, and discuss the presentation, diagnosis and management of the case. Such metastatic disease ultimately represents end-stage malignancy, and patients should be palliated.
Metastases of colorectal cancer to the nasal cavity is rare, with few case reports available in the literature. The principle challenge is in differentiating metastatic disease to the nasal cavity from primary cancer. Due to the rarity of this condition, there is no consensus on the optimal management for such distant metastases. Hence, we describe our experience in the diagnosis and management of these rare cases.
Between 2010 and 2015, two cases of nasal metastases from colorectal cancer were managed in our hospital. Patients were identified through a prospectively collected colorectal cancer database. Only patients with histology proven metastases at the nasal region were included. Data collected included basic demographic information, symptoms on initial presentation, laboratory and radiological reports, intraoperative notes and clinical progress. Informed consent was not obtained as the patients had passed away at the time of writing this manuscript.
Patient A is a 46-year-old Malay woman who first presented with intestinal obstruction from sigmoid cancer in June 2008. Computed tomography (CT) scan of the abdomen and pelvis showed a stenosing lesion at the sigmoid colon and she underwent an open anterior resection. Histology was pT4N0. She then underwent eight cycles of adjuvant capecitabine/oxaliplatin.
On March 2011, she presented again with headache and vomiting and was found to have brain metastases and a nasal mass (Fig. 1). The nasal mass was biopsied, and the histopathology showed moderately differentiated intestinal-type adenocarcinoma (ITAC), consistent with metastatic disease from a colonic primary. It stained positive for CDX2, cytokeratin (CK) 20, Vilin and was negative for CK7 (Fig. 2). She was further administered with ten cycles of bevacizumab/capecitabine/irinotecan.
In spite of further chemotherapy, she developed progressive disease with tumor in the lungs as well as recurrent disease in the brain. She was palliated with 35 Gy of whole brain radiation therapy. She also developed recurrence in the nasal cavity and was treated with palliative radiotherapy for a month. She demised 74 months after initial diagnosis of colon cancer.
Patient B is a 64-year-old Chinese man who presented with a perforated intestinal obstruction in November 2012. CT abdomen and pelvis shows a mid-descending colon irregular circumferential lesion causing large bowel obstruction (Fig. 3). There were small volume local lymph nodes and retroperitoneal adenopathy. He underwent subtotal colectomy with ileorectal anastomosis. Histology showed poorly differentiated adenocarcinoma, pT3N2b. He was commenced on adjuvant chemotherapy with xeloda/oxaliplatin.
On the 6th cycle of oxaliplatin and capecitabine he was found to have metastases at the mid-ureter and bladder. He was then started on two cycles of folinic acid, fluorouracil and irinotecan before he had disease progression again and developed metastases to the bone, left adrenal, mesenteric lymph nodes, and liver.
His first presentation of nasal cavity metastases was with epistaxis on November 2013, where nasoendoscopy of the anterior and posterior nasal space showed a central mass, and histology showed multiple islands and small groups of epithelial cells, with many of the cells demonstrating pleomorphic and prominent nuclei. The infiltrating cells stained positive for broad-spectrum cytokeratin (AE1/3), CK20 and CDX2 and was negative for CK7 and p63 (Fig. 4). Before any treatment could be instituted, he passed away due to per rectal bleeding, acute on chronic kidney disease, recurrent falls and seizures. He passed away 11 months after the initial diagnosis of his colorectal cancer.
Metastases to the nasal cavity from colorectal carcinoma is rare. Primary tumors of the nasal cavity are far more common than such metastases. It is therefore important to distinguish between both conditions, though this may be challenging. Both primary malignancy and metastatic malignancy can present with recurrent epistaxis, nasal obstruction and facial pain [1,2]. Secondly, the histology of colonic metastases to the nasal cavity is indistinguishable from primary sinus adenocarcinomas—more specifically the colonic variant of ITAC [3].
Primary ITAC is a rare primary malignancy of the nasal cavity and paranasal sinuses. They may occur sporadically or as an occupation-related hazard, especially in cases of wood dust exposure [4]. However, the histology of ITAC-colonic variant is difficult to differentiate from a metastatic colorectal cancer [3]. Hence, a diagnosis of metastatic adenocarcinoma from the nasal cavity requires correlation with any prior history of colorectal cancer. Diagnosis may also be aided by immunohistochemical studies of the biopsy specimens. Positivity for CDX2, CK20 and negativity for CK7 differentiates metastatic colonic adenocarcinoma from ITAC [3,5]. The CK20+/CK7− immuniprofile is considered to be specific for colorectal epithelial tumors [6]. As was the case in both our patients, correlation between the histologic features of the nasal lesion with that of the previous colonic lesion was paramount in order to confirm the diagnosis of metastatic disease.
A possible mechanism of metastasizing to the nasal cavity was first postulated by Batson [7] in 1940, through the low-pressure valveless system connecting deep pelvic veins, intercostal veins, vena cava and the azygous system. An increase in the abdominal or thoracic pressure can cause a retrograde spread of the tumor cells through the vertebral venous plexus to the cerebrospinal venous system to reach the nasal cavity.
The development of nasal metastases from colorectal cancer is associated with poor outcomes, with none of the case reports surviving past the five-year mark from the diagnosis of the nasal metastases. All cases were treated with palliative radiotherapy to the nasal region [2,3,8].
In the case reports listed in our literature research, all the patients received palliative radiotherapy to the nasal region, and they passed away between 2 to 18 months after diagnosis of the metastases [3,8]. These results are similar to our findings as well. Differentiating between the two is thus important—colorectal metastases to the nasal cavity is usually associated with disseminated metastases, poor prognosis and should be managed palliatively, while primary adenocarcinomas of the nasal cavity have a much better prognosis and could be treated with curative intent.
In conclusion, nasal metastasis is a rare condition that needs to be differentiated from ITAC. The presence of nasal metastases, unlike ITAC, represents advanced disease and carries a poor prognosis. Such patients should be palliated.
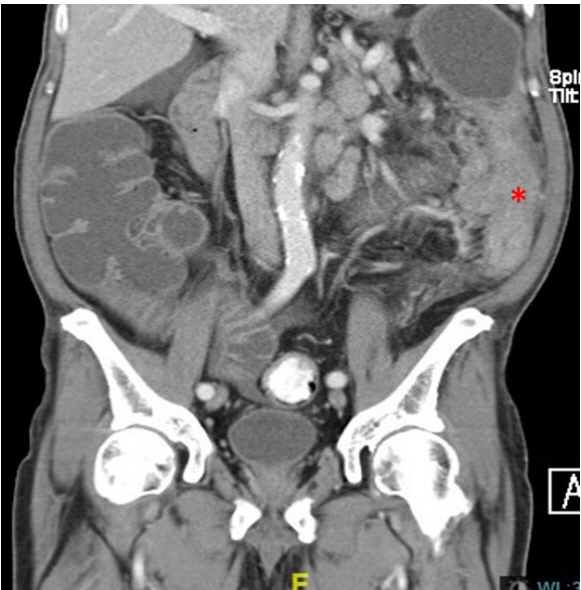
Fig. 1.
Coronal section of a magnetic resonance imaging demonstrating presence of the left nasal mass (asterisk). RPF, right, posterior, foot; LAH, left, anterior, head.

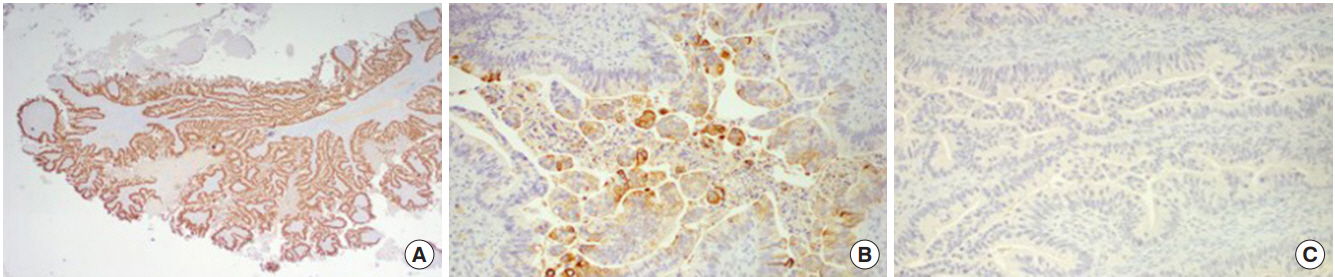
Fig. 2.
Case 1: immunohistochemically the malignant cells were positive for CDX2 (A), positive for cytokeratin (CK) 20 (B), and negative for CK7 (C).
REFERENCES
1. Azarpira N, Ashraf MJ, Khademi B, Asadi N. Distant metastases to nasal cavities and paranasal sinuses case series. Indian J Otolaryngol Head Neck Surg 2011;63:349–52.




2. Lopez F, Devaney KO, Hanna EY, Rinaldo A, Ferlito A. Metastases to nasal cavity and paranasal sinuses. Head Neck 2016;38:1847–54.


3. bin Sabir Husin Athar PP, bte Ahmad Norhan N, bin Saim L, bin Md Rose I, bte Ramli R. Metastasis to the sinonasal tract from sigmoid colon adenocarcinoma. Ann Acad Med Singapore 2008;37:788–90.


4. Barnes L. Intestinal-type adenocarcinoma of the nasal cavity and paranasal sinuses. Am J Surg Pathol 1986;10:192–202.


5. Bien S, Kaminski B, Okla S, Kopczynski J. Metastasis of rectal adenocarcinoma to the skull base and paranasal sinuses, with unusual clinical symptoms. Otolaryngol Pol 2005;59:627–30.

6. Kende AI, Carr NJ, Sobin LH. Expression of cytokeratins 7 and 20 in carcinomas of the gastrointestinal tract. Histopathology 2003;42:137–40.