Right-sided acute diverticulitis in the West: experience at a university hospital in Argentina
Article information
Abstract
Purpose
In the West, diverticular disease is located mainly in the left colon. However, it can also present in the right colon, with an incidence of 1% to 2% in Caucasians. The purpose of this study was to describe our experience in right-sided acute diverticulitis (RD).
Methods
In this retrospective study, 410 patients with acute diverticulitis treated from 2013 to 2020 were included in a university hospital in Córdoba, Argentina. Colonic diverticulitis was stratified into 2 groups; RD and left-sided acute diverticulitis. Demographic and clinical variables, laboratory and imaging findings, type of treatment, follow-up, and recurrence were analyzed.
Results
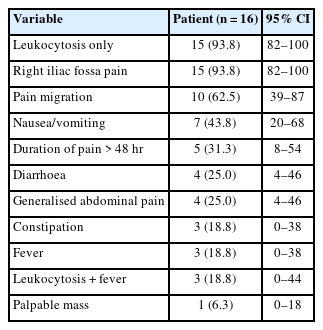
Sixteen patients (3.9%) with RD were identified; 62.5% were male and the mean age was 40.7±11.7 years. A total of 81.3% were Caucasian and 18.7% Native American. Significant differences were found between both groups of diverticulitis; patients with RD were younger (P=0.001), with lower BMI (P=0.01), comorbidity rate (P=0.01), Charlson comorbidity index (P=0.02), hospital stay (P=0.01), severity according to the Hinchey classification (P=0.001) and had a lower recurrence rate (P=0.001). There were no significant differences in sex (P=0.95), duration of pain until admission (P=0.05), laboratory findings (P=0.23) and treatment (P=0.34).
Conclusion
Conservative treatment predominated in RD, with a lower rate of complications and recurrences, providing data that support conservative therapy as initial treatment in RD in our environment.
INTRODUCTION
Diverticular disease (DD) has long been considered a common disease in Western countries. Worldwide, the major incidence is found in the United States, Europe, and Australia, affecting 50% of the population over the age of 60 years, with a prevalence of 15% to 35% [1]. The lifetime risk of acute diverticulitis in someone with diverticulosis is 10% to 25% [2].
Diverticulitis is a heterogeneous disease in both presentation and prognosis. Uncomplicated diverticulitis is the most common presentation and is characterized by acute onset of diverticular inflammation without abscess or perforation [3]. Among gastrointestinal diseases, is a leading cause of ambulatory visits, hospital admissions, and colon surgery, accounting for substantial healthcare expenditures [4].
Besides the geographical variability in the prevalence of DD, there is significant variability in the location of colonic diverticula in different regions of the world [5]. According to reports in the West, it affects primarily the left side (90%) predominating in the sigmoid colon, whereas in Asia it mostly affects the right-sided colon (80%) with a prevalence of 8% to 25% [6, 7]. However, with increasing migration, it has recently been detected a major frequency of right-sided DD in the West [8]. These differences suggest that the pathophysiology of DD may differ between populations.
In the past decade, our understanding of diverticulitis has expanded substantially with the publication of multiple observational studies and clinical trials. Historically, diverticulitis was thought to be an aggressive disease, and the threshold for surgical intervention was low. With a better understanding of prognosis, a shift toward nonoperative approaches has occurred [3].
The purpose of this study was to describe our experience in right-sided acute diverticulitis, carried out in a Western university hospital.
METHODS
Ethics statements
This study was conducted in accordance with the Declaration of Helsinki of 1964 (revised in 2013) and was approved by the Institutional Ethics Committee of the Clinica Universitaria Reina Fabiola (No. 2021-017).
Population and study design
A retrospective cohort study was performed, which included all patients aged ≥ 18 years with a diagnosis of acute diverticulitis treated from January 2013 to January 2020 at a University Hospital in Córdoba, Argentina. The information of the patients was collected through the review of medical records and an electronic database of the General Surgery Service. The colonic diverticulitis was stratified into 2 groups, according to the location of the diverticula: right-sided diverticulitis (RD) and left-sided diverticulitis (LD). The patients with DD associated with neoplasia and those with incomplete information in the electronic medical records were excluded.
Study of variables
The aspects analyzed were demographic variables (sex, age, and ethnic origin), body mass index (BMI), chronic comorbidities, Charlson comorbidity index (CCI), time of evolution of abdominal pain until admission to the emergency room, presence of peritoneal signs in the physical examination (abdominal palpation), body temperature, modified Hinchey classification, treatment (conservative or surgery), length of hospital stay, and symptom recurrence after discharge. Blood tests recorded the white cell count (WCC), the neutrophil-to-lymphocyte ratio (NLR), and the levels of C-reactive protein (CRP). The original diagnosis of the report made by the on-call radiologist was recorded, based on the initial abdominal ultrasound and computed tomography (CT).
Definition of variables
Modified Hinchey classification: The uncomplicated diverticulitis was defined as stage 0–Ia and complicated diverticulitis was defined as stage Ib–IV or associated with fistulas, stenosis, or colonic obstruction. RD is the colonic diverticulitis that spreads from the cecum to the transversal colon. LD is the colonic diverticulitis that spreads from the splenic flexure to the sigmoid colon. A body temperature of ≥38 °C was defined as fever, and a WCC of > 10.0×109/L was considered leukocytosis.
Treatment and follow-up
Clinical decisions were made by a senior surgeon at the moment of diagnosis and the patients with RD were treated according to our institutional protocol for DD (Fig. 1). In cases where the initial diagnosis was uncertain or the clinical condition of the patient justified it, emergency surgery was performed.
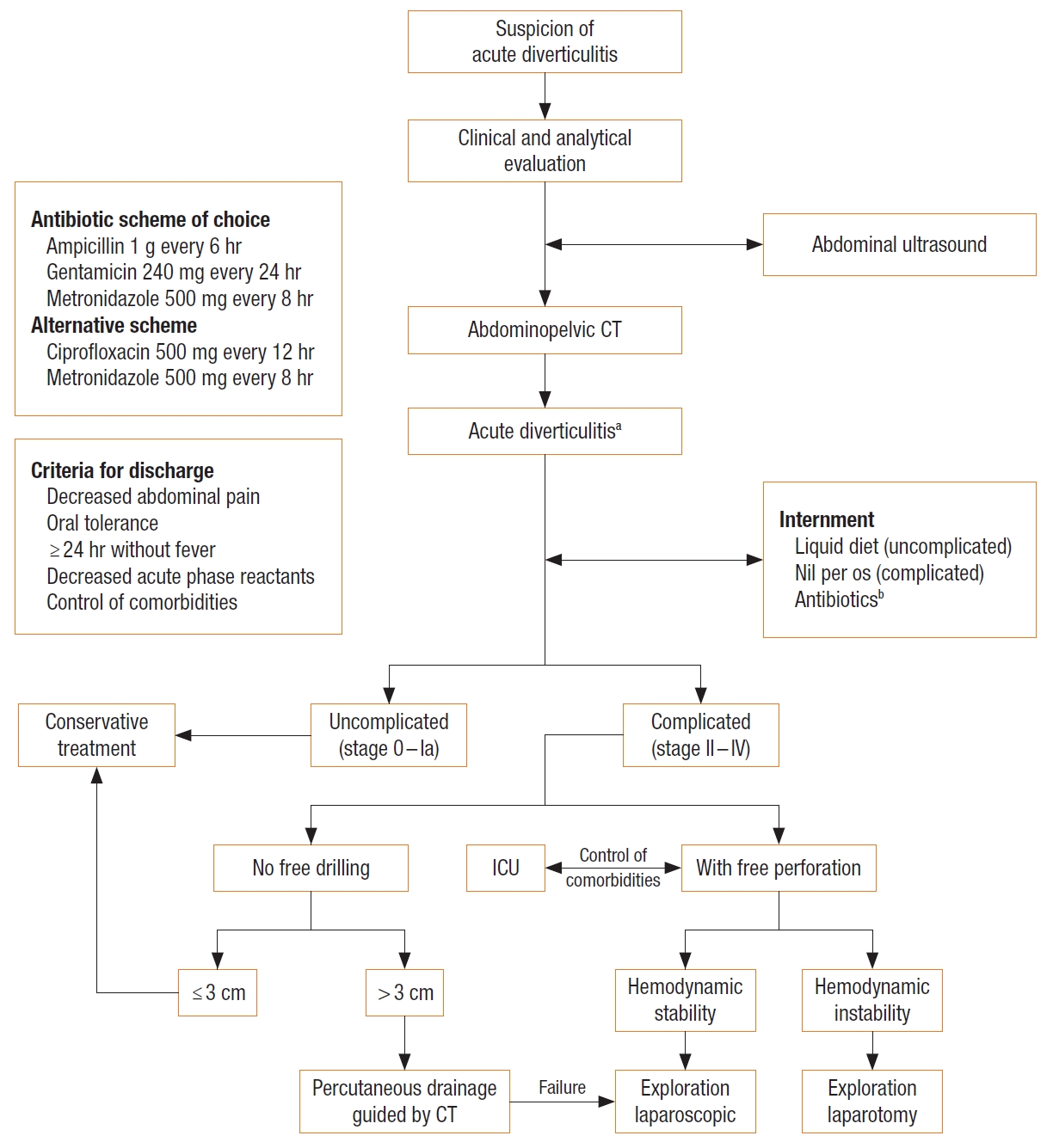
Institutional algorithm for the diagnosis and treatment of acute diverticulitis. CT, computed tomography; ICU, intensive care unit. aModified Hinchey classification by Wasvary. bAntibiotics adjusted according to the renal function.
Following discharge from the hospital, follow-up was performed by an experienced member of the General Surgery Service. We gathered the follow-up information from hospital records or by contacting patients or their relatives by phone.
Statistical analysis
The categorical variables were expressed as frequency (percentage), continuous variables as mean value±standard deviation or median (range). Mann-Whitney test was used to compare the continuous variables, and the chi-square test to compare categorical variables. A P-value of < 0.05 was considered significant. For statistical analysis, IBM SPSS ver. 26 (IBM Corp) and GraphPad ver. 7 (GraphPad Software) were used.
RESULTS
Demographic characteristics of patients
A total of 410 patients were included; 3.9% (n=16) had RD, confirmed by images or during surgery (Table 1). Of these patients, 62.5% (n=10) were male and the mean age was 40.7±11.7 years. Thirteen were Caucasian and three were Native American. This group was younger in comparison to the LD group (40.7±11.7 years vs. 65.4±12.7 years, P=0.001). Also, they presented a lower BMI (25.5±5.7 kg/m2 vs. 29.3±4.2 kg/m2, P=0.01), comorbidity rate (18.8% vs. 33%, P=0.01), CCI (0.4 vs. 2.8, P=0.02) and a lower severity of the stage according to the Hinchey classification (P=0.001) (Table 2). There were no significant differences between both groups in relation to sex (P=0.95). However, colonic diverticulitis was more common in males.
Presentation
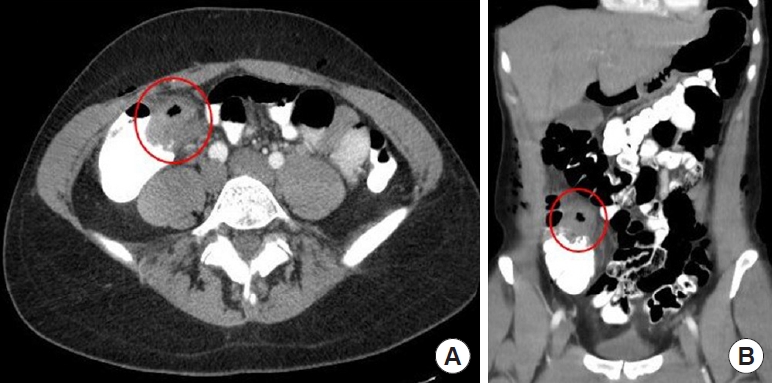
All the patients with RD had abdominal pain, with an average length of 47.3 hours. However, there was no significant difference between both groups in relation to the length of symptoms until consultation (P=0.05). Only 1 patient with RD presented right-hypochondrium pain with no radiation, associated with a palpable mass, simulating acute cholecystitis (Fig. 2). The main signs and symptoms are shown in Table 3. The initial clinical suspicion of acute appendicitis (AA) was present in 12 patients with RD.
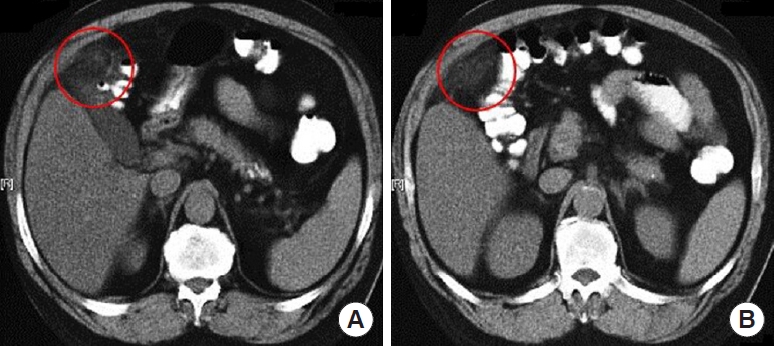
Computed tomotraphy of the abdomen and pelvis showing (A) a severe inflammatory process of diverticular origin at the level of the hepatic flexure of the colon (red circle), and (B) pericolonic fat involvement and thickening of the colon wall (red circle).
According to the distribution of diverticula, 75.0% were located in the sigmoid colon, 21.1% in descending colon or splenic flexure, 2.7% in ascending colon or cecum, and 1.2% in hepatic flexure.
Laboratory findings
Blood tests at the moment of consultation show leukocytosis in 93.8% (n=15) of patients with RD, with a mean value of 13.5×109/L. Twelve patients (75.0%) had a neutrophil count of > 70% with a mean value of 74.1%. Seven (43.8%) had a lymphocyte count of < 20% with a mean value of 18.4%. The levels of CRP presented a mean value of 56.4 mg/dL. We found a significant difference for NLR in the group of RD in relation to Hinchey stage, with a mean value of 3.1 for the stage O; 5.8 and 7.6 for the stages Ia and Ib, respectively (P=0.01). However, there was no significant difference between both groups regarding the laboratory findings (P=0.23).
Ultrasound findings
All the patients with RD underwent abdominal ultrasound at the moment of emergency consultation. Pericolonic fat alteration was found in 75.0% (n=12) and colon wall thickening was found in 68.8% (n=11) of the cases. A diverticulum image was described in 25.0% (n=4) of radiological reports and it was not possible to visualize the cecal appendix in 68.8% (n=11) of patients.
Tomographic findings
In 14 patients with RD, the study was completed with abdominal CT. Pericolonic fat involvement and thickening of the colon wall were seen in all patients. Mesenteric fat involvement was mild in 71.4% (n=10) and intense in 28.6% (n=4); and in 18.8% (n=3), a thickening of the right lateroconal fascia was observed. Colon wall thickening was mild (3–5 mm) in 4 patients, moderate (5–15 mm) in 7 patients, and marked (> 15 mm) in 3 patients. The diverticulum was seen in 13 cases (92.9%) (Fig. 3) and 1 patient presented a pericolic abscess of 3 cm, which received conservative treatment.
Treatment and follow-up
In the group of RD, 13 patients were treated conservatively and in 3, the diagnosis was erroneously confirmed during surgery. Of the patients who underwent surgery, 2 underwent only abdominal ultrasound, while the 3rd patient had a preoperative CT suggestive of cecal inflammation which affected the appendix. In these cases, the preoperative diagnosis was AA and the surgical approach was laparoscopic. In 2 patients, an appendectomy was not performed, because an inflammatory mass was detected on the external wall of the ascending colon without appendicular involvement; while in the 3rd case, the involvement of the appendiceal base was confirmed and prophylactic appendectomy was performed, with no findings of malignancy in the histopathological study.
After the surgery, the patients continued with intravenous antibiotics until the moment of hospital discharge, which was given when they presented clinical and analytical improvement. Two patients with RD were treated on an outpatient basis and the rest had a shorter hospital length of stay compared to the group of LD (3.2±1.2 days vs. 5.3±3.4 days; P=0.01).
Following the hospital discharge, all the patients continued with oral antibiotics (amoxicillin and clavulanic acid 875 mg/125 mg every 12 hours and metronidazole 500 mg every 8 hours). The length of antibiotic treatment presented a mean of 11±3.5 days. Only 2 patients were treated with the alternative antibiotic scheme due to allergy to penicillin.
With regard to the follow-up, the group with RD presented a mean of 22.8±8.4 months. However, 2 patients were lost to follow-up. Colonoscopy was performed in a period greater than 8 weeks after the acute episode in 11 patients, detecting diverticula in the right colon in all of them. None of the cases presented any signs of diverticulitis.
The rate of recurrence was significantly lower in comparison to that of the LD group (6.3% vs. 12.2%; P=0.001), and it occurred in a single patient who was initially treated conservatively.
DISCUSSION
The Western countries are characterized by having a heterogeneous population that has migrated from the East, so the incidence of DD might increase considerably in the future [5]. However, no studies have demonstrated an alignment in the incidence between the immigrants and the native population.
Argentina is considered as a “country of immigration,” in relation to the strong impact that various migratory flows have caused over the native composition of the population, especially of European origin, followed by American neighboring countries, and to a lesser extent Asian countries [9]. The 2010 census estimated that 3.4% of people of Asian descent lived in Córdoba [10] and this could explain why our study has no Asian patients.
Reports have indicated that RD incidence in Asian population is 55% to 70%, whereas in Caucasians is 1% to 2% [11]. In this series, the incidence in Caucasians was 3.2%, being above that reported in the literature. Besides, it is more frequent in males, with a male to female ratio of 2.0:1 to 3.2:1; and it affects mainly young population between 32 to 53.1 years old [11, 12]. Our findings were similar.
Pathologically, left-sided DD consists of pseudodiverticula and it has commonly been related to aging and a low dietary fiber intake, as risk factors [13]. On the other hand, the pathogenesis of DD is controversial and reports have indicated that they are congenital or “true” diverticula, with herniations that affect all the layers in the colonic wall and are not associated with aging [14]. In this cohort, the patients with RD were significantly younger than patients with LD, a fact that would support the theory that RD has a congenital nature.
The anatomic distribution of diverticula seems to vary according to race. In Caucasians, 75% are located in the sigmoid colon, 11% in the splenic flexure or descending colon, 6% in the transversal colon, and only 8% in ascending colon or hepatic flexure [15]. This study showed that 21.1% were located in splenic flexure or descending colon and 3.2% in the right-sided colon.
On a clinical basis, it is difficult to distinguish RD from appendicitis, due to the fact that signs and symptoms are similar at the moment of presentation, especially in young patients [16]; which requires a differential diagnosis between these 2 pathologies to determine a therapeutic plan and avoid unnecessary surgeries, due to a misdiagnosis of AA [17]. In this cohort, AA was suspected in 75% of the cases. However, only 3 patients required surgery.
Currently, few studies have reported differences between RD and appendicitis. It has been described that the prevalence of pain migration was significantly higher in AA (39.2%–82%) compared to that of RD (15.4%–54%) [18, 19]. In this cohort, 63% of patients with RD presented abdominal pain migration, similar to that reported. Also, it could be confused with other diseases such as cholecystitis, gastritis, or peptic ulcer, although less frequently [20, 21]. We found a patient with right hypochondrium pain who simulated cholecystitis, but without findings on biliary pathology ultrasound.
Among other differences existing with appendicitis, reports have indicated that the duration of abdominal pain is longer in RD (68.4±23.3 hours vs. 29.8±20.2 hours, P<0.01) [19]. In this work, the time interval from the onset of abdominal pain until admission was 47.3±10.2 hours, similar to the information reported in the literature.
On the other hand, reports indicate a low incidence of nausea and vomiting in RD (8%–16%) compared to AA (32%–72%) [18], which differs from our findings, since in this study 44% of patients with RD presented nausea and vomiting.
With regard to fever, it was reported that its prevalence is not significantly different between AA and RD (14.4% vs. 15.0%, P=0.87) [18], similar to our results.
From the analytical approach, reports show that leukocytosis in RD is in 58% to 69.2%; and in AA, it is presented in 68.5% to 90% [12, 22]. In this cohort, 94.0% of patients with RD presented a leukocyte count above 10.000×109/L. However, the number of studies that have demonstrated its utility to differentiate both illnesses is limited, is this still a controversial topic.
In a recent study, we demonstrated the utility of NLR to predict complicated LD, with a cutoff point of ≥ 4.2 as the best value of diagnostic approximation [23]. Similarly, in this cohort we found that NLR showed statistical significance in relation to the severity of Hinchey stage, validating its applicability as a reliable, noninvasive, and cost-effective marker in the early diagnosis of complicated DD.
After the clinical and analytical evaluation, radiological images are required to continue the assessment of patients with right lower quadrant pain. Reports indicate that the ultrasound performed by an experienced radiologist for RD diagnosis shows a sensitivity of 91.3% and a specificity of 99.8% [24], but other studies have not confirmed this information. Our diagnosis rate using the ultrasound scan was very low (25.0%), similar to the results in other studies [12]. CT is another technique used in case of colonic diverticulitis suspicion, which presents a specificity of 90% to 95%, but in some cases, it may establish a wrong diagnosis and confuse RD with an appendicular abscess, Crohn disease, epiploic infarction, or colon cancer [21]. CT has been reported to presents a preoperative diagnosis rate in RD of 78.4% to 83.3% [25, 26]. According to our institutional protocol, we completed the tomographic evaluation in 87.5% of our patients, with a preoperative diagnosis rate of 93%.
There are no established guidelines for the treatment of RD, and current recommendations commonly refer to the left-sided colon. It was shown that RD patients are less likely to have serious complications and therefore more likely to respond to conservative treatment with less need for emergency surgery and shorter length of hospital stay [5]. In our study, we found that the rate of complicated diverticulitis was significantly lower in the group of patients with RD (6.3% vs. 12.5%, P=0.04). Furthermore, the conservative treatment was feasible in most patients due to the high rate of preoperative diagnosis.
The observed differences in disease severity between RD and LD may be explained by various factors. Inflammation in RD is more likely to be localized to a single diverticulum, while the inflammation in LD is commonly described as diffuse, involving multiple diverticula with wall thickening [27]. Anatomically most of the right colon is retroperitoneal which may limit the spread of inflammation, while the sigmoid colon, the most common site of LD, is intraperitoneal which may result in the easier spread of inflammation [28].
Although it varies by investigator, the recurrence rate in RD after conservative treatments is shown to be approximately 3% to 15% [22]. During the follow-up, only 1 patient was detected recurrence but had no complications and a presented good evolution with the conservative treatment.
There is controversy as to the surgical treatment for RD found at the moment of surgery. Mainly 4 surgical strategies can be considered; diverticulectomy with appendectomy, ileocecal resection, right hemicolectomy, and prophylactic appendectomy [16]. From the patients who required surgery, 1 underwent prophylactic appendectomy for severe local inflammation, while in the other 2 patients, the exploration laparoscopic detected diverticulitis of the ascending colon that did not require appendectomy. However, appendectomy, alone, could be associated with an increase in the rate of recurrence and the need for a new surgery since this approach does not offer a definite treatment for RD [12]. None of the 3 patients operated on did present recurrences, nor complications during the follow-up, but it is difficult to establish the true recurrence rate in patients operated, due to the fact that in our experience we had a predominance of cases treated conservatively.
The limitations of our study were its retrospective design and that a small number of patients with RD treated in a single center in Córdoba, Argentina were recruited. However, the RD is considered a rare pathology in our environment. In addition, risk factors for diverticulitis, including eating habits, physical activity, and defecation habits, were not included in our research. Therefore, the results of our study cannot be generalized to the entire population.
In our environment, the incidence and complication rate of RD are lower compared to LD. Conservative treatment predominated in RD. However, the evidence is very limited and further studies are required to provide robust data.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
None.
AUTHOR CONTRIBUTIONS
Conceptualization: RMPH, DAPP; Formal analysis: RMPH, DAPP; Supervision: JEL, HAPM, RP, MP; Writing–original draft: RMPH, DAPP; Writing–review & editing: JEL, HAPM, RP, MP. All authors read and approved the final manuscript.