Perianal Actinomycosis: A Surgeon’s Perspective and Review of Literature
Article information
Abstract
Actinomycosis is a serious suppurative, bacterial infection caused by the gram-positive anaerobic Actinomyces species. Primary perianal actinomycosis is rare and challenging for the colorectal surgeon. We aimed to present our experience and compare this with available literature. All patients with isolated Actinomyces on microbiology reports, between January 2013 and February 2021, were identified and reviewed. Data collection was retrospective based on electronic patient records. The site of infection and treatment strategy were examined. Perianal cases were evaluated in depth. All publications available in the literature were interrogated. Fifty-nine cases of positive actinomycosis cultures were reviewed. Six cases of colonization were excluded. Actinomyces turicensis was the most common organism isolated. Five cases of perianal actinomycosis were identified requiring prolonged antibiotic and surgical therapy. Twenty-one studies, most case reports, published since 1951 were also reviewed. Diagnosis of perianal actinomycosis may be challenging but should be suspected particularly in recurrent cases. Prolonged bacterial cultures in anaerobic conditions are necessary to identify the bacterium. An extended course of antibiotic therapy (months) is required for eradication in certain cases.
INTRODUCTION
Perianal sepsis is a common problem for the general surgeon, presenting either as an acute abscess or anal fistula. In the United Kingdom, the annual incidence is estimated between 21.5 and 30.8 per 100,000 population [1]. Etiology, however, has not been fully proven. Chiari was the first to describe in 1878 the anal glands that discharge at the mucocutaneous junction of the anal canal into the anal crypts [2]. The pathogenesis of perianal suppuration is currently attributed to those anal crypts getting blocked by debris or stool, which subsequently results in cryptitis and formation of abscess [3]. The majority of cases can be usually dealt with without complications, yet a small number can pose a significant challenge for both patient and surgeon. These cases are often associated with a variety of conditions such as Crohn’s disease, lymphogranuloma venereum, tuberculosis, or actinomycosis.
Actinomycosis involving the perianal region can be quite complex with recurrent abscesses, multiple draining sinuses, and fistulas in ano. This form of the disease is a rare entity and not generally appreciated. Proper treatment is often delayed for months or even years as early detection requires a high index of suspicion [4]. The aim of this study was to review and present our experience in a district general hospital and raise awareness of this rare form of the disease.
METHODS
Our institution consists of 2 major hospitals that serve an area of more than 500,000 residents. All bacteriology reports related to Actinomyces species, between January 2013 and February 2021, were identified from a prospectively maintained electronic database. Only positive cultures for Actinomyces species were included in the study. Histology reports were also examined to confirm diagnosis wherever possible. Retrospective clinical data were additionally obtained from institutional electronic patient records by 3 separate clinicians. Site of infection, treatment strategy, and baseline characteristics of patients were collected. All cases of perianal disease were identified and a more detailed review of these was undertaken. No ethical approval was required for this study as it was performed as part of a local audit process. In order to publish relevant imaging, written informed consent was subsequently obtained from 2 patients.
A comprehensive literature search was also performed parallel to the study to identify publications relevant to perianal actinomycosis. The following keywords and Medical Subject Heading (MeSH) terms were used: “actino*,” “anorectal,” “anorectum,” and “perianal.” The online bibliographic databases of PubMed, Medline, and Embase were searched for all citations published in English between May 1950 and April 2021. Selection was limited to human studies only. Initially, titles and abstracts were screened to identify papers reporting actinomycosis of the perianal region. Subsequently, a full-text review of the remaining studies was performed.
RESULTS
One hundred and eighty bacteriology reports of Actinomyces species were scrutinized and 59 cases were identified with positive Actinomyces culture. Six of these cases represented colonization and were therefore excluded from the analysis. The median age of included patients was 33 years and male to female ratio was 1:3. Mixed growth of Actinomyces species was observed in 39.6% of the cases (21 of 53), but the most commonly isolated subtype was Actinomyces turicensis in 34.0% (18 cases). Other less common subtypes included Actinomyces neuii, Actinomyces meyeri, and Actinomyces israeli with 5, 3, and 2 cases, respectively.
Positive cultures for Actinomyces were identified predominantly in patients with soft tissue infections (breast, back and groin abscesses, or dehisced infected wounds). Natal cleft comprised the second commonest site, whereas only 5 cases involving the anorectum were detected (Table 1). Other areas where Actinomyces were isolated included cervicofacial and abdominal regions. There were 2 cases of systemic actinomycosis isolated in blood cultures. One death was recorded in the series and this was related to systemic actinomycosis with rapid hematogenous spread and formation of multiple brain abscesses.
In the perianal cohort (Table 2), 3 of the 5 patients with positive cultures suffered recurrent episodes of infection resulting in complex disease with chronic and multiple anal fistulas and sinus tracts requiring further surgery and prolonged treatment with antibiotics. Histological evidence of sulfur granules was detected in only 1 patient that validated the diagnosis.
The operation usually involved drainage of sepsis as a first step. None of the patients had macroscopic evidence of sulfur granules on the initial procedure. For those with a recurrence, a number of return journeys to theatre were required (range, 3–6). This usually comprised examination under anesthesia of the anorectum with either drainage of sepsis, seton insertions, and/or fistulectomy depending on the occasion and surgeon’s decision. Magnetic resonance imaging (MRI) with contrast of the pelvis was used in all 3 patients with the recurrence to guide surgical management. According to Parks classification of fistulae [2], patient 2 had multiple transsphincteric fistulae and 1 supralevator with 2 external openings, patient 3 had 4 transsphincteric and 1 intersphincteric fistula, and patient 5 had multiple sinus tracts extending to the perineum but no formal fistula was identified on MRI scan or surgery. Fig. 1 shows images of the 65-year-old male with deep intersphincteric and extrasphincteric fluid collections.

Magnetic resonance imaging of pelvis STIR sequence with contrast of the 65-year-old male with perianal actinomycosis. At 12:00, there is an intersphincteric and extrasphincteric 8 mm fluid collection which continues with a small tract to the right side of the buttock up to the subcutaneous surface around 8 o’clock.
Antibiotic choice varied between cases but usually a combination of either amoxicillin or cephalosporin and metronidazole was necessary. As no clear guidelines were available, antibiotic choice was planned in conjunction with the microbiologist. Initial length of treatment was decided for 6 weeks depending on response. However, the 26-year-old female patient suffered a recurrence while on antibiotics and therefore a course of 30 weeks in total was essential. In 1 of the male patients, the 49-year-old, the disease spread superiorly into the perineum and scrotal involvement was also observed (Fig. 2). All patients with recurrence were followed up in an outpatient setting to confirm resolution of the disease.
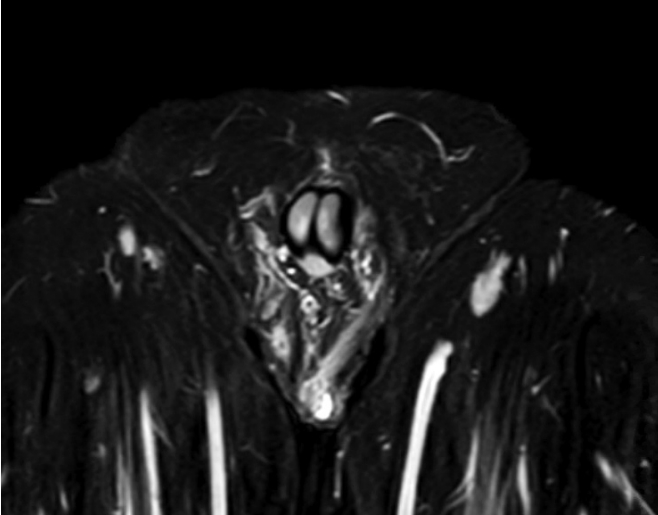
Magnetic resonance imaging of pelvis STIR sequence with contrast of the 49-year-old male. There is a small collection with a thick enhancing wall at the base of the left scrotum extending posteriorly to the left perineum. The collection measures 2.0 × 0.7 × 2.1 cm. No fistula or communication with the anus.
As shown in Table 1, prolonged antibiotic therapy combined with further surgery was also observed in patients with soft tissue (23.8%) and pilonidal infection (20.0%) for recurrence.
All publications related to perianal actinomycosis since 1951 are listed in Table 3 [4-24]. As anorectal involvement is a rare manifestation of the disease most of the studies included here are actually case reports or case series. No randomized trials or prospective studies were identified. Mean duration of antibiotic therapy was prolonged and varied from weeks to even years [16, 23]. The weighted average was 8.3 weeks across studies with available date. The largest cohort comes from Brewer et al. [4] with 9 patients over a 22-year period.
DISCUSSION
Actinomycosis is a rare but potentially serious bacterial infection caused by the gram-positive anaerobic filamentous bacilli of the Actinomyces species. The bacteria belong to the genus of the Actinobacteria group and they are predominantly anaerobic commensals of the oropharynx, urogenital, and gastrointestinal tract [25]. The exact mechanism that leads to overt disease is still unknown, as actinomycosis occurs in both immune-competent and immune-compromised patients [5]. Sanford and Voelker published the first large series of the disease in 1925, reporting 670 cases of actinomycosis of various anatomical sites. Interestingly they described only 5 cases related to the perianal region [21]. The infection commonly occurs in the cervicofacial, thoracic, and abdominopelvic regions [25]. Anorectal involvement may result from a secondary extension of an abdominal infection or may arise primarily in the anorectal region [4]. Primary perianal actinomycosis is generally quite rare with only a few reports published in the literature. Egal et al. [5] found the condition to represent less than 0.2% of all fistulae in ano in their series.
The anorectal form usually begins in an anal crypt where it evolves as a chronic bacterial infection that is characterized by slowly progressing suppurative fibrosing inflammation, development of draining sinus tracts that may discharge characteristic “sulfur granules” and direct dissemination via adjacent tissues [21, 26]. The acute phase is rarely recognized due to the insidious onset, and the diagnosis is nearly always late [21]. It can often be misdiagnosed because it mimics other more common conditions such as malignancy and tuberculosis [27].
Diagnosing actinomycosis is challenging. Patients may give a protracted history of operations involving repeated attempts for drainage of perianal abscess, or fistulotomies [4]. There is a wide range of differentials such as inflammatory bowel disease, tuberculosis, malignancy, hidradenitis suppurativa, lymphogranuloma venereum, and syphilis [12]. Patients usually undergo a variety of tests including endoscopic procedures with biopsies, computed tomography, and virology tests.
Isolation and identification of the causative bacteria occur in only a minority of cases. Demonstration of Gram-positive filamentous organisms and sulfur granules on histological examination is strongly supportive of the diagnosis of actinomycosis. Direct isolation of the organism from a clinical specimen or from sulfur granules is usually required for a definitive diagnosis. However, the failure rate of isolation is particularly high (>50%) for various reasons. These include previous antibiotic treatment, overgrowth of concomitant organisms, or occasionally inadequate methodology [27]. Gram staining of pus and pathology of infected tissue is of great value, as it is often more sensitive compared to plain culture which remains sterile in more than 50% of cases [25]. Common practice is to send only bacterial swabs from the perianal abscess, but rarely the tissue. Several series such as Egal et al. [5] have been published with good evidence that sending both specimens to the laboratory at every surgery is important.
Histological analysis of all excised tissue from fistulas and abscesses should be carried out routinely. Clinicians should alert the laboratory to expect the specimen and specifically request for actinomycosis to ensure that prolonged culture on appropriate media is performed; Actinomyces are slow-growing organisms that need to be cultured anaerobically for up to 3 weeks [27]. Molecular methods, such as polymerase chain reaction and 16s ribosomal RNA sequencing have been proven to be powerful tests in confirming and/or identifying Actinomyces species [28].
In our series, contradicting standard practice, all cases were identified from positive cultures, and in only 1 occasion diagnosis was confirmed histologically with positive gram stain. It is likely that some cases especially soft tissue infections, may not represent a typical actinomycetic lesion [28] but simple infections involving Actinomyces species. For the perianal group that we particularly focused on, we believe that only 3 out of 5 cases represent true actinomycosis and those are the patients with recurrent episodes requiring prolonged antibiotic treatment. Interestingly macroscopic evidence of sulfur granules was not identified in any of them. From our experience, culture of the affected tissue is of great significance, particularly in cases of diagnostic dilemma.
Currently, there are no standardized guidelines on antibiotic therapy. Our choice was usually amoxicillin with a combination of anaerobic cover (metronidazole or clavulanic acid). In vitro studies have reported that Actinomyces are susceptible to a wide range of antimicrobial agents. A UK study of 87 clinical isolates of Actinomyces showed that most were susceptible to β lactams (including benzylpenicillin, amoxicillin, ceftriaxone, meropenem, and piperacillin-tazobactam), doxycycline, clindamycin, erythromycin, and clarithromycin. Species identification was found to be crucial because of resistance to some antibiotics [29]. Although several antibiotic regimens may be used, the most preferable first choice remains intravenous penicillin, specifically 10–20 million units per day of penicillin G over 2 to 6 weeks, divided 4 times a day, followed by therapy with penicillin V or amoxicillin for 6 to 12 months [11]. Certain antibiotics are, nonetheless, considered to be ineffective, including quinolones, aminoglycosides, and first-generation cephalosporins [30]. Appropriate duration is a matter of debate although in most series patients received antibiotics for a minimum of 6 months. Regular long-term follow-up of the patient is paramount during extended antibiotic treatment to ascertain adherence and to assess for clearance of the infection. The mean duration of follow-up in the colorectal clinic in our series was 18 months.
CONCLUSION
Perianal actinomycosis is a rare form of the disease and has only been described in isolated case reports and small series. Surgeons should be aware of the possibility of actinomycosis as the cause of indolent perianal abscesses. Diagnosis is challenging requiring a high level of clinical suspicion. Prolonged bacterial cultures are necessary to identify the bacterium. Adequate surgical drainage and exploration of sinus tracts appear beneficial but not always adequate. Treatment strategy and decisions should be on a case-by-case basis as an extended course of antibiotic therapy may be required for eradication. Surgical drainage and exploration of sinus tracts appear beneficial but not always sufficient.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
Acknowledgements
The author would like to thank the Drs. Atia Khan and Henrietta Leslie for their valuable contribution in data collection as well as Dr. Sahar Musaad for her support and advice.



