Can the Heald anal stent help to reduce anastomotic or rectal stump leak in elective and emergency colorectal surgery? A single-center experience
Article information
Abstract
Anastomotic and rectal stump leaks are feared complications of colorectal surgery. Diverting stomas are commonly used to protect low rectal anastomoses but can have adverse effects. Studies have reported favorable outcomes for transanal drainage devices instead of diverting stomas. We describe our use of the Heald anal stent and its potential impact in reducing anastomotic or rectal stump leak after elective or emergency colorectal surgery. We performed a single-center retrospective analysis of patients in whom a Heald anal stent had been used to “protect” a colorectal anastomosis or a rectal stump, in an elective or emergency context, for benign and malignant pathology. Intraoperative and postoperative outcomes were reviewed using clinical and radiological records. The Heald anal stent was used in 93 patients over 4 years. Forty-six cases (49%) had a colorectal anastomosis, and 47 (51%) had an end stoma with a rectal stump. No anastomotic or rectal stump leaks were recorded. We recommend the Heald anal stent as a simple and affordable adjunct that may decrease anastomotic and rectal stump leak by reducing intraluminal pressure through drainage of fluid and gas.
INTRODUCTION
Surgical resection of the colon or rectum carries significant risks of postoperative complications. Anastomotic leak and the leakage or “blowout” of a rectal stump are 2 of the most feared early complications, which cause significant morbidity and occasional mortality [1, 2].
The risk of anastomotic leak is highest after low rectal surgery requiring a coloanal anastomosis, with a published incidence between 1% and 20% [2]. A leak often requires a return to the operating room, take-down of the anastomosis, and formation of an end colostomy. Rectal stump leak has a reported incidence of up to 5%, with resultant pelvic abscesses requiring drainage and extended antibiotic therapy [3, 4]. Both these complications contribute to prolonged and expensive hospital stays, decreased quality of life for the patient, and poorer oncological outcomes [5].
There are established technical factors that play a role in reducing anastomotic leak, including adequate blood supply, absence of tension, and meticulous operative technique [6]. However, the exact etiology remains unknown and is multifactorial. Recent hypotheses suggest a microbiome effect, with intraluminal microbes either inducing a proinflammatory state or producing collagenase enzymes, which are detrimental to anastomotic healing [7]. Similar factors are likely to play a role in rectal stump leak, but sphincter spasm causing back pressure of intraluminal gas and fluid may contribute.
Defunctioning stomas are commonly used to mitigate the risk, or severity, of an anastomotic leak, particularly after a low rectal or coloanal anastomosis. Stool diversion reduces effluent and gas at the anastomosis, thus reducing intraluminal pressure, and reports have suggested that defunctioning ileostomy may play a role in reducing leaks (or their sequelae) [8].
However, a defunctioning ileostomy has significant morbidity in some patients and can affect quality of life. High stoma output can lead to dehydration and electrolyte imbalance, and stoma retraction or herniation can require further surgery [9]. Reversal of a defunctioning stoma requires another operation, with potential complications. Decision-making around which patients to defunction and how (loop ileostomy vs. colostomy) continues to be debated, but generally a defunctioning stoma is considered after a low anterior resection, especially in patients who have had neoadjuvant chemo/radiotherapy [10].
Authors have described the use of transanal decompression devices instead of diverting stomas after rectal anastomoses, with a reduction in anastomotic leaks or mitigation of sequelae [11–13]. These devices range from Foley catheters to specialized devices such as the Heald anal stent. The Heald stent (Basingstoke Surgical Technology Ltd) is made of a hollow silicone elastomer, flanged at both ends, and was pioneered by Professor RJ Heald in Basingstoke, UK as a way of protecting a colorectal anastomosis [14].
In a randomized trial, the Heald stent was used specifically for low colorectal anastomoses as an alternative to a defunctioning stoma. It appeared to provide equal protection from dangerous leakage, with lower morbidity and shorter hospital stay when compared with patients who had a defunctioning stoma [15].
We have used the Heald anal stent selectively for protecting colorectal anastomoses and decompressing rectal stumps.
TECHNIQUE
Ethics statement
All patients included in the study had consented to anonymized use of their data as part of the National Health Service “opt-out scheme.”
The Heald anal stent, its indications and placement
The Heald stent (Fig. 1) is made of a soft silicone elastomer. It weighs 10 g. It is a 1-size-fit device suitable for high, mid, and low rectal or coloanal anastomoses and rectal stumps. There are no absolute contraindications to its use. It is placed under general anesthetic in the lithotomy position, after the formation of anastomosis or division of the rectum (if leaving a stump). The wider flange (50 mm diameter) is placed inside the rectum with the aid of lubricant jelly by folding it and passing it through the anal sphincters using fingers. The stent (30 mm length) keeps the anal sphincters gently open to facilitate the drainage of gas and liquid. The external flange (43 mm diameter) sits externally outside the anal sphincters to prevent it from being drawn back into the rectum. The stent can be left in place in this fashion, but the supervising author (John Bunni, consultant colorectal surgeon) chooses to fix the outer flange temporarily to the skin of the inner buttock with 1 silk suture on either side. Generally, the stent is left in situ for 3 to 4 days and flushed with 20 mL of warm water twice daily using a bladder syringe that fits snugly into the external opening of the lumen, before being easily removed on the ward (after cutting the sutures−if it has been fixed).
The study
This was a single-center retrospective study in a high-volume UK colorectal unit. We analyzed a database of colorectal resections, and selected patients who had a Heald anal stent at surgery between January 1, 2018 and November 19, 2022. Patient demographics, along with the indications and types of surgery, were recorded.
Analysis
Intraoperative and postoperative outcomes were analyzed using clinical notes, discharge summaries, and outpatient documentation. Radiological imaging or interventional procedures were cross-checked to ascertain whether an anastomotic or stump leak, or any other postoperative complication, was recorded. Standard criteria for identifying anastomotic or stump leak, such as pain, fever, raised serum inflammatory markers, clinical examination findings, and any radiological (computed tomography) records were used. Data were tabulated using Microsoft Excel (Microsoft Corp), and all statistical calculations (median, interquartile range, and percentages) were performed within this program.
Results
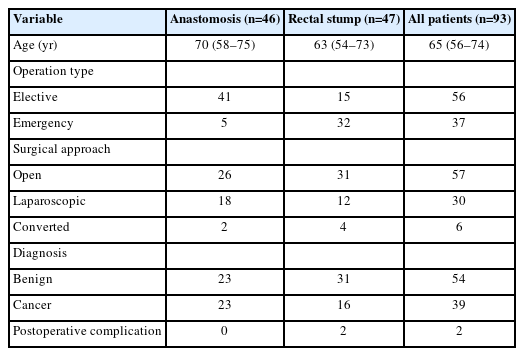
The Heald anal stent was used in 93 patients over 4 years by seven consultant surgeons, although 71 of the cases (76%) were under the care of the supervising author. The overall median age was 65 years (interquartile range, 56–74 years), with 49 men (53%) and 44 women (47%). There were 56 elective cases (60%) and 37 emergencies (40%). The underlying pathology was benign in 54 cases (58%) and malignant in 39 (42%). Overall, 57 cases (61%) had open and 30 (32%) had laparoscopic surgery, with 6 cases (7%) of laparoscopic converted to open surgery. In total, 46 patients (49%) had an anastomosis, and 47 (51%) had an end stoma with a rectal stump (Table 1). All except 1 patient had a colonic or rectal resection: this patient underwent repair of an iatrogenic full-thickness rectal injury during a complicated appendicectomy. The median number of days of the in situ stent was 4 (range, 1–5 days).
In the anastomosis group (n=46), the most common operation using a Heald anal stent was high anterior resection, with 22 cases. There were 11 reversals of Hartmann’s procedure (without defunctioning stomas), 7 sigmoid colectomies, 3 resection rectopexies, 2 reversals of Hartmann procedure (with defunctioning ileostomies), and 1 low anterior resection (as part of debulking surgery for ovarian cancer).
In the rectal stump group (n=47), Hartmann procedure was most common, with 20 cases. Eight patients had a subtotal colectomy with end ileostomy. There were 6 high anterior resections, 6 low anterior resections, 2 completion colectomies, and 2 left hemicolectomies. There was 1 unsuccessful reversal of Hartmann procedure, 1 sigmoid colectomy, and 1 suture repair of a full-thickness rectal injury.
There were no anastomotic or rectal stump leaks. Two patients with rectal stumps had pelvic collections, which were not considered due to rectal stump blowout. One was managed postoperatively with a radiological drain (after an emergency extended left hemicolectomy for colonic ischemia), and the other was treated with antibiotics only (after an elective Hartmann’s procedure for a contained perforated diverticular mass fistulating into the uterus).
DISCUSSION
The Heald anal stent was used in 92 patients undergoing colorectal resection, and 1 undergoing an emergency repair of an iatrogenic full-thickness rectal injury. No anastomotic or rectal stump leaks were detected. This is noteworthy, as some leaks might reasonably have been expected without a stent, based on previously published data (see introduction).
The soft atraumatic design of the Heald stent, designed specifically for this purpose, is a safer alternative compared with a Foley catheter (which is easily blocked by bowel content, and may risk pushing through the anastomotic line) or alternative transanal drainage tubes, and it is easy to place and secure in the operating room.
In this series, the Heald anal stent was used predominantly in higher anastomoses such as high anterior resections or reversals of Hartmann’s procedure. It was only used in one low anterior resection, as our favored method for a low rectal or coloanal anastomosis continues to be a defunctioning ileostomy (although we will consider a stent alone in a very fit and well patient). Due to the high-risk nature of low rectal surgery, and the evidence-based practice that guides it, a randomized trial comparing the Heald stent versus a defunctioning ileostomy in low rectal anastomoses is necessary prior to making a definite change to our operative practice in this subgroup.
In conclusion, the Heald anal stent offers a simple and effective way of protecting an anastomosis or a rectal stump, by reducing intraluminal pressure through facilitating drainage of gas and effluent through the anus.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
None.
Acknowledgments
The authors thank the consultant general surgeons at the Royal United Hospital (Bath, UK) who have supported use of the Heald anal stent, and Susan Thorne (senior business analyst, Royal United Hospitals Bath NHS Foundation Trust) for assistance with targeted searches of electronic patient records.
Author contributions
Conceptualization: JB, BM, RJH; Data curation: MJ, JB; Formal analysis: MJ; Methodology: MJ; Supervision: JB; Supervision: BM, RJH; Writing–original draft: MJ, BM, RJH; Writing–review & editing: all authors. All authors read and approved the final manuscript.