Initial local excision for clinical T1 rectal cancer showed comparable overall survival despite high local recurrence rate: a propensity-matched analysis
Article information
Abstract
Purpose
Local excision (LE) is an alternative initial treatment for clinical T1 rectal cancer, and has avoided potential morbidity. This study aimed to evaluate the clinical outcomes of LE compared with total mesorectal excision (TME) for clinical T1 rectal cancer.
Methods
Between January 2000 and December 2011, we retrospectively reviewed from multicenter data in patients with clinically suspected T1 rectal cancer treated with either LE or TME. Of 1,071 patients, 106 were treated with LE and 965 were treated with TME. The data were analyzed using propensity score matching, with each group comprising 91 patients.
Results
After propensity score matching, the median follow-up time was 60.8 months (range, 0.6–150.6 months). After adjustment for the necessary variables, patients who underwent LE showed a significantly higher local recurrence rate than did those who underwent TME; however, there were no differences in disease-free survival and overall survival. In the multivariate analysis, age (hazard ratio [HR], 9.620; 95% confidence interval [CI], 3.415–27.098; P<0.001) and angiolymphatic invasion (HR, 3.63; 95% confidence interval, 1.33–9.89; P=0.012) were independently associated with overall survival. However, LE was neither associated with overall survival nor disease-free survival.
Conclusion
LE for clinical T1 rectal cancer yielded a higher local recurrence rate than did TME. Nevertheless, LE provided comparable overall survival rate and can be proposed as an optional treatment in terms of organ-preserving strategies.
INTRODUCTION
Since the adoption of the concept of total mesorectal excision (TME), it has become the standard treatment for rectal cancers. TME involves the specification of precise definitions of distal margin, circumferential margin, and minimum number of excised lymph nodes, which lead to better oncological outcomes; however, many authors reported significant morbidity rates in the form of anastomotic leakage, genitourinary complications, and the need for temporary or permanent stoma [1-3]. Recent advances in screening colonoscopy have made it feasible to achieve early detection of rectal cancers and influenced to a large extent the shift in treatment plans seeking to minimize the morbidity rate associated with radical surgery. Local excision (LE) of clinical T1 (cT1) rectal cancer is an attractive option due to its potential advantages, such as better anorectal function, lower morbidity, and mortality, compared with TME. Additionally, LE avoids unnecessary wide rectal resection and decrease harmful complications, especially, in elderly or comorbid populations. However, on the other hand, there is a substantial risk of local recurrence or systemic metastasis in patients without additional treatment after LE and increased morbidity associated with surgical complications can be warned in the LE group when they received a radical surgery for local recurrence [4].
Although many authors have reported that the risk of lymph node metastasis is about 20% in stages T1 and even T2 rectal cancer, nearly 80% of these patients underwent radical surgery for no good reason. Many guidelines recommended LE for stage T1 rectal cancer without lymph node or distant organ metastasis; however, there is no consensus regarding the appropriate treatment for stage T2 rectal cancer [5, 6]. You et al. [7] reported that among patients with stage T1 tumors, there was a higher local recurrence rate after LE than after TME; however, the 5-year overall survival rates were the same. Regarding stage T2 rectal cancers, patients who received LE showed worse oncologic outcomes than those who received TME [7-9].
In spite of the recent technologies that are utilized in preoperative staging modalities such as endoscopic imaging, chest and abdominopelvic computed tomography (CT), pelvic magnetic resonance imaging, and rectal ultrasonography, it seems quite difficult to distinguish between stages T1 and T2 rectal cancers preoperatively. The risk of lymph node metastasis in stage T1 rectal cancer is 12.7% to 14.5% and that of stage T2 rectal cancer is 18.4% to 23.9%. This is correlated with the depth of tumor invasion, poor differentiation, angiolymphatic invasion, venous invasion, and perineural invasion, and also has a major influence on the disease-free and overall survival rates [10-12]. Some authors reported that LE and TME for early rectal cancers have comparable benefits, but with a lower morbidity rate after LE; they also reported that LE should be followed by a strict surveillance protocol [7]. We designed this study to evaluate the oncologic outcomes following both LE and TME for cT1 rectal cancers with great interest in determining the risk factors associated with recurrence.
METHODS
Patients and data collection
We retrospectively reviewed the medical records of patients who underwent LE or TME for cT1 rectal cancer with pathologically confirmed stage T1 or T2 rectal adenocarcinoma from January 2000 to December 2011. We enrolled patients who underwent LE at the National Cancer Center, Seoul National University Hospital, and Seoul National University Bundang Hospital in Korea. Patients who underwent TME at the National Cancer Center were used as the control group. We excluded patients with synchronous distant metastasis, those who were treated with neoadjuvant chemoradiotherapy and emergency operation, and radical treatment was recommended for patients who had adverse pathological features after LE, according to the guidelines. Salvage operation was defined as reoperation due to local or distant recurrence. A positive margin was defined as one with evidence of tumor within < 1 mm of the margin of the resected tumor. The study protocol was approved by the Institutional Review Board of National Cancer Center, Republic of Korea (NCC2015-0103). Written informed consent was waived due to its retrospective nature.
Preoperative evaluation
Pretreatment evaluation included physical examination, colonoscopy, chest and abdominopelvic CT scan, endorectal ultrasonography, and assessment of carcinoembryonic antigen level. Tumor staging and distance from the anal verge were established by considering the combined results of all modalities used. Radical resection was strongly recommended for patients who were diagnosed with stage ≥ T2 tumors or who had lymph node metastasis on preoperative evaluation. LE was recommended for patients who were diagnosed with stage ≤ T1 rectal cancer and who had no lymph node metastasis on preoperative evaluation. However, LE was indicated in patients who refused radical resection (TME or Miles operation) and those who were unfit for major surgery.
Treatment and follow-up
LE was performed via transanal excision or transanal endoscopic microsurgery. Almost all patients received treatment under general anesthesia except some patients with spinal anesthesia. These patients had high risk factors for general anesthesia or had their preference. Patients who underwent LE were positioned such that the rectal tumor was located posteriorly from the surgeon’s perspective. In the LE group, full-thickness resection of the rectal tumor was performed with a 1-cm gross surrounding margin of normal rectal wall. The specimens were fixed on a board, marked for orientation, and sent to the pathologist. The defect in the rectal wall was closed using absorbable sutures. Angiolymphatic invasion was defined as the presence of tumor cells within lymphatic or blood vessels and perineural invasion was defined as spread of tumor cells in, around, and through nerves [12].
Both groups underwent standardized follow-up. Patients were followed regularly every 3 months for the first 2 years and every 6 months thereafter. Moreover, we performed digital rectal examination, complete blood counts, biochemical profiles, and serum carcinoembryonic antigen level assays at every visit. Follow-up imaging studies, such as abdominopelvic CT and colonoscopy were performed as clinically indicated or at the physician’s discretion. Local recurrence was defined as any tumor recurrence within the true pelvis, including tumor recurrence within the obturator lymph nodes. Distant recurrence was defined as any tumor recurrence outside the true pelvis. The survival time was calculated as the interval from the date of operation to the date of last visit or confirmed death, whichever occurred first. Relapse-free survival was measured from the surgery to recurrence or death from any cause and recurrence-free survival included any recurrence from rectal cancer.
Statistical analysis
Differences in baseline characteristics between the 2 groups (LE vs. TME) were assessed using the chi-square test, Fisher exact test, or Wilcoxon rank-sum test, as appropriate. To adjust for the differences in baseline characteristics between the 2 groups, propensity score matching was applied using the logistic regression model. Variables used in the propensity model were sex, age, tumor location, tumor size, and T classification. Subsequently, a 1:1 propensity match between 2 groups was obtained using the nearest neighbor matching within a set caliper width. Survival analysis was performed using the Kaplan-Meier method with the log-rank test. Risk factors were evaluated using the Cox proportional hazards model. Univariate analysis was performed at first; only variables with P-values of ≤ 0.1 were included in the multivariate analysis. All statistical analyses were performed using SPSS version 14.0 (SPSS Inc., Chicago, IL, USA). Analysis items with a P-values of < 0.05 were considered statistically significant.
RESULTS
Clinicopathologic characteristics of the study population
From January 2001 to December 2011, 1,061 patients were enrolled in this study. A total of 106 patients underwent LE and 956 patients underwent TME. The baseline characteristics of both groups are presented in Table 1. Both groups had similar distributions of sex, age, and body mass index. Compared with patients who underwent TME, those who underwent LE had a lower tumor location (4.3 cm vs. 8.0 cm, P<0.001), smaller tumor size (2.0 cm vs. 2.8 cm), and a higher rate of stage T1 cancers (79.4% vs. 29.2%, P<0.001). The rate of angiolymphatic invasion was higher in the TME group (15.0% vs. 29.2%, P=0.003), while the patients who underwent LE had a higher proportion of threatened margin (28.9% vs. 2.2%, P<0.001). The period of postoperative hospital stay was significantly longer in the TME group (5.82 vs. 10.27 days, P<0.001). The rate of salvage operation after local recurrence was significantly higher in the LE group than in the TME group (16.0% vs. 2.1%, P<0.001).
After propensity score matching, 91 matched pairs of patients were selected. Table 2 shows the clinicopathologic characteristics of the matched patients. The variables used for propensity score matching were sex, age, tumor location, tumor size, and T stage classification. Among the matched patients, LE more likely led to a threatened margin (29.7% vs. 2.2%, P<0.001), shorter in-hospital stays (5.8 days vs. 11 days, P<0.001), and a higher rate of salvage operation (17.6% vs. 2.2%, P=0.001).
Treatment-specific recurrence and survival outcomes
At a median follow-up period of 60.8 months (range, 0.6–150.6 months), 17 patients who underwent LE experienced disease recurrence while 7 patients treated with TME did. Of 17 patients treated with LE, 11 developed local recurrence only, 4 developed distant recurrences only, and 2 developed synchronous local and distant recurrence. The details of patients who experienced tumor recurrence after LE are summarized in Table 3. Of 7 patients treated with TME, 6 developed distant recurrence only, and only 1 patient developed local recurrence. The 5-year overall survival rates were 96.0% in the LE group and 91.1% in the TME group (P=0.425) (Fig. 1A). The 5-year disease-free survival rate was 86.5% in the TME group and 79.9% in the LE group (P=0.359) (Fig. 1B).
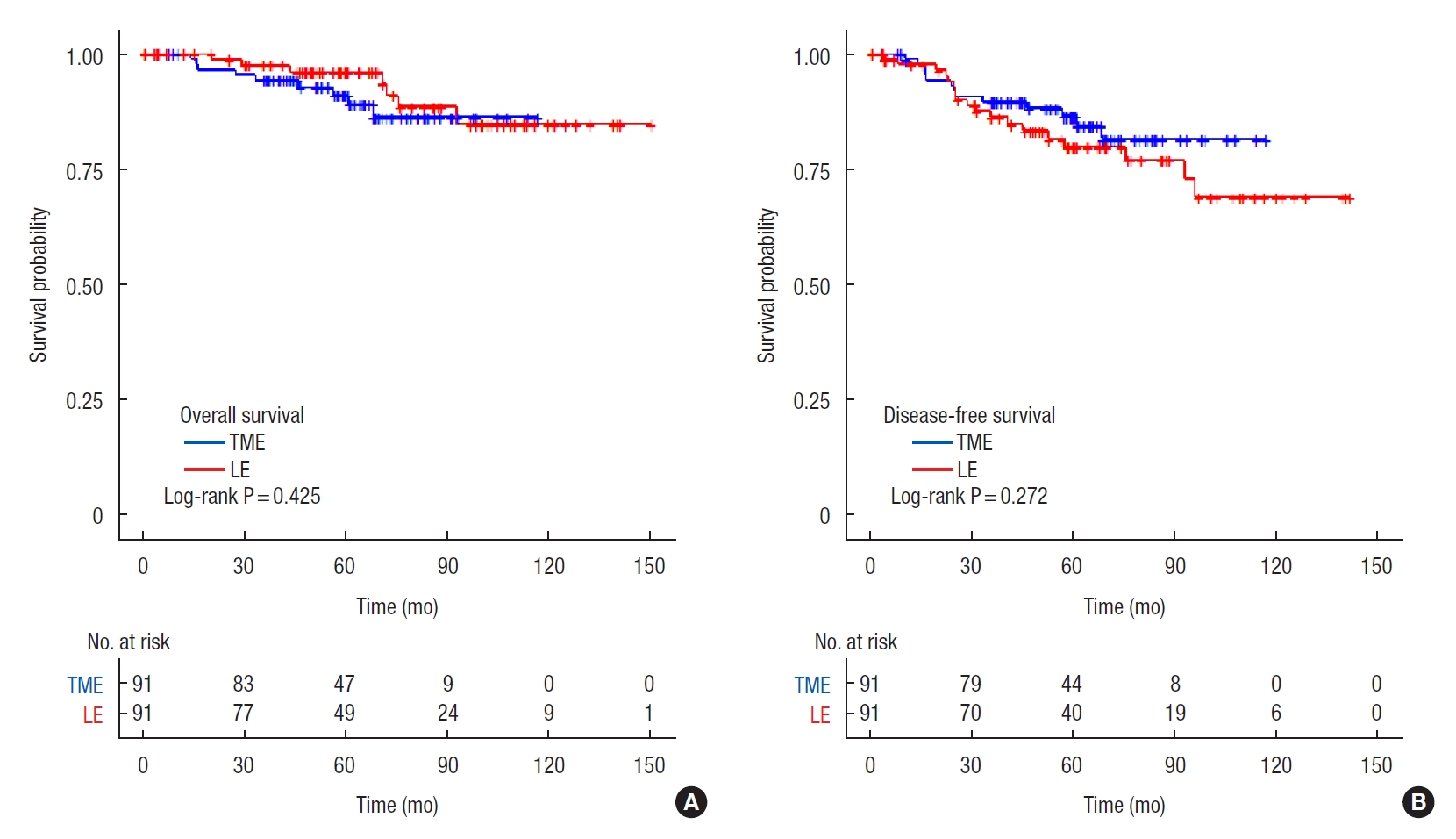
Comparison of overall survival (A) and disease-free survival (B). TME, total mesorectal excision; LE, local excision.
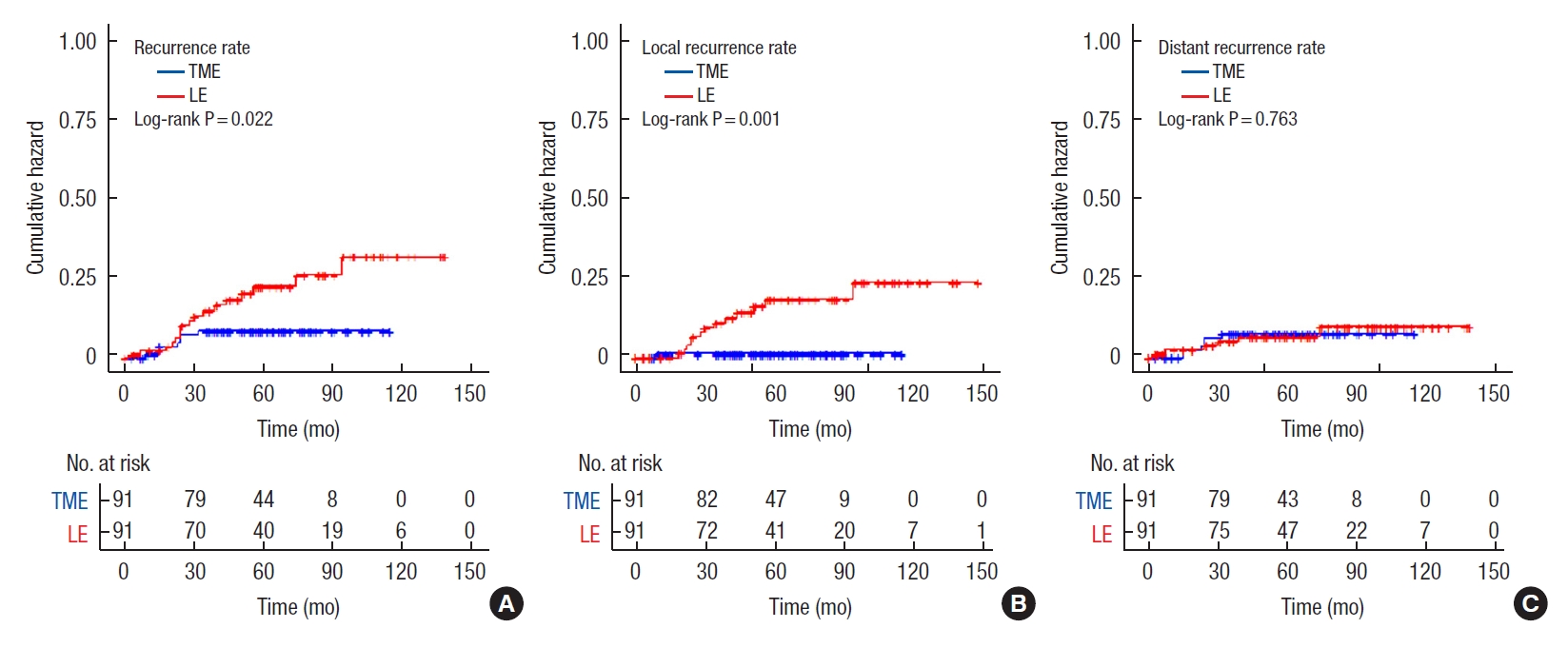
The median time to local recurrence was 30 months (range, 18–94 months) in the LE group compared with 9 months in the TME group, while the median time to distant recurrence was 27 months (range, 3–74 months) in the LE group compared with 71 months (range, 3–115 months) in the TME group (Fig. 2). The 5-year local recurrence-free survival rate was 83.3% in the LE group compared with 98.9% in the TME group (P=0.001). The 5-year distant recurrence-free survival rate was 93.7% in the LE group compared with 92.9% in the TME group (P=0.994).
Variables affecting patient survival
Logistic regression analysis was performed to identify the risk factors affecting the prognoses of patients. The independent risk factors for overall survival that yielded P ≤ 0.1 in the univariable analysis were sex, age, and angiolymphatic invasion, while those of disease-free survival were age, T classification, and angiolymphatic invasion (Table 4). In the multivariable analysis, age of ≥ 70 years (hazard ratio [HR], 9.620; 95% confidence interval [CI], 3.415–27.098; P<0.001) and angiolymphatic invasion (HR, 3.63; 95% CI, 1.33–9.89; P=0.012) were independent risk factors for overall survival. Also, age (HR, 2.676; 95% CI, 1.276–5.611; P=0.009) and angiolymphatic invasion (HR, 2.197; 95% CI, 1.052–4.591; P=0.036) were independent factors for disease-free survival. In addition, LE (HR, 13.752; 95% CI, 1.794–105.426; P=0.012) was the only independent risk factor for local recurrence (Table 5).
DISCUSSION
This study analyzed the oncologic data regarding LE among patients with early rectal cancer. The results showed a significantly higher local recurrence rate in the LE group; however, there was no statistically significant difference in overall survival between the 2 treatment modalities. The study findings could be used to support both patients and surgeons in decision-making.
The main goals of performing LE in rectal cancer are proper oncologic outcomes with minimum morbidity and mortality while avoiding a permanent colostomy and poor functional outcomes associated with TME. Although TME is considered the optimal treatment for rectal cancer, treatment of early rectal cancer requires a balance of the immediate complications, functional disadvantages including genitourinary dysfunction and permanent stoma with the oncological outcomes. LE is selected for patients who are unfit to undergo major surgery or patients with early rectal cancer as it offers rapid recovery, lower morbidity, and very rarely results in mortality. However, previous reports showed a higher local recurrence rate after LE compared with TME [7, 13, 14]. Table 6 summarizes the differences in local recurrence and disease-free survival between LE and radical surgery among patients with stage T1–2 tumors [7, 8, 13, 15-17]. Nash et al. [17] showed that the local recurrence rate is significantly higher after LE (13.2%) compared with TME (2.7%) in T1 rectal cancer. They also showed that LE was inferior to TME in terms of disease-free survival (87% vs. 96%, respectively). However, You et al. [7], Bentrem et al. [8], and Mellgren et al. [13] reported no significant difference in overall survival rate between the LE and TME groups of patients with stage T1cancer; our results corroborate those of these previous studies. An analysis of the Surveillance, Epidemiology and End Results database in the United States revealed that LE of T1 rectal cancer did not affect the cancer-specific survival (HR, 1.6; P=0.236). However, patients with T2 rectal cancer treated with LE showed an increased hazard of cancer-specific survival (HR, 1.71, P=0.001) [18]. Based on this finding, many guidelines recommended that T1 cancer with favorable characteristics is the only indication of LE [5, 6].
Our study revealed that the overall survival rate among early rectal cancer patients treated with LE was comparable to that among those treated with TME, but had higher rates of local recurrence and an inferior disease-free survival rate. It should be considered that in addition to the small sample size of the LE group, the patients were not randomly selected for either approach; the selection depended mainly on the surgeons’ and patients’ preferences. Among patients with stage T1 rectal cancer treated with LE, 10 (11.0%) experienced tumor recurrence (8, local recurrence; 2, distant metastasis). All local recurrences were treated with salvage surgery (6, radical resection; 2, re-LE). Only 1 patient with local recurrence treated by Miles operation experienced a second recurrence in the lung and died from cancer recurrence (Table 3). Among patients with stage T2 rectal cancer treated with LE, 7 (31.8%) experienced tumor recurrence, including 3 with local recurrence, 2 with distant recurrence, and 2 with local recurrence with synchronous distant metastasis. A majority of patients with local recurrence treated with salvage surgery showed cancer-free survival. All cases of local recurrence of T2 rectal cancer were treated with radical resection and one of them died due to cancer (Table 3). Compared to our study, You et al. [7] and Mellgren et al. [13] reported that patients with stage T2 rectal cancer showed a higher local recurrence rate and inferior overall survival after LE compared with TME. Our study enrolled only 22 patients (24.2%) with stage T2 rectal cancer and we suppose that the small sample size and the differences in tumor characteristics may have affected our results.
The higher local recurrence rates reported by many groups after LE may be related to unresected mesorectal lymph nodes, threatened resection margin, and potential tumor seeding during manipulation of the tumor. Preoperative imaging studies such as endorectal ultrasonography, CT scan, and magnetic resonance imaging do not always precisely reveal the depth of tumor and lymph node involvement. Also, LE does not offer information about the lymph node status which determines the need for postoperative adjuvant therapy. Lymph node metastasis occurred in 6% to 11% and 17% to 43% of patients with stages pT1 and pT2 rectal cancer, respectively [11, 19, 20]. In our study, 31.8% of patients with stage T2 rectal cancers experienced tumor recurrence, compared with 15.9% of patients with stage T1 rectal cancers. The high recurrence rate observed among patients with stage T2 rectal cancers treated by LE is likely to be related to the higher rate of lymph node metastasis compared with that among patients with stage T1 rectal cancers.
The pathological parameters that predict poor prognosis such as depth of invasion, angiolymphatic invasion [21], tumor budding [22], and threatened resection margin [23] are critical when selecting patients. Thus, we recommended TME for patients with high-risk tumor characteristics after LE. Pathological risk factors may not be evaluated using preoperative biopsy specimens; thus, detailed examination of the LE specimen is important to decide if further treatment is required. In this study, age and angiolymphatic invasion were associated with overall survival. Another study also showed similar results regarding the association between angiolymphatic invasion and distant metastasis of rectal cancer [21]. Rectal cancer with angiolymphatic invasion has more aggressive oncological features and can spread to the systemic circulation. Thus, patients with angiolymphatic invasion identified on pathological examination should be referred for more aggressive and radical therapy.
One of the most important factors that are associated with local recurrence is the status of the surgical margin [23]. However, in our study, 29.7% of patients in the LE group had threatened resection margin, this was not associated with local recurrence in the multivariable analysis. This may be due to shrinkage of the resected specimens before pathologic examination, in addition to the fact that fixation of the specimen on a cardboard and cauterization of the margin during LE makes it difficult to evaluate the margin status.
This study has several limitations, such as the inherent bias due to the retrospective nature of the study. To reduce bias, data were obtained from multiple centers and surgeons; we also applied the propensity score matching. After matching, the patients’ characteristics became homogeneously distributed. The small sample size is also a limitation of this study.
In conclusion, patients with cT1 rectal cancer treated with LE had a higher local recurrence rate compared with those treated with TME; however, the overall survival rates were similar. LE can be an optional initial treatment modality for early rectal adenocarcinoma, particularly low-risk T1 cancer. Salvage surgery after local recurrence might improve the chance of cancer cure. However, further large-scale randomized control studies are required to clarify the exact role of LE in the treatment of early rectal cancer.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
This research was supported by The Korean Society of Coloproctology Research Grant (KSCP-2020M003) and the National Cancer Center Grant (1810192-1).
Acknowledgements
The authors thank Kyoung Min Kang and Dong Eun Lee for their help in the statistical analysis.