Early Systemic Failure After Preoperative Chemoradiotherapy for the Treatment of Patients With Rectal Cancer
Article information
Abstract
Purpose
Distant metastasis can occur early after neoadjuvant chemoradiotherapy (CRT) in patients with rectal cancer. This study was conducted to evaluate the clinical characteristics of patients who developed early systemic failure.
Methods
The patients who underwent neoadjuvant CRT for a rectal adenocarcinoma between June 2007 and July 2015 were included in this study. Patients who developed distant metastasis within 6 months after CRT were identified. We compared short- and long-term clinicopathologic outcomes of patients in the early failure (EF) group with those of patients in the control group.
Results
Of 107 patients who underwent neoadjuvant CRT for rectal cancer, 7 developed early systemic failure. The lung was the most common metastatic site. In the EF group, preoperative carcinoembryonic antigen was higher (5 mg/mL vs. 2 mg/mL, P = 0.010), and capecitabine as a sensitizer of CRT was used more frequently (28.6% vs. 3%, P = 0.002). Of the 7 patients in the EF group, only 4 underwent a primary tumor resection (57.1%), in contrast to the 100% resection rate in the control group (P < 0.001). In terms of pathologic outcomes, ypN and TNM stages were more advanced in the EF group (P < 0.001 and P = 0.047, respectively), and numbers of positive and retrieved lymph nodes were much higher (P < 0.001 and P = 0.027, respectively).
Conclusion
Although early distant metastasis after CRT for rectal cancer is very rare, patients who developed early metastasis showed a poor nodal response with a low primary tumor resection rate and poor oncologic outcomes.
INTRODUCTION
Over the last several decades, preoperative chemoradiotherapy (CRT) has been accepted as a standard treatment for patients with locally advanced rectal cancer because it can reduce local recurrence postoperatively and enable sphincter-saving surgery in some patients who had been ineligible for the procedure before CRT [1, 2]. National Comprehensive Cancer Network guidelines recommend preoperative radiotherapy or CRT for all patients with rectal cancer suspected to be greater than stage T3 or with lymph node (LN) metastasis on preoperative evaluation [3].
Concern for failure of systemic control of the disease during CRT remains an issue in the treatment of patients with cancer because radiotherapy only involves local treatment of the disease and rectal cancer in patients who are eligible for preoperative radiotherapy is locally advanced. Moreover, a long period of about three to four months is typical between diagnosis and surgery. Thus, many other chemotherapy regimens added to fluorouracil-based CRT have been suggested in an effort to enhance systemic control during CRT or within the window from radiotherapy to surgery [4, 5].
Nevertheless, reports of early distant metastasis during or after preoperative CRT are rare. For that reason, we conducted this study to evaluate the incidence and the clinical significance of early distant metastasis after preoperative CRT in patients with rectal cancer and to provide data that will be useful in determining the evaluation or follow-up protocol for patients whose risk for early distant metastasis is high.
METHODS
Patient selection and classification
The medical records of patients who had undergone long-course CRT with neoadjuvant intention for rectal cancer at Korea University Anam Hospital, Seoul, Korea, between June 2007 and July 2015, were retrospectively reviewed. This study was approved by the Institutional Review Board of Korea University Anam Hospital (IRB No. 2018AN0158), and all patients provided written informed consent. Patients who had primary rectal cancer with a pathologically proven adenocarcinoma within 15 cm of the anal verge, subdivided as lower (<5 cm), mid (5–10 cm), and upper (>10 cm) rectum, were enrolled in this study. Patients who developed distant metastasis within 6 months after completion of CRT were assigned to the early systemic failure group (the EF group). All other patients were assigned to the control group.
Preoperative evaluation and preoperative CRT
All patients were evaluated preoperatively by using physical examination, total colonoscopy, abdominopelvic computed tomography (CT), chest CT, pelvic magnetic resonance imaging, and routine laboratory tests including tumor markers. Based on these studies, patients without distant metastasis were considered for preoperative CRT.
Preoperative CRT has been performed selectively at our institution; patients with resectable locally advanced rectal cancer including T3 stage and patients with LNs suggesting metastasis including the total mesorectal excision (TME) plane were considered for surgery without preoperative CRT [6]. The indications for preoperative CRT at our hospital were as follows: T4, circumferential resection margin involved or threatened, or suggestive metastasis of lateral pelvic LN, defined as a LN beyond the TME plane such as iliac and obturator LNs, on preoperative staging. Exclusion criteria in this study were as follows: radiotherapy for anal cancer or for recurrent rectal cancer; preoperative short-course radiotherapy or postoperative radiotherapy; initial distant metastasis; no restaging work-up after CRT preoperatively or no appropriate follow-up; or preoperative exposure to chemotherapy regimens other than 5-fluorouracil (5-FU) or capecitabine as a radiosensitizer, such as with oxaliplatin, irinotecan, or a target agent.
Patients were treated with neoadjuvant chemoradiation (5,040 cGy in 28 fractions) for 5–6 weeks, with intravenous or oral 5-FU (tegafur) or oral capecitabine as a radiosensitizer. Each patient was evaluated by restaging 8–10 weeks later for final decision; then, surgery was performed in most patients unless certain problems, such as distant metastasis or a locally refractory response, developed.
Adjuvant therapy and patient follow-up
In patients with stage II disease with risk factors or stage III disease postoperatively, oxaliplatin-based adjuvant chemotherapy was performed for 8–12 cycles. In patients who underwent chemotherapy, laboratory tests, including carcinoembryonic antigen (CEA) and abdominopelvic and chest CT, were performed at 3- to 4-cycle intervals during chemotherapy, and other examinations, such as sigmoidoscopy or total colonoscopy and positron emission tomography (PET)-CT, were added when necessary. After chemotherapy, follow-up examinations were carried out at 3-month intervals during the first 2 years postoperatively, at 6-month intervals until 5 years after surgery, and then annually if no evidence of recurrence was found.
Statistical analysis
Data for the early failure (EF) and the control groups were compared in terms of patient demographics, tumor characteristics, operative and postoperative outcomes, pathologic results, and oncologic outcomes. Descriptive results are presented as medians for continuous outcomes and as frequencies and percentages for categorical outcomes. For nonparametric statistics, Mann-Whitney U-test was used to compare continuous variables, and the chi-square test or Fisher exact test was applied for categorical variables. Five-year overall survival (OS) and recurrence-free survival (RFS) were analyzed using the Kaplan-Meier method, and comparison of survival between groups was performed by using the log-rank test. Statistical analyses were performed using IBM SPSS Statistics ver. 22.0 (IBM Co., Armonk, NY, USA). A P-value < 0.05 was considered statistically significant.
RESULTS
During the study period, 388 patients with rectal cancer received radiotherapy. Seven patients received radiotherapy for anal cancer or recurrent rectal cancer, 160 received preoperative short-course radiotherapy or postoperative radiotherapy, 72 were found to have an initial distant metastasis, 39 had no restaging work-up or follow-up, and 3 had preoperative exposure to other chemotherapy. Of these, 107 patients were enrolled in the study, and 7 developed early distant metastasis. In the patients with early distant metastasis, the lung was the most common site of distant metastasis (4 patients), followed by bone (3 patients), liver (1 patient), and intramural metastasis of the anal canal (1 patient) (Table 1). The median duration for metastasis was 78.1 days.
Patient demographics are detailed in Table 2. No significant differences were found between the EF and the control groups in terms of age, sex, body mass index, level of tumor location, initial clinical T and N stage, or tumor size. In addition, the duration and the doses of radiotherapy were similar. Preoperative CEA was significantly higher in the EF group than in the control group (5 mg/mL vs. 2 mg/mL, P = 0.010). Also, most patients in the control group received intravenous or oral 5-FU as a radiosensitizer, but patients who used capecitabine were relatively more frequent (EF: 28.6% vs. control: 3%, P = 0.002) in the EF group.
Only 4 of the 7 patients (57.1%) with early distant metastasis underwent a resection of the primary rectal lesion. Operative and postoperative outcomes in the patients who received a resection are compared in Table 3. The resection rate was significant lower in the EF group at 57.1% compared to the control group at 100% (P < 0.001). The median duration from CRT to surgery was significantly longer in the EF group (68.0 days vs. 54.7 days, P = 0.027). No significant differences were found between the groups in terms of type of approach, procedure, rate of sphincter-saving surgery, emergency surgery, operative time, or estimated blood loss. Postoperative morbidity and hospital stay were also similar between the 2 groups.
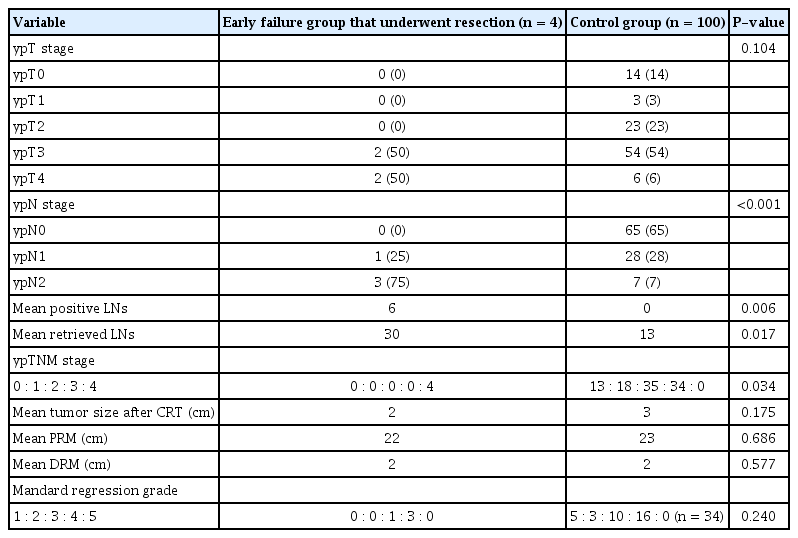
Among the postoperative pathologic results, the groups were not different in terms of ypT stage and tumor size (Table 4). However, both ypN stage and TNM stage were advanced in the EF group (P < 0.001 and P = 0.034, respectively), and the numbers of positive and retrieved LNs were significantly higher in the EF group (P = 0.006 and P = 0.017, respectively).
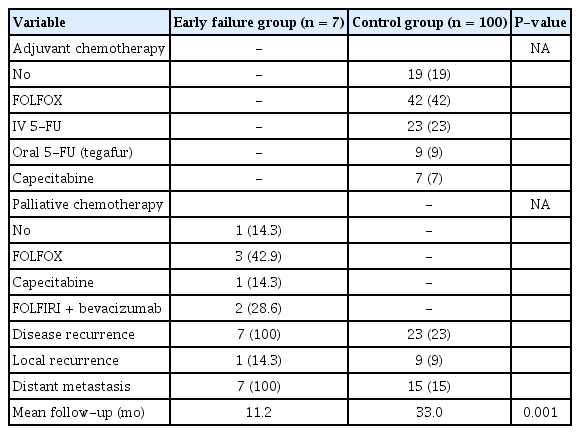
Adjuvant or palliative chemotherapy or the patients and their oncologic outcomes are described in Table 5. In the control group, local recurrence developed in 9 patients, distant metastasis in 15 patients, and disease recurrence in 23 patients. In the EF group, one patient experienced a local recurrence other than distant metastasis. The median follow-up duration was 11.2 months in the EF group and 33 months in the control group (P < 0.001). A significant difference between the 2 groups was found for RFS (P < 0.001), but not OS (P = 0.731).
DISCUSSION
In our study, early distant metastasis developed in 6.5% of the patients who had received preoperative CRT. This is a very rare report for early metastasis after preoperative CRT. Kang et al. [7] reported no patients who developed new metastatic lesions before or during surgery in a study comparing open and laparoscopic surgery for mid or low rectal cancer after neoadjuvant CRT. Bujko et al. [8] did not directly report the rate of early distant metastasis in their study comparing long-term oncologic outcomes of short-course RT and long-course CRT for patients with rectal cancer; instead, they reported that 140 of the 155 patients allocated to the short-course RT group and 143 of the 157 patients allocated to the CRT group had no distant metastasis at the time of surgery and were alive 30 days after surgery.
We identified the clinical characteristics of patients who developed early distant metastasis. Patients developing early metastasis had a significantly lower rate of primary tumor resection (57.1%) compared to the control group, possibly because patients with distant metastasis in the absence of symptoms frequently receive palliative chemotherapy without surgery. In addition, the time from CRT to surgery may have been longer in the EF group due to the design of new treatment plans for unexpected distant metastasis.
In this study, capecitabine as a radiosensitizer was frequently used in the EF group. The result could be misinterpreted to mean that capecitabine has weaker systemic effects than 5-FU, which differs from the results of previous studies. In general, capecitabine not only shows better oncologic outcomes than 5-FU at a systemically effective dose as an adjuvant treatment (1,250 mg/m2 twice daily), but its oncologic superiority continues at a lower dose (825 mg/m2 twice daily) as a radiosensitizer for CRT [9, 10]. Because the results in our study may have statistical bias due to the small number of patients in the EF group, further study including a larger number of patients is needed.
The lung was the most common site of distant metastasis in the EF group, which was unexpected given that, in general, the liver is the most common metastatic site of colorectal cancer. This difference might be attributed to the locations of the tumor levels in the lower rectum, which were a median of 4 cm above the anal verge. Generally, lymphatic drainage of the mid to lower rectum passes through the lateral pathway and drains directly to the lung [11, 12]. Bone metastasis was also common, second to lung metastasis, and both of these metastases are very aggressive, with short life expectancy [13, 14]. Thus, the metastatic lesions in the EF group indicated their potential aggressiveness, which was also represented by poor nodal response to preoperative CRT.
The possibility should be considered that early distant metastasis may arise from metastasis missed on initial imaging studies for work-up. Abdominopelvic and chest CT, as well as PET-CT, can be read as false negative for a distant metastasis with a small size [15, 16]. In particular, indeterminate nodules are common on chest CT, and pulmonary metastasis can be difficult to assess [6, 17]. Radiologic methods are limited in their ability to detect microscopic metastases. Accordingly, a relevant radiological diagnosis should be a prerequisite for formulating an appropriate treatment plan, as well as a follow-up imaging work-up.
The challenge remains to identify early which patients will experience a poor local response to CRT and develop a distant metastasis. We found no significant differences in any of the patients’ demographics or tumor characteristics between the EF and the control groups. If a meaningful intrinsic factor associated with the response to CRT, such as a gene expression profile, could be found, that factor could help in planning neoadjuvant therapy. However, studies on predictive factors are lacking. Some studies have suggested possible factors, but most of these remain at the experimental level [18, 19].
Accurate predictions of the responses to CRT or to craft tailored treatments are still not possible. The strategy of avoiding CRT due to concerns for early systemic failure may cause more injury than good for patients given that the rate of early systemic failure was very low in our study, at 6.5%. Therefore, a realistic alternative would be to conduct intensive follow-up for early detection of a distant metastasis during CRT, although debate is still ongoing regarding the utility of restaging images after neoadjuvant treatment for changing treatment strategy [20, 21]. Along with initial studies before CRT and preoperative restaging work-ups after CRT, additional abdominopelvic and chest CT could be added during the interval from 8 to 10 weeks after CRT.
This study had some limitations. First, it was a retrospective analysis with several biases and a small number of patients in the EF group. In particular, many patients received short-course radiotherapy at our institution and were excluded from this study. The results might have been different if they had also received CRT. Above all, evaluating the risk factors for early systemic failure was difficult because of the large difference in the numbers of patients between the EF and the control groups. Moreover, molecular biomarkers, such as epidermal growth factor receptor, K-ras/N-ras, microsatellite instabilities, and caudal type homeobox-2, were not analyzed due to insufficient data. Nevertheless, our study is very valuable because data on the clinical characteristics of early distant metastasis after CRT are rare.
In conclusion, unlike our concern, in this study, early distant metastasis rarely developed after CRT for the treatment of patients with rectal cancer. Patients who developed an early metastasis also had poor nodal responses to CRT, with low primary tumor resection rates and very poor oncologic results. For early detection and treatment, intensive follow-up imaging work-ups can be considered during the period between CRT and surgery. Furthermore, additional study is needed to identify intrinsic factors helpful for predicting the responses of patients to CRT and for tailoring treatment.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.