Preoperative Tattooing Using Indocyanine Green in Laparoscopic Colorectal Surgery
Article information
Abstract
Purpose
The aim of the present study was to evaluate the usefulness of indocyanine green (ICG) as a preoperative marking dye for laparoscopic colorectal surgery.
Methods
Between March 2013 and March 2015, 174 patients underwent preoperative colonoscopic tattooing using 1.0 to 1.5 mL of ICG and saline solution before laparoscopic colorectal surgery. Patients’ medical records and operation videos were retrospectively assessed to evaluate the visibility, duration, and adverse effects of tattooing.
Results
The mean age of the patients was 65 years (range, 34–82 years), and 63.2% of the patients were male. The median interval between tattooing and operation was 1.0 day (range, 0–14 days). Tattoos placed within 2 days of surgery were visualized intraoperatively more frequently than those placed at an earlier date (95% vs. 40%, respectively, P < 0.001). For tattoos placed within 2 days before surgery, the visualization rates by tattoo site were 98.6% (134 of 136) from the ascending colon to the sigmoid colon. The visualization rates at the rectosigmoid colon and rectum were 84% (21 of 25) and 81.3% (13 of 16), respectively (P < 0.001). No complications related to preoperative ICG tattooing occurred.
Conclusion
Endoscopic ICG tattooing is more useful for the preoperative localization of colonic lesions than it is for rectal lesions and should be performed within 2 days before laparoscopic surgery.
INTRODUCTION
Minimally invasive laparoscopic surgery for patients with colorectal cancer has become a popular alternative to conventional surgery due to its having comparable survival and recurrence rates [1, 2]. Intraoperative identification of lesions previously detected by endoscopy is often particularly difficult during laparoscopic surgery because of the inability of surgeons to palpate the colorectum intraoperatively. Therefore, accurate localization of colorectal tumors remains of critical importance during laparoscopic surgery. The lack of accurate lesion identification during laparoscopy may lead to resection of the wrong segment of the bowel [3-5]. For that reason, several methods, including barium enema, preoperative colonoscopy combined with fluoroscopy, placement of mucosal clips, and intraoperative colonoscopy, have been used to localize colorectal tumors during laparoscopic surgery. Each of these methods has drawbacks including, respectively, inaccurate localization, use of radiation, migration and poor cost performance of clips, and colonic insufflations [6-9].
Endoscopic tattooing of colonic lesions to assist in operative localization was first described in 1975 [10]. India ink tattoos properly placed in the colorectum are long-lasting and are reported to remain constant; therefore, they are being commonly used for colonic tattooing. However, India ink is currently not available in Korea because it has not yet been approved by the Ministry of Food and Drug Safety in Korea. Indocyanine green (ICG) has been described in some reports of animal models as an alternative candidate to India ink [11-13]. Although adverse reactions to ICG are known to occur [14], this dye has been used safely for more than 50 years in tests of cardiac and hepatic function, and more recently for sentinel node detection in cancer surgery [15, 16]. Furthermore, a saline test injection method involving injection of tattooing agents combined with prior and subsequent injections of saline into the submucosal layer has been found to result in better diagnostic accuracy and safety than are achieved with conventional methods [17]. For these reasons, we evaluated the usefulness of preoperative colonoscopic tattooing using the saline test injection method with ICG for colorectal lesion localization in laparoscopic surgery.
METHODS
Patients
This study was approved by the Ethics Committee of the National Cancer Center (approval number: NCC2015-0285). Informed consent was waived because of the retrospective nature of the study. The medical records and the operations videos of a total of 202 patients who had undergone preoperative colonoscopic tattooing using ICG (Diagnogreen Injection, Daiichi Sankyo Co., Ltd., Tokyo, Japan) before laparoscopic colorectal surgery at the National Cancer Center, Korea, between March 2013 and March 2015 were retrospectively reviewed. The visibility of the tattoos in 174 patients was reviewed by operation video or records, and a total 184 tattoo sites were identified and analyzed. Patient medical records and operation videos were retrospectively assessed to evaluate the visibility, duration, and adverse effects of the dye.
Methods
Visibility (tattoo success) was defined based on the identification of the serosal appearance of nonpalpable lesions marked with ICG during laparoscopic surgery; when tattoos were not visualized, we performed an intraoperative colonoscopy to identify the lesions. The duration was defined as the interval between endoscopic tattooing and the operation day. Probable adverse events associated with ICG marking, such as fever, chill, abdominal pain, and allergic reaction after tattooing and spillage of ICG, as well as focal peritonitis, abscess, or intraoperative adhesions visualized during the surgery, were observed and recorded.
All patients underwent mechanical bowel preparation before colonoscopic tattooing. On the day before the colonoscopy, no solid food was allowed after lunch. Four expert endoscopists, who had performed more than 3,000 colonoscopies, participated in this study. These endoscopists performed colonoscopic preoperative tattooing in close cooperation with the surgeons.
The saline test injection method was used to inject tattooing agents into the submucosal layer of the colon [17]. With a 23-gauge injection needle (Optimos Injector, Taewoong Medical Co., Ltd., Gimpo, Korea), 1 mL of saline was injected into the submucosal layer to form proper submucosal elevation. With a second syringe and needle, 1 to 1.5 mL of ICG was injected. The syringe was replaced by the first syringe containing saline, and 1 mL of saline was injected to flush out the ICG remaining in the needle device. The ICG was injected at 2 opposite sites (180 degrees apart) or 3 circumferential sites 120 degrees apart and distal to the neoplasm or previous endoscopic mucosal resection site. Information about the types of tumor sites, tattooing sites, and biopsies were obtained retrospectively from medical charts, endoscopy and pathology reports, and operation videos. Statistical analysis was performed using the chi-square test. A P-value less than 0.05 was considered statistically significant.
RESULTS
The 174 patients enrolled in this study included 110 men (63.2%) and 64 women (36.8%), with a median age of 65 years (range, 34–82 years). The mean body mass index was 23.9 kg/m2. Of 174 patients, 168 (96.6%) had colorectal adenocarcinomas, 4 (2.4%) had adenomas, and 2 (1.0%) had neuroendocrine tumors. Patients’ characteristics, pathologic TNM classification, and tattoo sites are shown in Table 1. No complications related to preoperative ICG tattooing were observed.
All patients had undergone laparoscopically assisted surgery. During the procedure, the aim was to identify the lesion through the laparoscope by looking for the serosal surface stained or inflamed by ICG injection. A total of 184 tattoo sites were identified, of which 172 (93.5%) were visualized intraoperatively. We were unable to locate the lesions of 12 tattoos laparoscopically, and in those cases, we performed an intraoperative colonoscopy in order to locate the lesions.
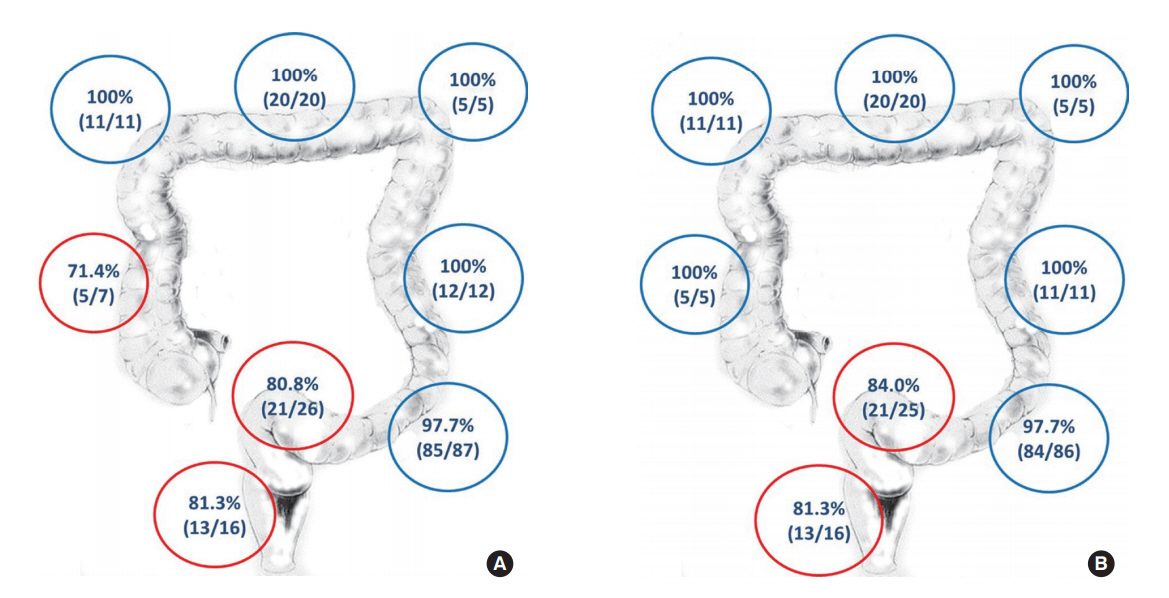
The median time between ICG tattooing and surgery was 1 day (range, 0–14 days), with 37 patients (21.3%) having been tattooed on the day of surgery. In this study, the visualization rates according to the intervals between tattooing and surgery were significantly different (P < 0.001). For tattoos placed within 2 days of surgery, the visualization rate was 95% (170 of 179). In contrast, if tattoos were placed more than 2 days before surgery, the visualization rate was only 40% (2 of 5) (Table 2, Fig. 1). Fig. 2A and B show the serosal appearance and invisibility of a nonvisible colonic lesion marked with ICG, as seen during laparoscopic surgery.

Visualization rate according to the intervals between preoperative indocyanine green tattooing and surgery.
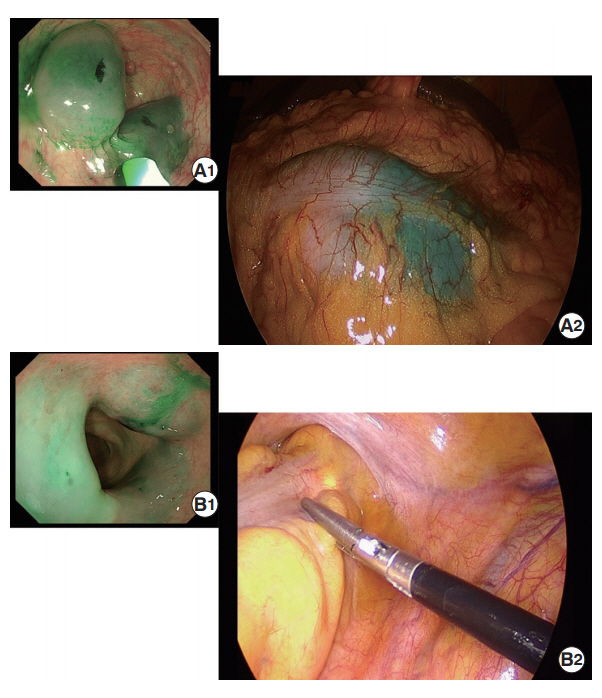
(A1-2) Identified tattoos and (B1-2) invisibility of a nonpalpable colonic lesion marked with ICG: (A1) endoscopic view after tattooing, (A2) serosal appearance during laparoscopic surgery (tattoo-operation interval: 1 day), (B1) endoscopic view after tattooing, and (B2) invisible lesion during laparoscopic surgery (tattoo-operation interval: 5 days).
ICG tattoos at the sigmoid colon were most common, and 97.7% (85 of 87) of them were visualized. Tattoos at the ascending colon were visualized in 5 of 7 cases (71.4%), and tattoos at the rectosigmoid colon and rectum were visualized in 21 of 26 (80.8%) and 13 of 16 cases (81.3%), respectively. Tattoos at the hepatic flexure, transverse colon, splenic flexure, and descending colon were all visualized intraoperatively. The dependence of the visualization rate on the tattoo site differed significantly between colonic sites (P = 0.002) (Table 2, Fig. 3A). The visualization rate by tattoo site for tattoos placed within 2 days before surgery was 98.6% (134 of 136) for the ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, and sigmoid colon taken together. However, at the rectosigmoid colon and rectum, the visualization rates within this time interval were 84.0% (21 of 25) and 81.3% (13 of 16), respectively (P < 0.001) (Fig. 3B).
DISCUSSION
The injection of a dye into the colonic wall is the most commonly used and effective method for marking colorectal lesions. Some tattooing agents, including India ink, ICG, methylene blue, indigocarmine, toluidine blue, and isosulfan blue, have been evaluated in animals, but only India ink and ICG were visible up to 48 hours after marking [11, 18, 19]. Several experimental studies comparing ICG and India ink for colonic tattooing in animals have reported that India ink is superior to ICG because of its higher visibility and longer duration [12, 13]. However, a longer duration is not always necessary for surgical resection. In terms of human cases, endoscopic tattooing with ICG in 12 patients was visible at 36 hours after injection and resulted in only minimal inflammation [18]. In another study of 39 patients, Miyoshi et al. [20] reported that the ICG marking was clearly observed in all 29 patients who underwent surgery within 8 days. However, at 9 days or later, the staining was clearly seen in only 2 of the 10 remaining patients (20%), and the markings in 3 patients were undetectable.
In this study, ICG tattooing was well observed intraoperatively in 95% of the patients within 2 days after endoscopic injection, but in only 40% of patients within 3 or more days after injection. For patients with tattoos placed within 2 days of surgery, the tattoo site also affected the visualization rate; namely, the visualization rates of the rectosigmoid colon and rectum were lower than those of other sites in the colon. On the whole, positive staining of water-soluble ICG tended to be weaker and fainter over time, finally dissipating without forming foreign material, as described previously in animal models [11-13]. These results support the proposition that ICG can be a safe option for endoscopic marking.
Clinically relevant complications of tattooing are considered to be rare [21]. Fu et al. [17] reported the rate of India ink leakage into the peritoneal cavity to be 1.8%, which is comparable with the rate reported in Miyoshi et al.’s ICG series (2.6%, 1 of 39) [20]. Askin et al. [22] reported that SPOT (GI Supply, Camp Hill, PA, USA), approved for human use by the U.S. Food and Drug Administration, is a safe and effective marker because no adverse effects were observed after 118 SPOT injections in 113 patients. Unfortunately, SPOT has not yet been approved by the Ministry of Food and Drug Safety in Korea.
ICG has been used for more than 50 years in a large number of patients and is considered to be extremely safe [14-16]. An absolute contraindication to ICG injection is an allergy to iodide such as an iodinated contrast material. Miyoshi et al. [20] reported that no surgical adverse effects of ICG injection were encountered in a sample of 39 patients. In our study, no complications due to ICG injection were observed. Therefore, ICG is a suitable alternative to India ink for colonoscopic tattooing, albeit with lower durability.
Some techniques of endoscopic tattooing may improve visualization of ICG compared with the conventional technique [17]. In our experience, using more than 1 mL enabled us to achieve 100% visualization, and we used amounts of 1 to 1.5 mL for injection after introduction of SPOT [4]. In this study, the saline test injection method was used, and 1 to 1.5 mL of ICG were injected into the submucosal layer of the colon. The visualization rate of this study was 93.5%, and no complications related to endoscopy or tattooing occurred.
ICG has been used less frequently and provides a less durable tattoo than India ink. Further studies are needed to improve the duration and the visualization rate of ICG tattooing. Recently, endoscopic marking using autologous blood was reported in several studies that used approximately 10 mL of patients’ coagulated blood immediately prior to colonoscopy, and the exact location of the lesion was visualized by the trail of blood during surgery in 92% (23 of 25) of the patients [23, 24]. The study described previously included a small number of cases; therefore, further research is needed to validate those results.
The utilization of fluorescence imaging with ICG may improve the detection rate of tumors compared with gross macroscopic color perception and may be a new method of colonic marking [25, 26]. Nagata et al. [25] reported that near-infrared-induced fluorescence with ICG showed tumor location clearly and accurately in all 24 patients while ten patients had negative staining of India ink. The authors suggested that fluorescence imaging offers high sensitivity in the determination of tumor location even though black or green staining cannot be seen by white light.
This study has several strengths and limitations worth noting. First, the present study included a large number of cases with no complications after tattooing. Second, we identified differences in the visibility of tattoos based on tattoo site and duration from injection to surgery. However, this study was limited by its retrospective and single-center design.
In conclusion, preoperative colonoscopic tattooing using ICG is a useful method for localization of colorectal lesions, and ICG marking may be suitable for surgical resections of the colon within 2 days after injection. Furthermore, this study highlights the need for endoscopists and surgeons to consider the effect of tattoo site on intraoperative visualization during laparoscopic surgery.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This work was supported by a National Cancer Center grant (NCC-1510150, NCC-1511670).