Laparoscopic right hemicolectomy with aortocaval lymphadenectomy, and pelvic peritoneum partial resection for ascending colon cancer
Article information
Abstract
The aim of this video is to present the procedural details of laparoscopic right hemicolectomy with aortocaval (infrarenal aortic bifurcation) lymphadenectomy, partial resection of the pelvic peritoneum (peritoneal carcinomatosis index, 3), and hyperthermic intraperitoneal chemotherapy in a patient who received neoadjuvant chemotherapy for stage IVc colorectal cancer. The total operation time was 290 minutes, and the patient was discharged on a postoperative day 13 without any complications. No postoperative complications occurred until postoperative day 60. The pathological stage of the tumor was determined to be T3N2bM1c. The pelvic peritoneal nodule was pathologically confirmed as a metastatic lesion. Among the 12 harvested aortocaval lymph nodes, 6 were metastatic lymph nodes. The minimally invasive approach was safe and feasible in this highly selected patient with colon cancer, aortocaval lymph nodes, and peritoneal metastases.
The presence of multiple types of metastases or peritoneal metastases in colorectal cancer is defined as stage IVb or IVc, respectively, in the eighth edition of the American Joint Committee on Cancer tumor-node-metastasis staging system because of its poorer prognosis than stage IVa [1]. Although stage IV colorectal cancer represents the end-stage of disease progression, multidisciplinary treatment can lead to better survival than palliative chemotherapy alone, and cure in some cases when radical resection is involved [2–6]. Although minimally invasive surgery is prevalent for the treatment of colorectal cancer, its use in patients with synchronous distant lymph nodes or peritoneal metastases has rarely been reported. This video presents the procedural details of laparoscopic right hemicolectomy with aortocaval lymphadenectomy, partial resection of the pelvic peritoneum, and hyperthermic intraperitoneal chemotherapy (HIPEC) for a patient with colorectal cancer and synchronous distant metastases, for which neoadjuvant chemotherapy was used (Supplementary Video 1). This study was conducted in accordance with the tenets of the Declaration of Helsinki and its amendments. The Ethics Committee of Kyungpook National University Chilgok Hospital approved this study (No. KNUCH 2022-05-031). Written informed consent was obtained from the patient for publication.
A 60-year-old man was diagnosed with ascending colon cancer, and synchronous aortocaval lymph node (intrarenal aortic bifurcation) and peritoneal (single nodule at the rectovesical fossa) metastases were found on both abdominopelvic and chest computed tomography (CT) and 18-fluoro-deoxyglucose positron emission tomography CT scans (Fig. 1). The tumor was a moderately differentiated, microsatellite-stable, KRAS-mutated, and NRAS-wild type adenocarcinoma. While the serum carcinoembryonic antigen level (1.75 ng/mL) was within the normal range, the serum carbohydrate antigen 19-9 (151.3 U/mL) was elevated. The patient received 9 cycles of FOLFOX (5-fluorouracil/leucovorin/oxaliplatin) plus bevacizumab, following which the CT scan showed that the primary tumor and metastatic lesions had partially regressed. His carbohydrate antigen 19-9 level decreased and was within the normal range (15.1 U/mL). A multidisciplinary team determined radical resection of all the lesions and HIPEC as the appropriate management strategy. Additional 4 cycles of FOLFOX without bevacizumab were administered before surgery. During the preoperative meeting for informed consent, the patient was informed that the imaging studies could underestimate the extent of peritoneal metastases [7] and that the surgery would be carried out only when gross radical resection of all the lesions was possible. The patient underwent mechanical bowel preparation 1 day before surgery and received prophylactic antibiotics before the incision on the day of operation. After administration of general anesthesia, the patient was placed in the lithotomy position. On initial laparoscopic exploration of the abdominal cavity through the umbilical camera port, the stage of the ascending colon cancer was clinically assessed as T3. There was a 1 cm-sized single metastatic nodule on the parietal peritoneum at the rectovesical fossa. Four 5-mm trocars were placed in each quadrant. The surgeon and an assistant, for holding the camera, stood on the left side of the patient during the entire procedure.

Abdominopelvic computed tomography and 18-fluoro-deoxyglucose (FDG) positron emission tomography (PET) computed tomography (CT) show ascending colon cancer (A), enlarged aortocaval lymph nodes (B, C), and peritoneal nodules at the rectovesical fossa (D). Abdominopelvic CT and FDG PET-CT show (A, B) ascending colon cancer and enlarged aortocaval lymph nodes (yellow arrows), and (C, D) peritoneal nodules at the rectovesical fossa (white arrows).
Partial excision of the pelvic parietal peritoneum was initially performed for intraoperative pathological diagnosis and to determine whether HIPEC must be administered. The parietal pelvic peritoneum, on which the whitish nodular lesion was found, was dissected using monopolar scissors with clear resection margins and without injury to the bladder or rectum (Fig. 2A). Frozen section biopsy revealed metastatic adenocarcinoma. Because the microscopic omental spread of peritoneal metastasis is possible even in the absence of macroscopic omental nodules [8], en bloc resection of the total greater omentum was performed during right hemicolectomy. The greater omentum was dissected off the stomach using an ultrasonic dissector while preserving the gastroepiploic vessels. Dissection was facilitated by caudal retraction of the greater omentum by the operator and cephalic countertraction of the stomach. Right hemicolectomy was initiated by incising the parietal peritoneum at the retrocecal recess. A caudal-to-cephalad mesocolic dissection was performed up to the pancreatic head and duodenum on the medial side and up to the hepatocolic ligament on the superior side. Central vascular ligation and D3 lymphadenectomy were performed from the ileocolic vessels to the mid-colic vessels using an ultrasonic dissector. The ileocolic vein and artery were ligated at their origin, and the right branch of the mid-colic artery and veins branching into the right colon were ligated.
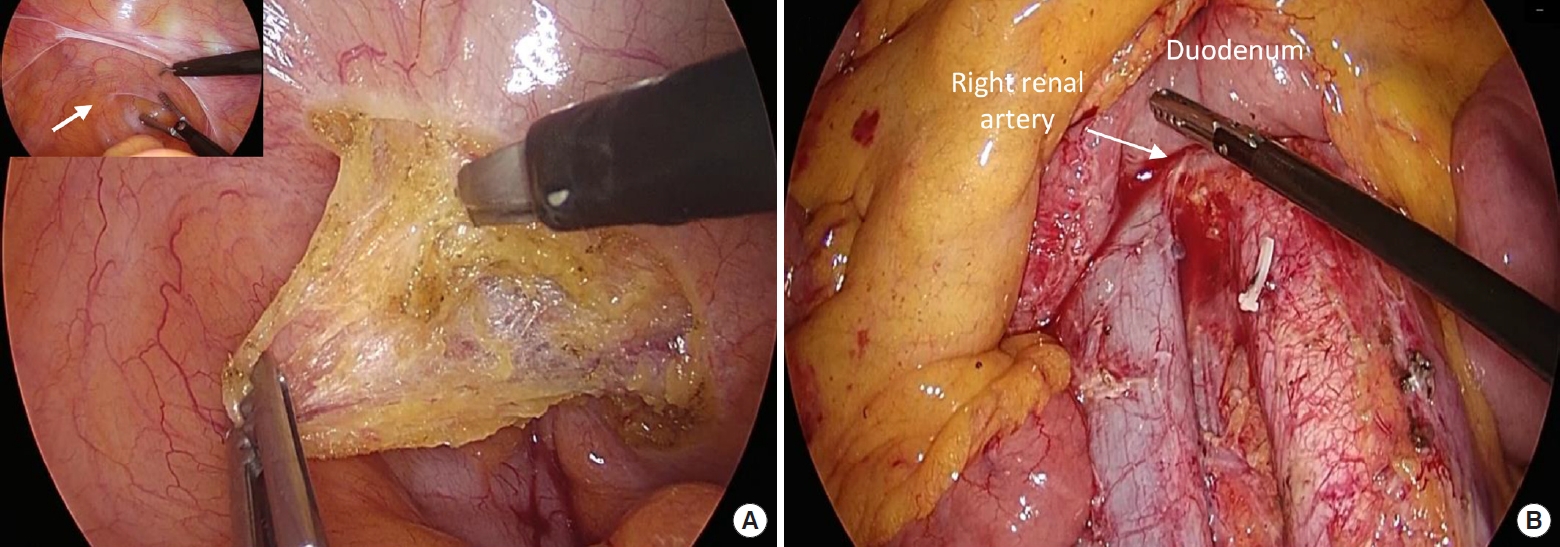
(A) Dissection of the parietal pelvic peritoneum including metastatic peritoneal nodule (arrow in inset). (B) Lymphadenectomy at the aortocaval area between the inferior border of the left renal vein and aortic bifurcation (arrow).
The Kocher maneuver was performed to approach the aortocaval lymph nodes. Complete mobilization of the right colon during right hemicolectomy and the Kocher maneuver allowed excellent exposure of the inferior vena cava, aorta, and aortocaval space. Metastatic lymph nodes were presented only on the anterior side of the inferior vena cava and aortocaval area. Lymphadenectomy was initiated from the inferior border of the left renal vein and dissection was performed down to the level of the aortic bifurcation (Fig. 2B). The small feeding vessels of the lymph nodes were ligated using surgical clips. The specimens were removed through umbilical mini-laparotomy, and extracorporeal ileocolic side-toside stapled anastomosis was performed. The last step of the operation was the administration of HIPEC for 90 minutes using 35 mg/m2 mitomycin C, which was heated to 41 to 42°C for 90 minutes. Circulation catheters were introduced into the abdominal cavity, of which the 2 input catheters were placed in the right upper quadrant and pelvic cavity, whereas the 2 output catheters were placed in the left upper quadrant and pelvic cavity.
The total operation time was 290 minutes, including the time required for HIPEC, and the total blood loss was 50 mL. The patient started oral intake of a small amount of clear water on postoperative day 2 and resumed a soft diet on a postoperative day 4. The patient was discharged on a postoperative day 13 without any complications, and no complications were observed until postoperative day 60. The pathological stage of the tumor was determined to be T3N2bM1c. There were metastatic cancer cells in the pelvic peritoneum and the serosa of the appendix (peritoneal carcinomatosis index, 3). There were 2 metastatic regional lymph nodes in the pericolic area among the 37 harvested regional lymph nodes, while there were 6 metastatic lymph nodes among the 12 harvested aortocaval lymph nodes. Postoperative systemic chemotherapy using the same regimen as preoperative chemotherapy was initiated in the 7th postoperative week.
A minimally invasive approach was safe and feasible in this highly selected patient having colorectal cancer with aortocaval lymph nodes and peritoneal metastases. R0 resection has been proven to contribute to the survival outcomes of stage IV colorectal cancer [9, 10], and these findings were reproduced in selected patients with multiple metastases [11]. The risk of radical surgery for multiple metastases and the difficulty of postoperative recovery can be reduced when minimally invasive surgery was applied. The findings of this study suggest that it is feasible for colorectal surgeons to enhance their skill in managing complex stage IV disease cases, as minimally invasive techniques have advanced during the last 3 decades. Further studies are needed to validate the clinical implications of this approach.
SUPPLEMENTARY MATERIALS
Supplementary video clip 1 is presented online (available at https://doi.org/10.3393/ac.2022.00780.0111).
Laparoscopic Right Hemicolectomy with Aortocaval Lymphadenectomy, and Pelvic Peritoneum Partial Resection for Ascending Colon Cancer
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
None.
AUTHOR CONTRIBUTIONS
Conceptualization: SYP; Data curation: HK, ANS; Formal analysis: SYP; Investigation: SYP, ANS; Methodology: SYP, HK; Project administration: SYP, HK; Resources: SYP; Validation: SYP; Visualization: SYP, HK; Writing–original draft: SYP, HK; Writing–review & editing: all authors. All authors read and approved the final manuscript.
Acknowledgements
The authors thank the patient cited in this video article who provided written informed consent for the publication of his clinical information and video. We also thank Suhyun Park for her sincere efforts in assisting with the operation.
