Effect of reinforcement suture on anastomotic healing in rectal and sigmoid tumor resections, single-center experience: a retrospective case-controlled study
Article information
Abstract
Purpose
Healing of colorectal anastomosis is burdened with a number of risk factors and the development of leak carries with it higher morbidity and mortality associated not only with worse quality of life but also worse oncological outcomes. The purpose of the study was an evaluation of the effect of reinforcement suture in the healing of stapler rectal anastomosis and risk factors for anastomotic leakage in mid, upper rectal and sigmoid tumors.
Methods
A total of 357 patients who underwent curative resection for rectal or sigmoid tumor over 5 years were analyzed. Risk factors, baseline demographic and clinical data, and presence of leak were recorded. All statistical analyses were carried out at a significance level of P<0.05.
Results
One hundred fifty-one patients met the inclusion criteria for the study. The overall incidence of leak was 2.8% in the group with reinforcement suture and 6.3% in the group without reinforcement suture. Because of the low incidence of leak the results were not statistically significant. Further results also show that none of the monitored parameters (age, weight, body mass index, tumor distance, preoperative radiotherapy, tumor stage, type of surgery) were significant predictors of anastomotic leakage.
Conclusion
Although the reinforcement suture is a safe and simple option for anastomosis protection after rectal or sigmoid resections, its statistical significance was not demonstrated in our study.
INTRODUCTION
According to the latest global statistics, colorectal cancer is the fifth most common cancer and the fourth most common cause of cancer death [1]. The mainstay of surgical treatment in locally advanced rectal cancer is tumor-specific mesorectal excision (total or partial) [2], whereas in low-risk cancer (maximum stage T1, sm1), local surgery is acceptable because there is an acceptable risk of lymph node invasion (3.1%) [3] and local recurrence (6.7%) [4].
Anastomotic leakage (AL) is still a dreaded complication that causes considerable concern to surgeons, especially coloproctologists. AL occurs more frequently in rectal (8%–14%) than in colonic resections (3%–7%) [5]. The occurrence of AL implies serious complications significantly affecting morbidity and mortality. According to Park et al. [6], colonic leakage was not associated with an impact on 5-year disease-free survival, but there was a significant difference in patients after rectal resection (67.0% vs. 76.6%, P= 0.005, respectively).
In our experience, the basic requirements for proper anastomotic healing are adequate blood supply to the anastomosed stumps, absence of anastomotic tension, prevention of sepsis, prevention of stool contamination, and prevention of distal obstruction. Nutritional status is also important. There are many other options in the literature to prevent or reduce leakage, these include various forms of endoluminal devices, transrectal sutures loaded intraluminally or intraabdominally, or the currently preferred indocyanine green fluorescence to assess organ perfusion. The aim of this nonrandomized study was to evaluate the effect of intraabdominally applied seromuscular reinforcement sutures on anastomotic healing after stapler resections for upper and middle rectal and sigmoid tumors.
The main attributes of the study were the incidence of leakage in the group with and without reinforcement suture and the evaluation of the effect of reinforcement suture on the development of leakage. A secondary outcome was the evaluation of risk factors for AL formation.
METHODS
Ethics statements
The study was performed in accordance with the 1964 Helsinki Declaration. All patients were guaranteed anonymity, provided consent for data processing and publication and signed informed consent. The research approval from Institutional Review Board was not required according to institutional policy.
Database
Patients for inclusion in the study were identified by searching the institutional database. The database contains patient demographics, admission and dismissal data, diagnoses based on the International Classification of Diseases, initial disease stage based on the classification system of the Union for International Cancer Control, operating room procedures, laboratory and radiology results, and definitive histology.
Patients
Using an institutional database, we identified patients who were diagnosed with rectal or sigmoid tumors between January 1, 2016 and December 31, 2020 and met the primary curative resectability requirement with subsequent stapled anastomosis on the rectum, realizing the differences in healing after resections of rectal and sigmoid tumors. Records were individually reviewed by the first author to collect data on age, body mass index (BMI), American Society of Anesthesiology (ASA) physical status (PS) classification, TNM stage, lymph node count, the distance of tumor from the anal verge, presence of a corner seromuscular suture, and presence of leak according to the grading system proposed by the International Study Group for Rectal Cancer. Patients who had undergone diversion, ileo or colostomy, or rectal amputation were excluded.
Statistical analysis
Statistical analyses were carried out using IBM SPSS ver. 22.0 (IBM Corp). Statistical significance was tested using the Fisher exact test for categorical variables and the Mann-Whitney U-test for continuous variables. Normal distribution was verified by the Shapiro-Wilk test. Binary logistic regression was used to analyze potential factors associated with the leak. All tests were carried out at a significance level of P< 0.05.
Preoperative management
The bowel was prepared preoperatively with 4 L of polyethylene glycol (Fortrans, Beaufour Ipsen Industrie) and intravenous antibiotics were administered to the patients 30–60 minutes before incision, a combination of ampicillin/sulbactam and metronidazole in a prophylactic dose. The next application was after 4 hours and the next 4 hours, for a total of 3 times.
Operative management
In the case of conventional surgery, we prefer the lateral to medial approach. The abdominal cavity is accessed by a midline incision from the umbilicus to the symphysis. The small bowel is retracted to the right and a lateral incision is made along the white line of Toldt with the left colon retracted medially. The incision is extended from the pelvis to the left upper quadrant. The exposed plane of the avascular tissue allows us to perform a blunt dissection, thus releasing the bowel from the retroperitoneum and exposing the ureter and the gonadal vessels within. Subsequently, the left colon is lifted and is pulled to the patient’s left, exposing the medial aspect of the left colon mesentery over the aorta. The superior rectal artery is identified at the level of the origin from the inferior mesenteric artery and divided between ties. In the case of laparoscopic surgery, we prefer the reverse, medial to lateral approach. At the level of the rectum, we proceed with tumorspecific mesorectal dissection according to current oncosurgical principles. We resect upper rectal tumors at the cost of partial mesorectal excision, tumors located lower down at the cost of total mesorectal excision. If necessary, to perform tension-free anastomose, we take down the splenic flexure, but usually a thorough lateral incision and release of the left colon up to the splenic flexure are sufficient, or in the case of an oral rectal tumor we can help by releasing the remaining rectum from the pelvic excavation. The anastomosis is always performed by the circular double stapling device in an end-to-end fashion, the stapled donut is checked, and the silastic drain is placed along with the anastomosis.
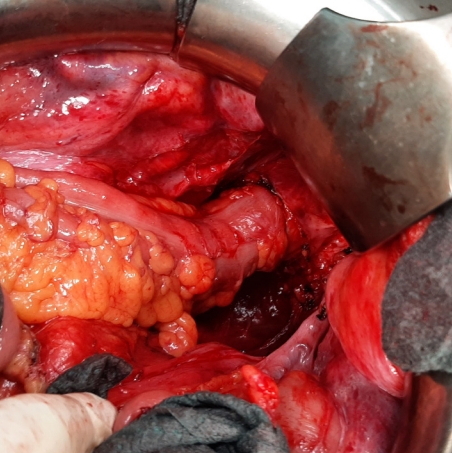
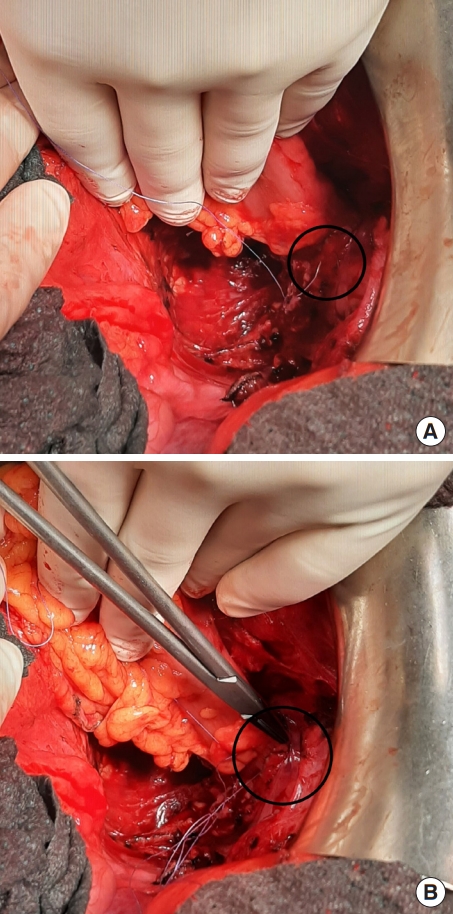
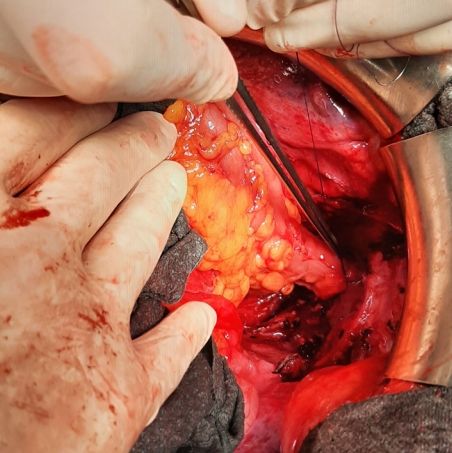
Regardless of whether the surgery is open or laparoscopically assisted, after construction of the stapler circular colorectal anastomosis, we suture its margins, left and right, which are most at risk of inadequate blood supply, with a separate seromuscular suture using absorbable Saphil 4/0 suture (Safil, B.Braun Surgical).
The suture is loaded about 1 cm below and then above the anastomosis in the manner of a U-stitch and tightened, thus incorporating the original stapler suture in the sense of a second layer known from anastomoses in the free bowel. The condition is the availability of the anastomosis from the intraabdominal cavity and the possibility of loading the suture, therefore, it was indicated for surgery of sigmoid, proximal rectal and some of the mid-rectal tumors specially favored for the female pelvis (Figs. 1–3).
Perfusion testing by indocyanine green was not routinely introduced during the period. This was subjectively assessed by the quality of blood flow during resection of the specimen (cold steel test) and pulsations in the mesentery. The intraoperative air-leak test was performed purely at the discretion of the surgeon.
All 6 operators were experienced coloproctologists with at least 7 years of experience. The cases were distributed over the entire period of data collection without being influenced by the learning curve factor.
RESULTS
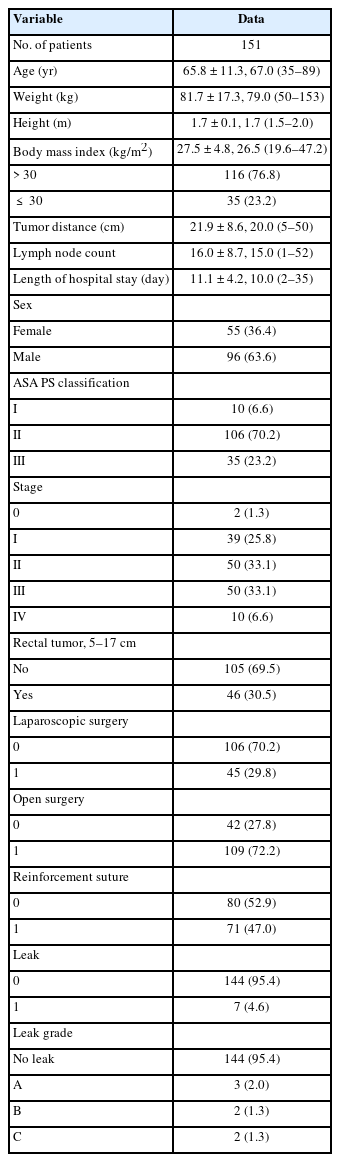
A total of 357 patients underwent curative resection for the rectal or sigmoid tumor in the reporting period, of which 151 patients met the inclusion criteria. There were almost twice as many male patients as female patients (96 vs. 55). Mean age was 65.8 years, weight 81.7 kg with an average BMI of 27.5 kg/m2. A quarter of the subjects were overweight, with a maximum BMI of 47.2 kg/m2. The risk of surgery and anesthesia classified by ASA PS classification was low in 76.8% of patients. ASA PS classification I, II, and III were 6.6%, 70.2%, and 23.2%, respectively. The pathological stage was evenly divided into stages I, II, and III, accounting for 25.8%, 33.1%, and 33.1%, respectively. Stage 0 (as a pathological complete response) and IV (patients with resectable or border resectable metastases) accounted for 1.3% and 6.7%, respectively. The mean lymph node count was 16 (range, 1–52) and the mean length of hospital stay was 11.1 days (Table 1).
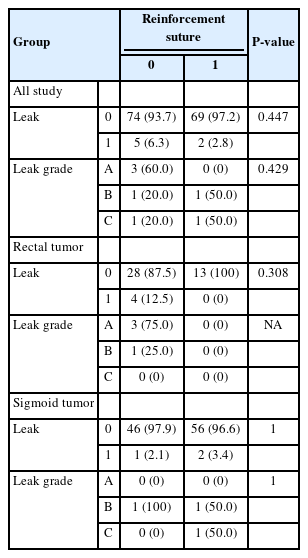
When comparing the groups with and without the reinforcing suture, leakage was higher in the group without intervention (2.8% vs. 6.3%), but the results did not reach statistical significance. Even when patients were divided into groups with rectal and sigmoid tumors, no statistically significant results were found due to the low incidence of a leak and the small definitive sample size. Interestingly, not a single dehiscence was detected in the group with rectal tumors, and in the group with sigmoid tumors 2, even 1x type C. This series further demonstrates that the reinforcement suture, although unable to prevent more severe leakage, does not allow low-grade leakage to occur (Table 2).
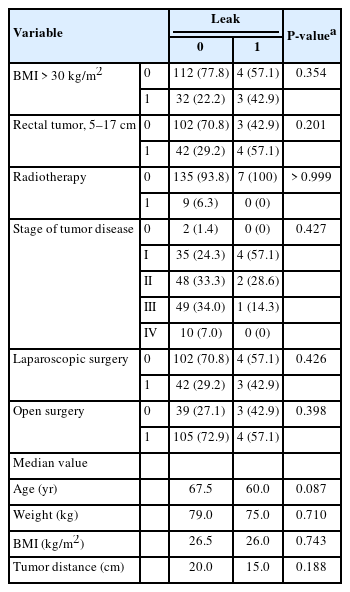
Further evaluation of possible risk factors (age, weight, BMI, tumor distance, preoperative radiotherapy, tumor stage, and type of surgery) shows none of them to be significant risk predictors for AL. Incomplete stapled donut as a risk factor for the leak was not included in the report because in the whole report, according to the relevant surgical protocols, it was not present, each time it was a complete, circular ring of tissue so it was not expected to affect statistical significance (Table 3).
There were a total of 13 revisions in the dataset, which corresponds to 8.6%. The most common reason was the dehiscence of laparotomy and leak, both 5 times in total (3.3%), followed by 2 times for early mechanical adhesive ileus (1.3%) and once for hemoperitoneum (0.7%). No colovesical, colovaginal, or colocutaneous fistula developed in this or the previous 5-year period. Stenosis of the anastomosis occurred 4 times, once in the reinforcement suture group, none of them required surgical revision and, as histologically benign, was successfully resolved by endoscopic balloon dilatation.
There were 5 revisions for type A and B leak (38.5% of all revisions), although in type A leak conservative approach should be taken (in the group of patients without protective loop ileostomy) revision was performed for a computer tomography finding suspicious of anastomosis dehiscence and mild clinical signs. An abdominal toilet was performed, if the leak was present, and if possible, the anastomosis was resewn and the loop ileostomy was performed. The same procedure was followed for type B leaks. Type C leak (15.4% of all revisions) required a more extensive procedure due to the dehiscence of more than half of the anastomosis circumference. The rectal stump was blindly closed and the colon descendens was diverted out as a terminal stoma in the sense of Hartman resection.
Investigation of the generally valid risk factors for anastomotic healing in patients where leakage occurred also failed to confirm to us their causal relationship. Diabetes mellitus (DM) was present in 1 patient, as well as 2 staplers firing, inflammatory bowel disease (IBD), steroid or nonsteroidal anti-inflammatory drugs use, and preoperative radiotherapy was not present in any patient. Males predominated in a ratio of 5:2 compared to females. The only thing that all patients had in common was a laboratory worse nutritional status with no significant clinical manifestations or weight loss. Unfortunately, since there is only 1 patient with DM, and 2 with 2 staplers firing among the risk factors in the leakage cohort and no other risk factors are present, it is not possible to analyze the data and demonstrate a significant relationship (Table 4).
DISCUSSION
International Study Group of Rectal Cancer defined AL as a defect in the intestinal wall integrity at the colorectal or coloanal anastomotic site (including suture and staple lines of neorectal reservoirs), which leads to communication between the intraand extraluminal compartments. A pelvic abscess close to the anastomosis is also considered as AL. The group suggested grading the severity of AL based on its effect on the patients’ clinical management. Grade A was defined as a leakage that requires no active therapeutic intervention. Grade B was defined as a leakage that requires active therapeutic intervention but can be managed without relaparotomy. Grade C was defined as a leakage that requires relaparotomy [7]. AL can also occur in the presence of protective ileo or colostomy. The incidence of a significant leak is almost 14% carrying the higher risk in a patient with IBD, postradiation colitis, trauma, or ischemia. The rates of AL in colorectal surgery vary widely depending on several factors, particularly whether the anastomosis is intra- or extraabdominal (2.7%–8.7% and 3.6%–13.3%, respectively) [6].
To provide an objective assessment of the risk of AL a clinical scoring system is mandatory. The colon leakage score is unique to detect high-risk patients preoperatively and objectively assesses the necessity for diverting ileostomy or non-restorative surgery [8]. Dutch leakage score shows patients with a higher risk of AL requiring intensive clinical observation or radiological evaluation. This scoring system reduced the delay in diagnosis of an AL from 4 to 1.5 days [9].
The mandatory principles of good anastomotic healing as mentioned above are adequate blood supply to anastomosed stumps, no tension of the anastomosis, prevention of sepsis, fecal contamination, prevention of distal obstruction, and nutrition status. The dogma that stapled anastomose compared to hand-sewn is safer was not evaluated. The highest level of evidence exists for the equality regarding AL, without confirmation that one technique is being superior to the other [10]. Following this statement, stapling techniques should be of preference because the technique is uniform, easy to learn, making it ideal for teaching young surgeons.
In a nutshell, risk factors for AL are divided into preoperative (male sex, BMI, radiochemotherapy, use of corticosteroids and immunosuppressive drugs), perioperative (level of anastomosis, multiple stapler firings, precompression before stapler firings, diameter of circular stapler, blood loss, and anastomotic tension) and postoperative (diverting stoma, abdominal drains) [11].
Healing of anastomosis with phases of inflammation, proliferation, and remodeling is similar to healing in other parts of the body. Collagen synthesis, which is the most important component of healing, predominates physiologically about 1 week after surgical trauma. At this critical time, the anastomosis is at high risk of damage, causing most leaks to occur between postoperative days 5 and 8. After approximately 4 weeks, collagen reorganization is almost complete, and wound healing is approximately 90% complete. A number of factors influence anastomosis healing, including control of the systemic disease. Regarding systemic disease, both bursting pressure and collagen content of colonic anastomoses are negatively affected in the presence of abdominal sepsis [12]. To prevent impaired healing, reinforcement methods have been investigated. In a porcine intestinal anastomosis model, the use of reinforcement significantly increased anastomotic burst pressure from 53 to 83 mmHg [13].
The use of reinforcement aims to reduce the tension in the anastomosis, cover the gaps between sutures causing stool contamination, and reduce bleeding. It can be applied in an intraabdominal or transanal way. Circular bioabsorbable Seamguard (CBSG, W.L. Gore and Associates) is a fully absorbable, biodegradable film that is inserted with a circular stapler during the reconstructive phase and covers the anastomosis with staple clips.
The results are promising; Portillo and Franklin [14] reported leakage in 3.4% of 117 patients, 2 recovered spontaneously, and 2 required protective ileostomy. Senagore et al. [15] compared patients with and without this foil with the result that the incidence of a leak is comparable (11.4% vs. 12.6%). The same results without statistical significance were obtained and published by Placer et al. [16], in a study of 304 patients.
The biodegradable colorectal drain (C-seal) works on the same principle. Unfortunately, the high cost of these devices impedes their wider clinical use. A current point of interest are biodegradable stents, which are clinically tested, but their biggest disadvantages are the high risk of migration. The latest modality is tissue adhesives, which are now being experimentally tested in an animal model.
A purely surgical option is to stitch over the anastomosis with absorbable sutures by intraabdominal or transanal approach and to varying degrees. It is believed that the reinforcement of the stapler line by the mucosa creates a barrier against intestinal bacteria with collagenolytic enzymes. Then there is a tension-free anastomosis effect. Baek et al. [17], in his study at only laparoscopic rectal resections, concluded that the risk of leakage was practically the same, but with a slight advantage for the suture group (6.4% vs. 7.9%), and in particular reported that the number of protective ileostomies was significantly lower (12.8% vs. 30.2%). Kim et al. [18] also reported good results for this reinforcement method. Although a small sample (63 patients with rectal cancer, intracorporeal reinforcing sutures were used in 15 cases, transanal in 48 cases) shows only 1 leak healed by conservative treatment. We see the high number of protective ileostomies (48 cases) as a disadvantage.
Our results are more or less the same. No significant dependence was found; however, the number of lesions was lower in the group with reinforcement suture, unfortunately only in type A, as the methods are absolutely equivalent for type B and C lesions requiring our attention and intervention.
This study has several limitations. First, it is a retrospective study conducted at a single center. Secondly, it is not a randomized controlled trial. The placement of a reinforcement suture or not was at the discretion of the surgeon based on a subjective assessment of the potential risk of anastomosis leakage, so there was a selection bias between groups. Next, we compared a group of patients with rectal and sigmoid tumors with different healing rates, but we tried to minimize this bias by selecting patients in order to select a group suitable for intraabdominal reinforcement suture. And finally, a small group of patients meeting the inclusion criteria did not allow to reach statistically significant conclusions.
In our opinion, studies should focus on techniques that are easy to learn, easy to perform, and have high reproducibility. Additional stapler anastomosis sutures meet these criteria, are simple to perform, inexpensive, and their influence on anastomosis healing was assumed. They can be applied intraabdominally or transanally for super low anastomoses. They are widely applicable in laparoscopic or conventional surgery. Unfortunately, its effect on anastomosis healing has not been confirmed, at least not with statistical significance, and it cannot be recommended otherwise than that its use is purely at the discretion of the surgeon.
Any beneficial method that brings improvement in proctology is desirable and therefore further randomized multicenter controlled research is still needed.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
This study was supported by Ministry of Health, Czech Republic; conceptual development of research organization (FNOl, 00098892).
AUTHOR CONTRIBUTIONS
Conceptualization: TR, PZ, DK; Data curation: TR, PS, MS; Formal analysis: all authors; Visualization: TR, PZ, RV; Writing–original draft: TR, RV; Writing–review & editing: all authors. All authors read and approved the final manuscript.
Acknowledgements
Special thanks to Mrs. Jana Zapletalova, Department of Medical Biophysics, Faculty of Medicine and Dentistry, Palacky University Olomouc for statistical work.