- Search
| Ann Coloproctol > Volume 39(5); 2023 > Article |
|
Abstract
Purpose
This study was performed to investigate the convergent validity, discriminative validity, and reliability of the Brazilian version of the low anterior resection syndrome (LARS) score in a population with low educational and socioeconomic levels.
Methods
The LARS score was translated into the Portuguese language by forward- and back-translation procedures. In total, 127 patients from a public hospital in Brazil completed the questionnaires. The convergent validity was tested by comparing the LARS score with the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire Core Module 30 (QLQ-C30) and with patients’ self-reported quality of life. For the discriminative validity, we tested the ability of the score to differentiate among subgroups of patients regarding neoadjuvant radiotherapy, type of surgery, and tumor distance from the anal verge. The test-retest reliability was investigated in a subgroup of 36 patients who responded to the survey twice in 2 weeks.
Results
The LARS score demonstrated a strong correlation with 5 of 6 items from the EORTC QLQ-C30 (P<0.05) and good concordance with patients’ self-reported quality of life (95.3%), confirming the convergent validity. The score was able to discriminate between subgroups of patients with different clinical characteristics related to LARS (P<0.001). The agreement between the test and retest showed that 86.1% of the patients remained in the same LARS category, and there was no significant difference between the LARS score numerical values (P=0.80), indicating good reliability overall.
Colorectal cancer is one of the most frequent types of cancer in the Brazilian population. In women, it is the second most common, with 17,620 cases estimated for 2016; in men, it occupies the third position, with 16,660 new cases [1].
Resection of the rectum with an anastomosis is still the gold standard curative operation for tumors without involvement of the anal sphincter. Tumors of the lower and middle part of rectum can be treated by low anterior resection (LAR) with sphincter preservation following the total mesorectal excision (TME) principles whereas in tumors of the upper part of rectum (10–15 cm from the anal verge), we can perform a partial mesorectal excision (PME) [2–4].
Advances in diagnosis, staging systems, and treatment have led to better oncological outcomes after rectal cancer treatment and increased the population of long-term survivors in recent decades. Unfortunately, many patients complain of disordered bowel function after surgery known as the LAR syndrome (LARS) resulting in quality of life (QoL) deterioration [5–7]. Up to 90% of patients develop LARS symptoms in the postoperative period, which are characterized by frequent bowel movements, evacuatory urgency, incontinence for flatus or feces, and stool fragmentation and clustering [8, 9].
In 2012, Emmertsen and Laurberg [10] developed and validated the LARS score, a symptom-based scoring system assessing bowel dysfunction after rectal cancer resection from a patient’s perspective. Since its publication, the LARS score has been translated and validated in many languages worldwide and became an important common international instrument in clinical trials for LARS studies. But it has not yet been validated in Brazil or in a population with a low educational and economic profile [9, 11–13].
Validated acceptable measurement tools are crucial in international research and clinical settings and should be applicable across different countries despite cultural and socioeconomic discrepancies. Although LARS score is already considered an international instrument, its validation in a poor population from South America will confirm its safe applicability in most various populations.
The aim of this study was to investigate convergent and discriminative validity and the test-retest reliability of the Brazilian translated version (Portuguese language) of LARS score. This validation study will add a very important angle by testing the LARS score in a population with a low educational and socioeconomic profile.
This study was approved by the Ethics Committee of the Federal University of Minas Gerais under the protocol CAAE-14500613. 9.0000.5149. The patients were included in this study after having signed the informed consent form.
We conducted the forward and back-translation procedures according to recommendations from the World Health Organization (WHO) and the European Organization for Research and Treatment of Cancer (EORTC) to guarantee the semantic equivalence [14, 15]. Two native Brazilian translators fluent in English translated the English version of LARS score to Portuguese and a single common version was obtained after discussion and consensus [11]. Then, this Portuguese version was translated back to English by another professional translator whose mother tongue was English. The back translation was compared with the original score to identify semantic discrepancies.
Inclusion criteria were age 18 years or older, rectal adenocarcinoma between 0 and 15 cm from the anal verge, and a restorative resection of the rectum (TME or PME) with a functional anastomosis. Exclusion criteria were local recurrence or distant metastasis, patients receiving adjuvant chemotherapy at the time of the study, other bowel diseases, or presence of an intestinal stoma. A temporary stoma should have been reversed at least 3 months prior to the inclusion in this study.
Due to the low educational level with a relatively high rate of illiterate patients, and the historically low response rates in postal surveys in Brazil, the eligible participants were invited for an appointment at the outpatient clinic at the hospital to be interviewed for the study. The EORTC Quality of Life Questionnaire Core Module 30 ver. 3.0 (QLQ-C30) and the LARS score were filled in in collaboration with the investigator during the interview. Patients were systematically asked during the interview about any difficulty interpreting or understanding the meaning of LARS score questions.
Sociodemographic and economical status were obtained from patient’s report at the time of the interview and clinical information was verified from local hospital databases.
The validation of the Brazilian version of LARS score was obtained by evaluating the convergent validity, discriminant validity, and the test-retest reliability [16].
In order to investigate the convergent validity, the LARS score was compared to the global QoL item and the 5 functional subscales of the EORTC QLQ-C30, which is a widely used and validated tool to measure QoL in cancer patients. We expected negative correlations between the LARS score and the QLQ-C30 global QoL, physical, emotional, cognitive, social, and role functioning subscales.
To test the LARS score’s convergent validity further, an additional question was asked to assess patient’s QoL as done in previous LARS score validation studies: “How much does your bowel function affects your quality of life?” There were 4 response options: “not at all,” “a little,” “some,” and “a lot,” which were categorized into 3 groups similar to LARS score to investigate the agreement between them [11]. The self-reported QoL groups and LARS score corresponding categories are as follows: no impact (“not at all”), no LARS; minor impact (“a little”), minor LARS; and some or major impact (“some or a lot”), major LARS.
The agreement was considered perfect when the QoL category matched the LARS category; moderate agreement was defined when we had difference by one category; and finally, no agreement was defined when LARS score and the impact on QoL differed by 2 categories, for example having no LARS and some or major impact on QoL.
For discriminant validity, we investigated the ability of the LARS score to differentiate between groups of patients. It has previously been shown that low tumor level, radiotherapy, and TME as opposed to PME is associated with poor functional outcome [5, 12, 17]. Thus the groups were based on tumor level (above 5 cm vs. below 5 cm from the anal verge), type of surgery (TME vs. PME) and preoperative radiotherapy (yes vs. no) [5, 10].
To investigate the test-retest reliability a randomly selected subgroup of 40 participants was invited to complete the questionnaires twice by the same interviewer (2 weeks after the first interview). Thirty six of them agreed to come back and participated.
Statistical analyses were performed using SPSS ver. 16.0 (SPSS Inc). Descriptive statistics were used to assess clinical and demographic variables that were shown as mean±standard deviation, median with interquartile range (IQR), numerical values, and percentages.
To assess the convergent validity, the EORTC QLQ-C30 data was presented as median and IQR and the difference in LARS groups was tested by the Kruskal-Wallis test. The association between LARS score and the categories according to self-reported bowel function on QoL was also tested by the Kruskal-Wallis test.
The nonparametric Mann-Whitney test was used for hypothesis testing of the discriminative validity and a box and whisker plot analysis was used to illustrate the LARS score values.
The test-retest reliability of LARS score and its category was evaluated by assessing the proportions with 95% confidence intervals (CIs) of agreements (perfect, moderate, or no agreement) between the first and second tests. The LARS score values in the test and retest were illustrated by a Bland-Altman plot and the correlation between them were also evaluated by the intraclass correlation coefficient (ICC). ICC values of ≥0.8 are traditionally considered adequate according to previous validation studies of LARS scores [11, 18].
A P-value of <0.05 was considered statistically significant.
The double forward translations and the backward translation confirmed the original meaning of each question of LARS score. The back translation was checked and accepted by the investigators and there were no semantic discrepancies, which led to the final Portuguese version of the score (Fig. 1).
We had 217 potentially eligible patients operated from 2000 to 2014 and identified through local databases at a tertiary public hospital in Brazil (Hospital of Clinics of the Federal University of Minas Gerais). However, 127 patients could be contacted and answered the questionnaires. Among the 90 patients who did not attend to answer the questionnaire, 22 had died, 6 refused to participate, and 62 patients could not be contacted. Clinical information is shown in Table 1.
Regarding the level of education, 66 patients (52.0%) did not complete elementary school which is 8 years of study in Brazil, and 31 (24.4%) stopped studying during high school which is 3 years of study after elementary school. Sixteen patients (12.6%) were illiterate and could not read. The family monthly income was up to Brazilian minimum salary in 25 participants (19.7%), which corresponds to about 251 US dollars. Data regarding educational and economic status is shown in Table 2 [19].
No patient reported difficulty interpreting and understanding the meaning of LARS score questions despite their socioeconomic and educational status.
The EORTC QLQ-C30 results are shown in Table 3. The LARS score had statistically significant negative correlations with global QoL item (P<0.001), physical function (P=0.015), role function (P=0.001), emotional function (P=0.011), and social function (P<0.001) subscales from EORTC QLQ-C30. The P-value was 0.057 for the cognitive function.
Regarding the proportion of agreement between QoL category and LARS score category, we had perfect agreement in 70.9% (95% CI, 63.0%–78.0%), moderate agreement in 24.4% (95% CI, 17.0%–32.0%), and only 4.7% (95% CI, 1.0%–8.4%) of the participants had no agreement (Table 4). Boxplots in Fig. 2 show the numerical value of LARS score and the QoL category. A higher LARS score has more impact of bowel function on QoL (P<0.001).
The ability of LARS score to differentiate among different groups of patients regarding bowel function was tested to assess the discriminative validity of LARS score (Fig. 3). A statistically significant difference (P<0.001) was found comparing patients with TME and PME; with and without neoadjuvant radiotherapy and patients with tumors of >5 and ≤5 cm from the anal verge. Groups treated with TME, those who underwent radiotherapy and those with tumors of ≤5 cm from the anal verge had worse bowel functional results (higher LARS score values).
A total of 36 participants completed the questionnaire twice in a 2-week interval to verify the reproducibility. The agreement between the first and second replies for each LARS score question is shown in Table 5.
The differences between the first and second test values are illustrated in Fig. 4 by means of a Bland-Altman plot. The mean difference was 0.2, the 95% limits of agreement were –10.1 to 10.5 and there was no significant difference between the test and retest values of the score (P=0.80). The ICC of LARS score was 0.94 (95% CI, 0.83–0.96) indicating adequate reliability.
This is the first study to show that the recently developed LARS score is a valid and reliable tool for measuring LARS in a population with a low educational level and a high proportion of illiterates and emphasizes the importance of using standardized translation and validation procedures before applying the questionnaire internationally.
An objective and effective evaluation of bowel function after rectal cancer surgery is currently considered mandatory due to the high prevalence of LARS symptoms and its detriment to QoL. Patients should be informed preoperatively about the risk of postoperative pelvic dysfunction including bowel, urinary and sexual problems leading to physical and social impairment [10, 20].
In order to have a common international scientific instrument for research and clinical practice on LARS, the use of specific and validated questionnaires was proposed instead of other scores that are time-consuming for calculating, completing, and interpreting by patients. The validated LARS score offers an easy, quick, and accurate tool that could be applied to various populations worldwide despite cultural differences [21].
In this validation procedure, results showed that 76.4% of the 127 patients did not complete high school and almost 20% of them had a family monthly income up to one Brazilian minimum salary, which is considered by the government the minimum amount of money for someone to live in the country. These profiles of poor and low educational patients represent the largest part of the population in Brazil and are predominant in Brazilian public hospitals since they usually cannot afford private health insurance [19]. This study validated LARS score for the Brazilian population after translation to Portuguese language and showed good psychometric properties regarding convergent validity, discriminative validity, and reliability.
The cognitive function was the only EORTC QLQ-C30 functional scale that was not statistically associated with the Brazilian version of LARS score (P=0.057). Considering the physiological changes that lead to LARS impact on QoL we previously expected less influence on the cognitive function compared with emotional and social functions for example. However, Emmertsen et al. [22] had shown minor but clinically relevant difference in cognitive function and LARS after 1-year of follow-up. Juul et al. [7] also demonstrated this association after at least 16 months past LAR for rectal cancer. These results suggest that bowel function may over time decrease both memory and concentration affecting cognitive function later than physical, role, emotional, and social functions.
LARS score categories had 70.9% of perfect agreement with the self-reported QoL categories and only 4.7% of no agreement confirming its convergent validity and correlation with QoL, as reported in other validation studies [11, 13].
Regarding the discriminative validity, the LARS score was demonstrated to be an adequate tool to identify subgroups of patients previously known to have worse functional outcomes based on specific clinical variables. Subgroups of participants who underwent TME surgery, neoadjuvant radiotherapy, and those with tumors located up to 5 cm from the anal verge had higher values of LARS scores (P<0.001). Previous studies have shown that the closer the tumor is to the anal verge the higher is the risk of LARS [23, 24].
The test-retest reliability had an excellent ICC (0.92) and very low proportion of no agreement between the first and second responses of LARS category and LARS 5 questions (0% to 5.5%). There was no statistical difference (P=0.80) between the numerical score values in the test and retest proving that the Brazilian version of LARS score is a stable and reproducible measure instrument.
Since the results of this study are very similar to previous LARS score validations in wealthy societies, this study contributes to the widespread of LARS score irrespective of educational and socioeconomic levels discrepancies [9, 11–13].
The limitation of this study was that 90 out of 217 eligible patients did not attend the survey and were not included. Most of them (62 patients) are probably no longer in clinical follow-up and could not be contacted due to the lack of updated phone numbers or other means of contact on hospital databases. This low attendance rate is probably a proxy for a population with low sociodemographic and economic profile. Moreover, the fact that an interviewer involved in the survey filled out questionnaires on behalf of the patient may also lead to bias in data collection.
The results of this validation of the Brazilian version of the LARS score only apply when used face-to-face with the patients as was done in this study and not if the score is sent out as a mailed questionnaire for self-completion by the patients. This new LARS score validation clearly emphasizes the importance of using standardized translation and validation procedures before applying a questionnaire to each population with different cultural and socioeconomic characteristics [25].
The definition of LARS and the development of standardized scores for bowel dysfunction were the first step to enabling new studies aiming at the identification of patients at higher risk for LARS. An example is the preoperative LARS score which was recently published and is the first nomogram and online tool to predict bowel dysfunction severity preoperatively [17, 26, 27].
In conclusion, the Brazilian version of LARS score has good psychometric properties regarding convergent validity, discriminative validity, and reliability when applied face-to-face to a low-level educational and socioeconomic population of rectal cancer survivors in Brazil.
Fig. 1.
The Brazilian version (Portuguese language) of low anterior resection syndrome score (LARS).

Fig. 2.
Association between low anterior resection syndrome (LARS) score and impact of self-reported bowel function on quality of life. *P<0.001 (Kruskal-Wallis test).
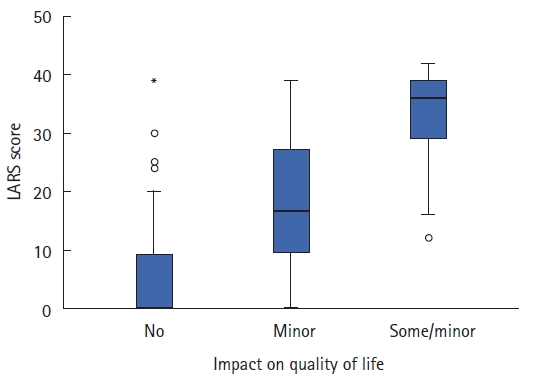
Fig. 3.
Association between low anterior resection syndrome (LARS) score and clinical subgroups. (A) Type of surgery (partial mesorectal excision [PME] or total mesorectal excision [TME]). (B) Neoadjuvant radiotherapy (no or yes). (C) Tumor level ( ≤5 or >5 cm of distance from the anal verge). P<0.001 (Mann-Whitney test).
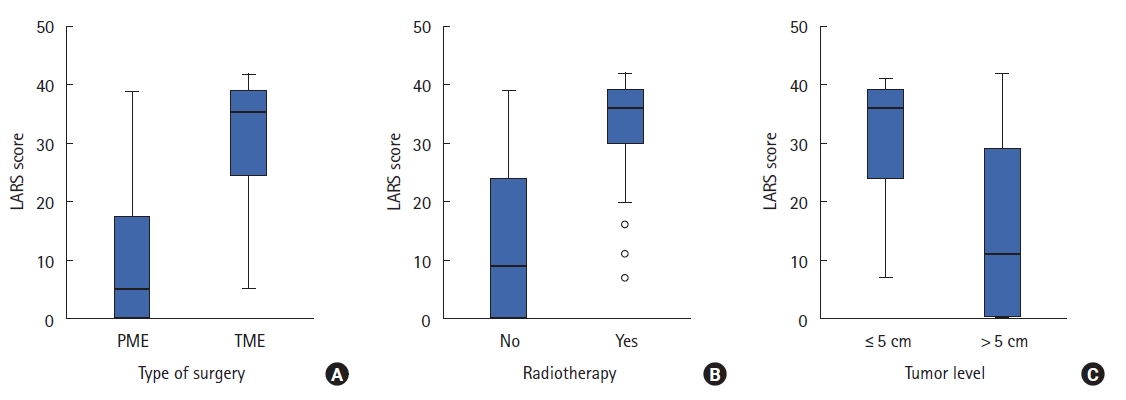
Fig. 4.
Bland-Altman plot illustrating the difference between low anterior resection syndrome (LARS) scores at the first and second test (n=36). SD, standard deviation.
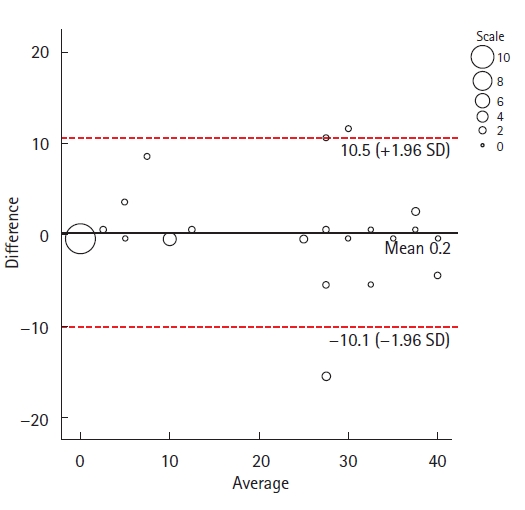
Table 1.
Clinical characteristics of patients (n=127)
Table 2.
Educational and economic features (n=127)
| Variable | No. of patients (%) |
|---|---|
| Literate | |
| Yes | 111 (87.4) |
| No | 16 (12.6) |
| Level of education | |
| Unschooled or incomplete elementary school | 66 (52.0) |
| Complete elementary school or incomplete high school | 31 (24.4) |
| Complete high school or incomplete college | 21 (16.5) |
| Complete college or higher education | 9 (7.1) |
| Family monthly incomea | |
| Up to 1 salary | 25 (19.7) |
| More than 1 and up to 5 salaries | 88 (69.3) |
| More than 5 and up to 10 salaries | 13 (10.2) |
| More than 10 salaries | 1 (0.8) |
Table 3.
The EORTC QLQ-C30 score results (global QoL item and functional scales) by LARS groups (n=127)
| Scale |
Score |
P-valuea | ||
|---|---|---|---|---|
| No LARS | Minor LARS | Major LARS | ||
| Global QoL scale | 91.7 (25.0) | 75.0 (25.0) | 66.7 (33.3) | <0.001 |
| Physical function | 100.0 (0) | 100.0 (20.0) | 100.0 (8.3) | 0.015 |
| Role function | 100.0 (0) | 100.0 (33.3) | 100.0 (33.3) | 0.001 |
| Emotional function | 91.7 (16.7) | 91.7 (29.2) | 79.2 (35.9) | 0.011 |
| Cognitive function | 100.0 (16.7) | 100.0 (25.0) | 83.3 (33.3) | 0.057 |
| Social function | 100.0 (0) | 100.0 (16.7) | 100.0 (33.3) | <0.001 |
Table 4.
Fit between LARS category and quality of life category (n=127)
| LARS category |
Quality of lifea |
|||
|---|---|---|---|---|
| No impact | Minor impact | Some or major impact | Total | |
| No LARS | 49 (38.6) | 15 (11.8) | 4 (3.1) | 68 (53.5) |
| Minor LARS | 2 (1.6) | 5 (3.9) | 10 (7.9) | 17 (13.4) |
| Major LARS | 2 (1.6) | 4 (3.1) | 36 (28.3) | 42 (33.1) |
| Total | 53 (41.7) | 24 (18.9) | 50 (39.4) | 127 (100) |
Table 5.
Agreement between first and second responses for LARS category (no, minor, or major) and for the 5 questions of LARS score (n=36)
REFERENCES
1. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA); Ministério Da Saúdea. Estimativa 2016: Incidência do Câncer no Brasil. INCA; 2016 [cited 2017 Jan 20]. Available from: https://www.ibge.gov.br
2. Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery: the clue to pelvic recurrence? Br J Surg 1982;69:613–6.



3. Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1986;1:1479–82.


4. Kanso F, Lefevre JH, Svrcek M, Chafai N, Parc Y, Tiret E. Partial mesorectal excision for rectal adenocarcinoma: morbidity and oncological outcome. Clin Colorectal Cancer 2016;15:82–90.


5. Bregendahl S, Emmertsen KJ, Lous J, Laurberg S. Bowel dysfunction after low anterior resection with and without neoadjuvant therapy for rectal cancer: a population-based cross-sectional study. Colorectal Dis 2013;15:1130–9.


6. Chen TY, Wiltink LM, Nout RA, Meershoek-Klein Kranenbarg E, Laurberg S, Marijnen CA, et al. Bowel function 14 years after preoperative short-course radiotherapy and total mesorectal excision for rectal cancer: report of a multicenter randomized trial. Clin Colorectal Cancer 2015;14:106–14.


7. Juul T, Ahlberg M, Biondo S, Espin E, Jimenez LM, Matzel KE, et al. Low anterior resection syndrome and quality of life: an international multicenter study. Dis Colon Rectum 2014;57:585–91.


8. Sturiale A, Martellucci J, Zurli L, Vaccaro C, Brusciano L, Limongelli P, et al. Long-term functional follow-up after anterior rectal resection for cancer. Int J Colorectal Dis 2017;32:83–8.



9. Samalavicius NE, Dulskas A, Lasinskas M, Smailyte G. Validity and reliability of a Lithuanian version of low anterior resection syndrome score. Tech Coloproctol 2016;20:215–20.



10. Emmertsen KJ, Laurberg S. Low anterior resection syndrome score: development and validation of a symptom-based scoring system for bowel dysfunction after low anterior resection for rectal cancer. Ann Surg 2012;255:922–8.


11. Juul T, Ahlberg M, Biondo S, Emmertsen KJ, Espin E, Jimenez LM, et al. International validation of the low anterior resection syndrome score. Ann Surg 2014;259:728–34.


12. Hou XT, Pang D, Lu Q, Yang P, Jin SL, Zhou YJ, et al. Validation of the Chinese version of the low anterior resection syndrome score for measuring bowel dysfunction after sphincter-preserving surgery among rectal cancer patients. Eur J Oncol Nurs 2015;19:495–501.


13. Juul T, Battersby NJ, Christensen P, Janjua AZ, Branagan G, Laurberg S, et al. Validation of the English translation of the low anterior resection syndrome score. Colorectal Dis 2015;17:908–16.


14. Dewolf L, Koller M, Velikova G, Johnson C, Scott N, Bottomley A. Eortc Quality of Life Group translation procedure 2009. 3rd ed. EORTC Quality of Life Group; 2009.
15. World Health Organization (WHO). WHO guidelines on translation. Process of translation and adaptation of instruments 2012 [Internet]. WHO; 2012 [cited 2017 Jan 20]. Available from: http://www.who.int/substance_abuse/research_tools/translation/en/
16. Cook DA, Beckman TJ. Current concepts in validity and reliability for psychometric instruments: theory and application. Am J Med 2006;119:166.

17. Battersby NJ, Bouliotis G, Emmertsen KJ, Juul T, Glynne-Jones R, Branagan G, et al. Development and external validation of a nomogram and online tool to predict bowel dysfunction following restorative rectal cancer resection: the POLARS score. Gut 2018;67:688–96.


18. Zidan M, Thomas RL, Slovis TL. What you need to know about statistics, part II: reliability of diagnostic and screening tests. Pediatr Radiol 2015;45:317–28.



19. Instituto Brasileiro de Geografia e Estatística (IBGE). Censo demográfico 2010 [Internet]. IBGE; 2010 [cited 2017 Jan 20]. Available from: http://www.ibge.gov.br/home/estatistica/populacao/indic_sociosaude/2009/default.shtm
20. Emmertsen KJ, Chen TY, Laurberg S. Functional results after treatment for rectal cancer. J Coloproctol 2014;34:55–61.

21. Chen TY, Emmertsen KJ, Laurberg S. What are the best questionnaires to capture anorectal function after surgery in rectal cancer? Curr Colorectal Cancer Rep 2015;11:37–43.




22. Emmertsen KJ, Laurberg S, Rectal Cancer Function Study Group. Impact of bowel dysfunction on quality of life after sphincter-preserving resection for rectal cancer. Br J Surg 2013;100:1377–87.



23. Digennaro R, Tondo M, Cuccia F, Giannini I, Pezzolla F, Rinaldi M, et al. Coloanal anastomosis or abdominoperineal resection for very low rectal cancer: what will benefit, the surgeon’s pride or the patient’s quality of life? Int J Colorectal Dis 2013;28:949–57.



24. Battersby NJ, Juul T, Christensen P, Janjua AZ, Branagan G, Emmertsen KJ, et al. Predicting the risk of bowel-related quality-of-life impairment after restorative resection for rectal cancer: a multicenter cross-sectional study. Dis Colon Rectum 2016;59:270–80.


25. Kim CW, Jeong WK, Son GM, Kim IY, Park JW, Jeong SY, et al. Validation of Korean version of Low Anterior Resection Syndrome Score Questionnaire. Ann Coloproctol 2020;36:83–7.




- TOOLS
-
METRICS

- Related articles in ACP
-
Low anterior resection syndrome: is it predictable?2023 October;39(5)
Validation of the Vietnamese version of the low anterior resection syndrome score questionnaire ;0()
Validation of Korean Version of Low Anterior Resection Syndrome Score Questionnaire 2020 April;36(2)







