Gland Attenuation, a Novel Morphological Feature of Colorectal Cancer: Evidence for an Epithelial-Mesenchymal Transition
Article information
Abstract
Purpose
Along the invasive margin, colorectal cancer may show distinctive morphologic changes characterized by an asymmetrically attenuating tumor gland with loss of polarity. The author coined the term ‘gland attenuation (GA)’ for these peculiar changes. The aims of this study were to compare the immunoreactivity of the epithelial-mesenchymal transition (EMT) markers E-cadherin and β-catenin and thus determine whether EMTs occurs at tumor budding (TB) or GA sites and to assess the association of TB and/or GA levels with clinicopathological parameters and prognosis.
Methods
Expression patterns of E-cadherin and β-catenin in the tumor centers at GA and TB sites were examined in 101 patients with well or moderately differentiated CRCs, and the prognostic significance of TB and/or GA was statistically evaluated.
Results
GA foci, as well as TB foci, revealed loss of membranous and cytoplasmic E-cadherin expressions and aberrant β-catenin expression with reduced membranous expression and increased localization to the nucleus, suggesting that EMTs occur in GA as well as in TB. The high-TB and the TB-dominant groups were significantly correlated with advanced invasion depth, presence of lymph node metastasis, advanced pathologic staging and presence of lymphovascular invasion. The high-TB and the TB-dominant groups showed poor overall survival (OS) and recurrence-free survival (RFS), and high TB was an independent prognostic factor in the multivariate analyses for OS and RFS.
Conclusion
This study showed evidence that EMTs occurs at GA sites as well as TB foci. TB is a strong and independent prognostic factor, and TB-dominance may be an indicator of adverse clinical outcome.
INTRODUCTION
Colorectal cancer (CRC) is the third most common malignancy and is one of the leading causes of cancer deaths in the world [1]. In South Korea, CRC is the third most frequent cancer and the fourth leading cause of cancer deaths [2]. Metastasis is directly associated with poor outcomes [3] and about 90% of all cancer deaths are related to metastatic spread of the primary tumor [4]. Therefore, identifying histologic findings and molecular features of metastasis that can be used to predict the prognosis in patients with CRC is important. Metastasis is a multistep process and involves interactions of tumor cells with the host’s microenvironment [5]. Recent studies have reported that the epithelial-mesenchymal transition (EMT) is a key step toward cancer metastasis, and EMTs have been suggested to initiate the metastasis of cancer cells [6, 7].
The EMT is a key step during several phases of embryonic development and is involved in chronic fibrosis, tissue remodeling, wound repair and tumor invasion and metastasis [7-9]. Epithelial cells lose their characteristic cell-cell adhesion, and such an event may be crucial for cancer cells to acquire an invasive and motile phenotype [10-13]. Loss of E-cadherin expression and nuclear localization of β-catenin are emerging as two of the most common indicators of EMTs [14] and have been associated with more aggressive dissemination and poor prognosis in a variety of cancers [15-17]. Tumor budding (TB) is a characteristic histopathological feature identified by the presence of a detached isolated single or small clusters (less than 5) of tumor cells that have detached from the tumor’s epithelium and migrated a short distance into a stroma at the margin of the invasive tumor [18-20].
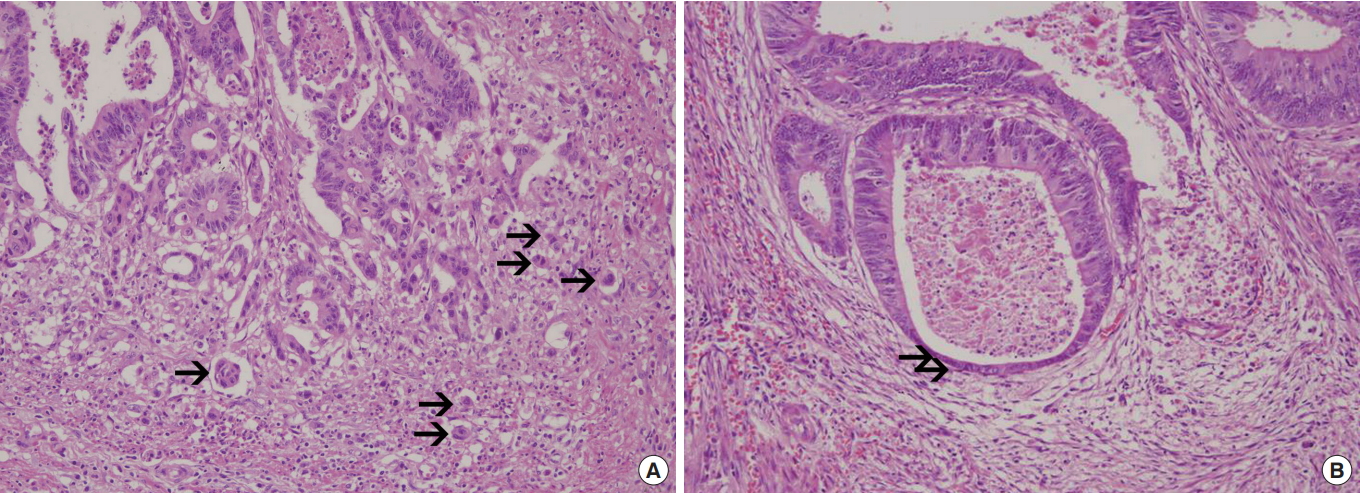
The adverse prognostic significance of TB in patients with colon cancer has consistently been reported [18, 21-26] and has been recognized by the American Joint Committee on Cancer/Union International Contre le Cancer (AJCC/UICC) as an additional prognostic factor to complement TNM staging [27]. In addition to TB, in our experience, another distinctive morphological change has been observed in the invasive front of CRC. Such a change is characterized by asymmetrically attenuating tumor glands distinctively in the region adjacent to the stroma at the invasive front that lose their characteristic polarity. The attenuated part of the gland is lined by a single layer of the partially low cuboidal, partially flattened epithelium, and the nuclei of the cells in that layer are arrayed haphazardly or parallel to the basement membrane. The opposite-side of the gland reveals a relatively well-preserved structure without distinct morphological change. This location-specific change, between the gland at the invasive front and TB, may suggest an interaction between the tumor and the host’s microenvironment. For this constellation of changes, the author coined the term ‘gland attenuation (GA).’
The author hypothesized that GA may involve EMTs and be considered as a transitional step between the conventional tumor gland at the invasive front and TB foci. The aims of the present study were to compare the immunoreactivities of the EMT-associated markers E-cadherin and β-catenin at TB foci, GA foci and the tumor’s center and thus to determine whether EMT occurs at TB or GA sites. Furthermore, the correlation of TB and/or GA levels with the clinicopathological characteristics and the prognostic impact of TB and/or GA on patients with CRC were assessed.
METHODS
Patients and specimens
From among the patients who had undergone curative surgery for a colorectal adenocarcinoma at Eulji University Hospital from January 2001 to December 2004, the present study comprised a nonconsecutive series of 130 cases. With the exception of 1 case (stage IV, pT4N1M1, R1 resection), an R0 resection was performed in all cases, including stage IV patients. Specimens obtained from patients who received preoperative chemoradiotherapy (CRT) were excluded from this study because CRT affects histopathologic findings. Postoperative CRT was recommended in patients with completely resected stage II or III rectal cancers. The patients who were diagnosed with a poorly differentiated adenocarcinoma (n = 29) were excluded from the study because they showed highly irregular glands or an absence of glandular differentiation, causing difficulty in measuring GA. Thus, 101 patients histologically confirmed to have had well or moderately differentiated colorectal adenocarcinomas were finally included in the study. We obtained various clinicopathological parameters for the patients, including age (less than 50/50 or more), sex (male/female), tumor size (less than 5 cm/5 cm or more), distant metastasis (absent/present), tumor invasion depth (pT1/pT2/pT3/pT4 or pT1, 2/pT3, 4), lymph node metastasis (pN0/pN1/pN2 or absent/present), pathological stage (I/II/III/IV or I, II/III, IV) and lymphovascular invasion (absent/present) from the patients’ medical records and pathology reports. The histological slides from the specimens were re-examined independently by one pathologist. Informed consent was obtained from each subject according to the institutional guidelines, and the Ethics Committee of the Eulji University Hospital (EMC 2018-08-010) approved the research protocols.
Evaluation and quantification of TB and GA
TB was evaluated using an Olympus BX41 microscope (Tokyo, Japan) with ×4, ×10, ×20, and ×40 objectives and ×10 ocular magnification. TB was defined as a single or a small cluster of <5 detached tumor cells at the invasive front (Fig. 1A). The presence of budding was determined according to the ‘rapid counting method’ proposed by Wang et al. [24]. In brief, each of 3 slides was examined in 5 different areas at ×200 magnification; then, the area was considered either positive (≥1 buds present) or negative (no buds present) for TB. The cases were categorized as high TB if at least 50% of the areas were positive for budding and as low TB if less than 50% of the areas were positive for budding. GA was defined as an asymmetrically attenuated gland at the invasive front with polarity loss, flattened epithelium and nuclei arrayed haphazardly or parallel to the basement membrane (Fig. 1B). The same scoring method was used to assess the degree of GA. For further evaluation of combined TB and GA sets, 2 categories of TB and 2 categories of GA were combined into a new variable with 3 categories including TB-dominant (high TB with low GA), balanced (high TB with high GA or low TB with low GA), and GA-dominant (low TB with high GA) cases.
Immunohistochemical staining and evaluation
Immunohistochemical stainings were performed on formalin-fixed, paraffin embedded blocks by using LSAB detection kits (DAKO, Carpentaria, CA, USA) according to the manufacturer’s instructions. The primary antibodies were anti-E-cadherin (1:500 dilution, DAKO) and β-catenin (1:100 dilution, DAKO). Tissue sections of 4 μm in thickness were cut and mounted on ProbeOn slides (Fisher Scientific, Pittsburgh, PA, USA). The sections were deparaffinized and rehydrated using a series of xylene and graded alcohols. After deparaffinization, slides were treated in 10 mM/L sodium citrate buffer (pH 7.0) for 20 minutes with the autoclave at 120°C for antigen retrieval. All sections for immunohistochemistry were incubated in 3% H2O2 for 10 minutes, to inactivate internal peroxidase, washed with 10-mM/L phosphate buffered saline buffer (pH 7.4), and incubated with normal bovine serum to reduce nonspecific antibody binding. The sections were then incubated with a primary antibody for 60 minutes, at room temperature. Detection of the immunohistochemical staining was performed with 3-amino-9-ethyl-carbazole (red color) or 3,3´-diaminobenzidine (brown color), as chromogen. The sections were counterstained for one minute with Mayer’s hematoxylin and then mounted.
Immunohistochemistry results were analyzed in a semiquantitative manner by 1 pathologist who was blinded to outcome. The staining intensities of membranous, cytoplasmic and nuclear β-catenin were graded as negative (0), weak (1), moderate (2), or strong (3). The staining intensities of membranous and cytoplasmic E-cadherin were also graded into 4 categories. The immunoreactivities of E-cadherin and β-catenin were separately analyzed for the tumor center, the preserved parts of the invasive margin glands, the attenuated parts of the invasive margin glands (GA sites), and the TB sites (Fig. 2).
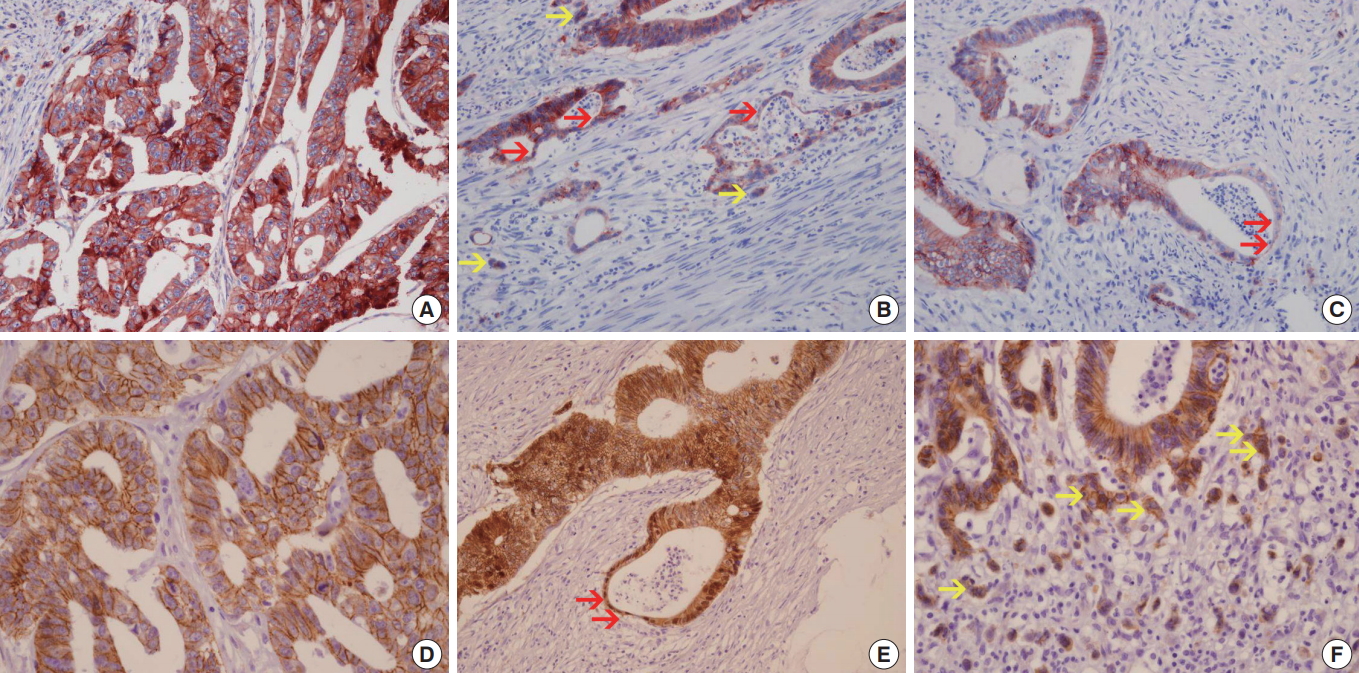
(A-C) Immunohistochemical staining of E-cadherin in a colorectal cancer specimen: (A) E-cadherin expression in the tumor’s center and (B, C) E-cadherin expressions in tumor budding (yellow arrows) and gland attenuation (red arrows). (D-F) Immunohistochemical staining of β-catenin in a colorectal cancer specimen: (D) β-catenin expression in the tumor’s center and (E, F) β-catenin expressions in tumor budding (yellow arrows) and gland attenuation (red arrows) (A-F, ×200).
Statistical analyses
Statistical analyses were performed using IBM SPSS ver. 18.0 (IBM Co., Armonk, NY, USA). Correlations between E-cadherin or β-catenin expression and the various clinicopathological parameters were analyzed by using Pearson chi-square test or Fisher exact test. Overall survival (OS) was defined as the period between treatment initiation and either the date of death or the last follow-up. Recurrence-free survival (RFS) was defined as the period from treatment initiation to first evidence of recurrence of a CRC or death from any cause. For the analyses of OS and RFS, Kaplan-Meier curves were plotted, and log rank tests or Breslow tests were used to calculate P-values. Multivariate analyses for OS and RFS were performed using the Cox proportional hazard model. In all statistical analyses, P-values < 0.05 were considered as statistically significant. Data are presented as the means ± standard deviations.
RESULTS
Baseline characteristics of the study group
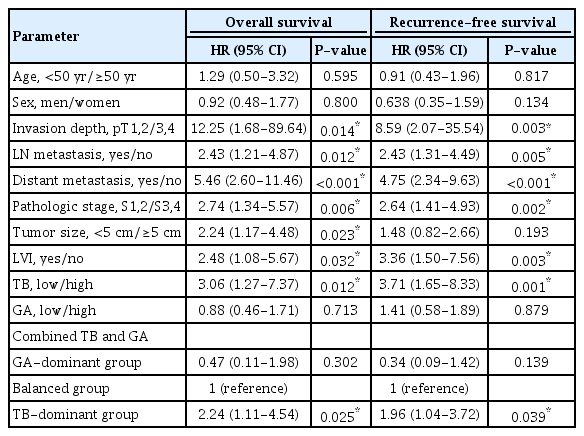
The study included 101 patients (51 men and 50 women), with a mean age of 62.7 years (range, 31–86 years). The mean tumor size was 4.8 cm (range, 0.8–9.0 cm) in maximum diameter. Sixty-seven patients (66.3%) had lymphovascular tumor invasion. TB was present in 97 patients (96.0%), low TB in 32 (31.7%), and high TB in 69 (68.3%). GAs were observed in 87 patients (86.1%). Low GA was found in 42 patients (41.6%) and high GA in 59 (58.4%). According to the 7th edition of the TNM classification of the AJCC/UICC [28], 5 patients (5.0%) were pT1, 17 (16.8%) pT2, 72 (71.3%) pT3, and 7 (6.9%) pT4. Fifty patients (49.5%) showed regional lymph node metastasis and 12 (11.9%) revealed distant metastasis. Eighteen patients (17.8%) were pathologic stage I, 32 (31.7%) stage II, 40 (396%) stage III, and 11 (10.9%) stage IV. Twenty-four patients (23.8 %) were treated with postoperative CRT, and the mean follow-up period was 57.4 months (range, 1–111 months). Table 1 summarizes the demographic and clinicopathological characteristics of the 101 patients with well or moderately differentiated CRC.
Immunoreactivity of E-cadherin and β-catenin
The immunoreactivities of E-cadherin and β-catenin were compared by tumor center, preserved part of the invasive front gland, attenuated part of the invasive front gland (GA), and TB sites (Fig. 2). The immunoreactivities of membranous and cytoplasmic E-cadherin decreased steadily from the tumor’s center (membranous: 2.33 ± 0.78; cytoplasmic: 2.27 ± 0.69) via the preserved parts of the invasive front glands (membranous: 1.89±0.80; cytoplasmic: 1.58 ± 0.78) to GA (membranous: 0.76 ± 0.49; cytoplasmic: 0.74 ± 0.56). However, in TB, the immunoreactivity of membranous E-cadherin was slightly higher (0.82 ± 0.78, P = 0.518) than in GA, and the cytoplasmic E-cadherin expression was significantly higher (1.15 ± 0.62, P < 0.001) than in GA. Membranous β-catenin expression was highest in the tumor’s center (2.48 ± 0.65) and then decreased steadily via the preserved parts of the invasive front glands (2.10 ± 0.77) and GA sites (0.91 ± 0.59) to TB sites (0.83 ± 0.72). Expressions of cytoplasmic and nuclear β-catenin showed steadily increasing trends from the tumor’s center (cytoplasmic: 0.56 ± 0.63; nuclear: 0.60 ± 0.24) via the preserved parts of the invasive front glands (cytoplasmic: 0.81 ± 0.64; nuclear: 1.57 ± 0.89) and GA sites (cytoplasmic: 0.94 ± 0.71; nuclear: 1.45 ± 0.93) to TB sites (cytoplasmic: 1.26 ± 0.72; nuclear: 1.78 ± 0.89).
Correlation of TB and/or GA with clinico-pathological parameters
The relationships between the TB level and the clinicopathological characteristics were analyzed, and the results are summarized in Table 2. In brief, high TB was significantly correlated with advanced invasion depth (P = 0.022), presence of lymph node metastasis (P = 0.001), advanced pathologic staging (P < 0.001) and presence of lymphovascular invasion (P = 0.005). Other variables, such as age, sex, tumor size, tumor site, tumor grade, distant metastasis, and tumor border, did not show any significant correlation with high TB. No statistically significant relationship was found between GA level and any clinicopathological parameter except for tumor site. High GA was more common in patients with rectosigmoid colon than in patients with right/transverse colon, and this finding was statistically significant regardless of pathologic stage (P = 0.031) (Table 2). High TB was significantly associated with high GA (P = 0.001). Of the 11 patients with stage IV cancer, 5 were TB-dominant, and 6 were balanced (5 patients with high TB and high GA, 1 with low TB and low GA). The relationships between the combined TB and GA variable and the clinicopathological characteristics were also analyzed. The positive rates of lymph node metastasis, pathologic stage and lymphovascular invasion increased linearly from GA-dominance to TB dominance, and the linear trends were statistically significant (Table 3).
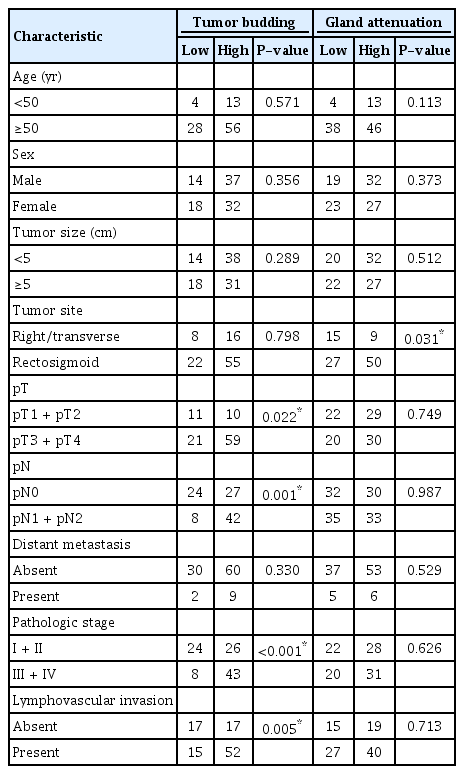
Correlations between tumor budding and gland attenuation levels and the clinicopathological parameters
Survival analyses for TB and/or GA
After an average follow-up of 57.4 months (range, 1–111 months), we found that 33 patients (32.7%) had died of CRC with or without metastasis, 4 (4.0%) had died of unrelated causes, 13 (12.9%) were still alive with local recurrence and/or distant metastasis, and 51 (50.5%) remained alive and were recurrence-free. The 5-year OSs were 79.6% for patients in the low-TB group and 57.6% for those in the high-TB group, and the 5-year RFSs were 77.3% for patients in the low-TB group and 43.9% for those in the high-TB group. Analyses of the Kaplan-Meier survival curves showed significant differences in the adverse OS rates (log rank test, P = 0.008) and the RFS rates (log rank test, P = 0.001) between patents with high and low TB (Fig. 3). The 5-year OSs were 62.5% for patients in the low-GA group and 65.7% for those in the high-GA group, and the 5-year RFSs were 56.5% for patients in the low-GA group and 53.5% for those in the high-GA group. Kaplan Meier survival curves were not statistically different between patients with low GA and those with high GA (Fig. 3). With regard to the combined TB and GA variable, the 5-year OSs were 81.8% for patients with GA-dominance, 66.9% for those with balanced TB and GA, and 47.6% for those with TB-dominance. Likewise, the 5-year RFSs were 81.8% for patients with GA-dominance, 55.5% for those with balanced TB and GA, and 38.1% for patients with TB-dominance. Analyses of the Kaplan-Meier survival curves also showed significant differences in adverse OS rates (Breslow test, P = 0.020) and RFS rates (log rank test, P = 0.015) among the 3 types of patients (Fig. 3). Other conventional risk factors, including tumor size, grade, lymphovascular tumor invasion, lymph node metastasis, invasion depth and TNM stage, were also significantly correlated with survival rates (data not shown). Univariate analyses were performed for the OS and the RFS survival by using the Cox proportional hazard model.
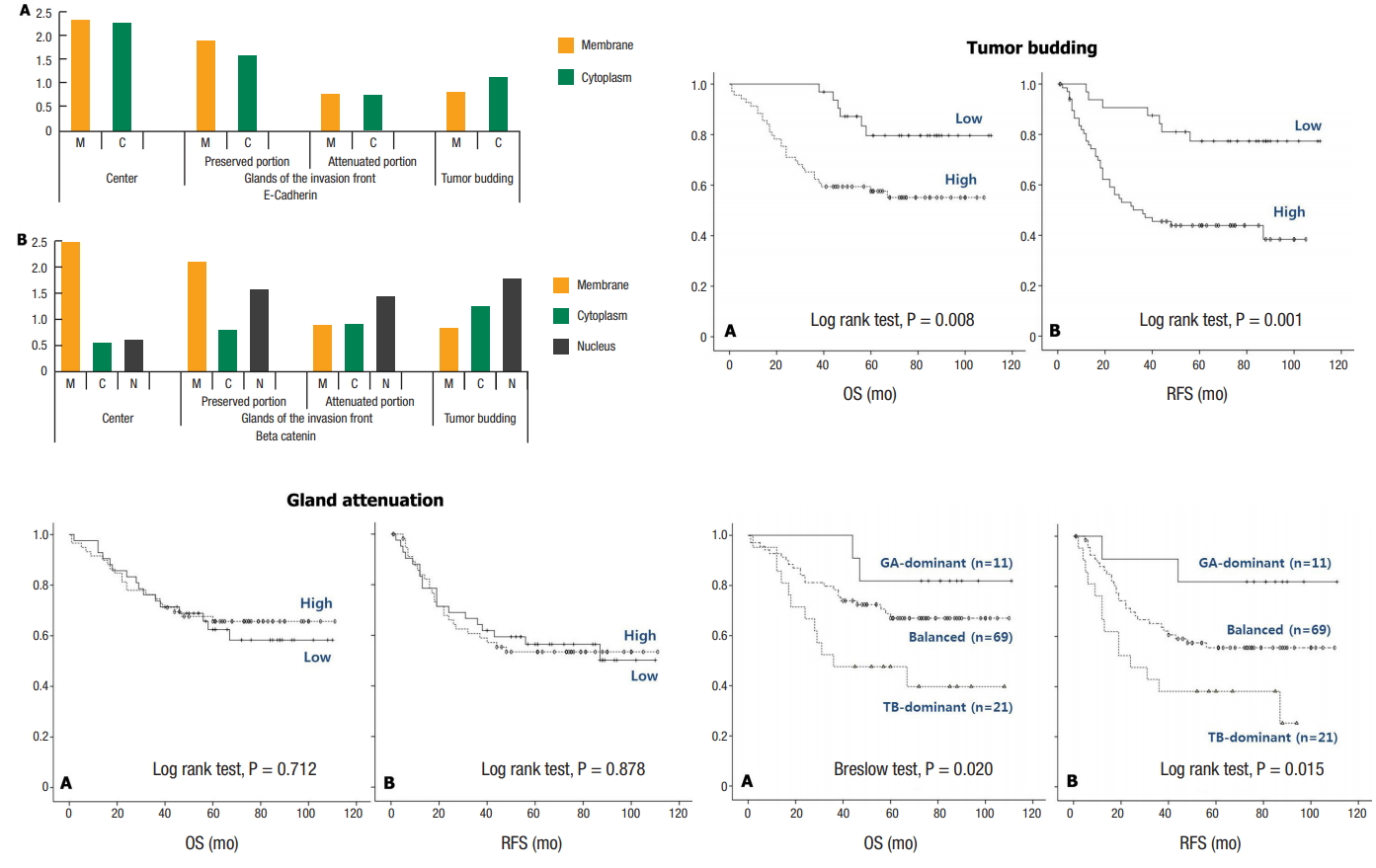
(Upper left) Immunohistochemical scores of b-catenin (A) and E-cadherin (B) in patients with colorectal cancer. (Upper right) Comparison of Kaplan-Meier survival rates between patients with colorectal cancer with high and low levels of TB: overall survival (A) and recurrence-free survival rates (B). (Lower left) Comparison of Kaplan-Meier survival rates between patients with colorectal cancer with high and low levels of GA: overall survival (A) and recurrence-free survival rates (B). (Lower right) Comparison of Kaplan-Meier survival rates between patients with colorectal cancer with three degrees of combined TB and GA: overall survival (A) and recurrence-free survival rates (B). TB, tumor budding; GA, gland attenuation.
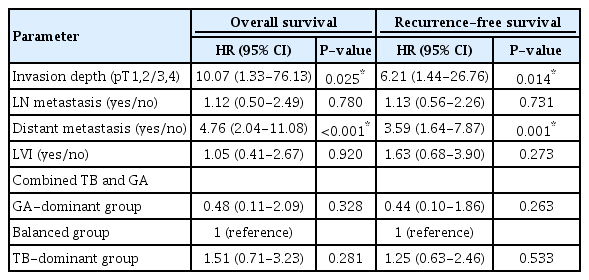
The univariate analyses showed that lymph node metastasis, distant metastasis, pathologic stage, lymphovascular invasion, TB and combined TB and GA had significant associations with both overall and RFSs and that tumor size had a significant association with OS. The results of the univariate analyses of the clinicopathological parameters are listed in Table 4. After initial screening by using univariate analyses, TB and conventional pathological variables, including invasion depth, lymph node metastasis, distant metastasis and lymphovascular invasion, were used in the multivariate analyses for OS and RFS by using the Cox proportional hazard model. High TB was independently associated with shorter OS (hazard ratio [HR], 2.82; 95% confidence interval [CI], 1.14–6.99; P = 0.025) and RFS (HR, 2.71; 95% CI, 1.15–6.39; P = 0.023). Invasion depth (HR, 11.28; 95% CI, 1.51–84.45; P = 0.018 for OS; HR, 7.03; 95% CI, 1.64–30.09; P = 0.009 for RFS) and distant metastasis (HR, 5.91; 95% CI, 2.54–13.72; P < 0.001 for OS; HR, 3.90; 95% CI, 1.81–8.38; P < 0.001 for RFS) were also independently correlated with both OS and RFS (P < 0.05). In addition, another multivariate model was used, and the combined TB and GA variable, instead of TB, was included in the model. In that model, invasion depth (HR, 10.07; 95% CI, 1.33–76.13; P = 0.025 for OS; HR, 6.21; 95% CI, 1.44–26.76; P = 0.014 for RFS) and distant metastasis (HR, 4.76; 95% CI, 2.04–11.08; P < 0.001 for OS; HR, 3.59; 95% CI, 1.64–7.87; P = 0.001 for RFS) were significantly correlated with both OS and RFS (P < 0.05). The combined TB and GA was an independent prognostic factor in the univariate model, but the association was no longer significant in the multivariate model (Table 5).
DISCUSSION
Malignant tumors are well known to be able to invade adjacent tissues through a variety of mechanisms including cohesive invasion, amoeboid invasion and EMT-mediated invasion. EMT is now considered an important step in tumor invasion and metastasis in many cancers [29, 30]. In CRC, EMT can be identified histologically by the presence of TB which occurs predominantly at the invasive front [31]. One of the major pathways of carcinogenesis of CRC involves loss of function mutations of the APC gene, which activates the WNT/wingless signaling pathway. In addition, its main downstream effectors, E-cadherin and β-catenin, are considered integral components and the most common indicators of EMT [32].
An important finding of this study is the histomorphological changes within the invasive front glands seen in many patients with CRC are well or moderately differentiated colorectal carcinomas, and to the best of our knowledge, this characteristic feature has never been mentioned in any previous study involving such patients. Therefore, I used the term ‘gland attenuation’ to describe this change. GA in the invasive front glands of CRC is characterized by partial loss of conventional glandular structure, asymmetric attenuation of the tumor’s epithelium, loss of polarity, and nuclei arrayed haphazardly or parallel to the basement membrane. Additionally, all of the above features are most frequently seen along the invasive front of the tumor and are typically observed in the part of tumor glands that are against the stroma of the invasive margin. In contrast, the opposite side of the same gland tends to show relatively well-preserved gland architecture with adherent polarized tumor cells. The tumor-host interaction at the invasive front of CRC represents a dynamic interface between pro- and anti-tumor factors [31]. On one hand, the colonic tumor invades the adjacent tissue by TB, which reflects tumor progression. On the other hand, the host attempts to defend against this situation by mounting an ‘anti-tumor’ inflammatory response [31, 33]. In this context, the above-mentioned, location-specific change along the invasive margin, which the author calls GA in this paper, may suggest the presence of an interaction between the tumor and the host’s microenvironment.
Evidence of EMT markers at the invasive edge of the tumor has been provided by several investigators. Jang [25] reported that expressions of cytoplasmic and nuclear β-catenin were significantly higher at TB sites than in the tumor’s center and that expressions of membranous and cytoplasmic E-cadherin were significantly lower at TB sites. Gao et al. [34] reported that the nuclear β-catenin expression level was significantly lower in the tumor’s center than at the tumor’s invasive front, and Lee et al. [35] also reported that losses of E-cadherin expression and nuclear β-catenin expression were higher in the invasive margin than in the tumor’s center. According to the results of the present study, both of the different morphological patterns at the invasive margin, TB and GA, are accompanied by a loss of membranous E-cadherin expression and a nuclear accumulation of b-catenin in the cells within the attenuated tumor’s epithelium. The above results strongly suggest that EMT might occur at GA sites as well as TB sites. What is interesting about the immunohistochemical results is that membranous and cytoplasmic E-cadherin expressions were their lowest in the GA area (membranous, 0.76 ± 0.49; cytoplasmic, 0.74 ± 0.56) and that the expression was slightly higher in the TB foci (membranous, 0.82 ± 0.78; cytoplasmic, 1.15 ± 0.62). Cytoplasmic and nuclear expressions of β-catenin were also increased for TB (cytoplasmic, 1.26 ± 0.72; nuclear, 1.78 ± 0.89) compared to GA (cytoplasmic, 0.94 ± 0.71; nuclear, 1.45 ± 0.93). These results are consistent with the results of an earlier study which reported that metastatic tumor cells derived from tumors originally expressing nuclear β-catenin were found to re-express E-cadherin and that their β-catenin became cytoplasmic by a phenotypic transition process, such as modulating the intracellular β-catenin distribution in tumor cells [36]. With few exceptions, GA is observed between a conventional tumor gland and TB along the invasive margin. Taken together, these results suggest that, during a complex stepwise EMT process, GA may represent an intermediate stage between an invasive front gland and TB.
Additionally, this study reconfirms TB as a strong adverse prognostic marker in patients with well or moderately differentiated CRCs. The adverse prognostic impact of TB on patients with CRC has been repeatedly reported [21, 22, 24, 37]. Furthermore, TB is frequently linked to lymph node involvement, lymphovascular invasion, high-grade tumors, and local tumor recurrence [21, 26, 38]. In this study, although GA was not statistically related to the clinicopathological parameters or survivals, the combined TB and GA variable was associated with several adverse prognostic parameters, including invasion depth, lymph node metastasis, pathologic staging and lymphovascular invasion. In the survival analysis, patients in the high-TB and the TB-dominant groups showed poor OS and RFS, and high TB was an independent prognostic factor in the multivariate analyses for OS and RFS.
In conclusion, the present study proposes the novel concept of a histomorphological change in the invasive front of CRC, called GA, and shows evidence that EMTs occur at GA sites as well as TB foci. In addition, the author suggests the possibility that GA may be an intermediate stage in the EMT process between the invasive front gland and TB. High TB is confirmed to be an important prognostic factor for predicting a poor prognosis in patients with CRC. Moreover, combined TB and GA findings may be good markers for predicting a prognosis for such patients.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.