Prognostic Impact of Carcinoembryonic Antigen Levels in Rectal Cancer Patients Who Had Received Neoadjuvant Chemoradiotherapy
Article information
Abstract
Purpose
Carcinoembryonic antigen (CEA) is a useful marker for rectal cancer. The aim of this study was to investigate the prognostic impact of CEA level according to neoadjuvant chemoradiotherapy (nCRT) in rectal cancer patients who underwent radical surgery.
Methods
A total of 245 patients with rectal cancer who underwent radical surgery were retrospectively evaluated. Serum CEA level was measured preoperatively and postoperatively. We compared survival outcomes based on CEA level before and after surgery according to nCRT.
Results
Of the 245 patients, elevation of CEA level was observed preoperatively in 79 and postoperatively in 30, respectively. Eighty-seven (35.5%) patients received nCRT, and elevated CEA level was a significant prognostic factor both before and after surgery. In patients who had not received nCRT, an elevated CEA level was a significant prognostic factor before surgery but was not significant after surgery. In a multivariate analysis for prognostic factors, elevation of preoperative CEA level was an independent prognostic factor of disease-free survival (DFS) regardless of nCRT. Postoperative CEA level was an independent prognostic factor of DFS in patients who had received nCRT but was not a factor in patients who had not received nCRT.
Conclusion
Serum CEA level was an independent prognostic factor both preoperatively and postoperatively in rectal cancer patients who had received nCRT.
INTRODUCTION
Colorectal cancer (CRC) is the third most frequently diagnosed malignancy and one of the leading causes of cancer-related mortality worldwide [1]. Rectal cancer accounts for approximately 30% of CRC and is associated with worse clinical outcomes [2, 3]. The standard treatment of locally advanced rectal cancer is neoadjuvant chemoradiotherapy (nCRT) followed by total mesorectal excision to improve resectability, anal sphincter preservation, and local control. Selecting optimal treatments for individuals remains challenging for clinicians due to lack of effective markers [4]. In recent years, several markers have played an increasingly vital role in detection and management of CRC [5].
Carcinoembryonic antigen (CEA) is one of the most common and convenient indexes in patients with CRC. It has been used for diagnosing cancer, evaluating response to therapy, prognostic prediction, or detecting recurrence [6-9]. Although CEA is not a disease-specific marker, change from preoperative to postoperative CEA level has been suggested as an indicator of a higher recurrence rate. Many studies have investigated the role of CEA as a prognostic factor in CRC [10, 11]. In earlier reports, elevated serum CEA level has been shown to have an adverse effect on prognosis of CRC [12, 13], whereas other studies have reported no relationship between serum CEA and oncologic outcomes [14, 15]. However, a few studies have focused on rectal cancer and described the prognostic effect of CEA according to nCRT.
In this study, we investigated the prognostic impact of CEA level according to nCRT in rectal cancer patients who underwent radical surgery.
METHODS
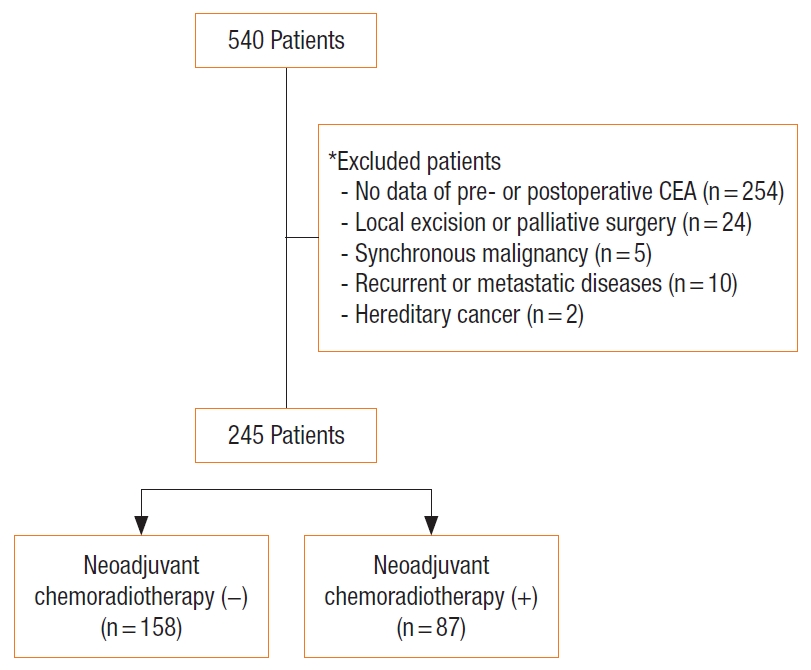
The present study is based on the colorectal database of Hallym University Sacred Heart Hospital in Anyang, Korea. A total of 245 records of patients with rectal cancer who underwent radical surgery from January 2007 and December 2017 was retrospectively reviewed. All had histologically confirmed adenocarcinoma of the rectum. Patients were excluded if they had no data for preoperative or postoperative CEA, local excision, palliative surgery, synchronous malignancies, recurrent or metastatic disease, or hereditary cancer. This study was approved by Institutional Review Board of Hallym University Sacred Heart Hospital (No. 2020-07-027). The requirement for informed consent was waived because of the retrospective nature of the study.
Patients with locally advanced rectal cancer were treated with nCRT that consisted of radiotherapy at a total dose of 50.4 Gy and concomitant chemotherapy based on either intravenous 5-fluorouracil or oral capecitabine. All had radical surgery between 6 and 10 weeks after completing nCRT. In contrast, patients with early rectal cancer immediately underwent radical surgery without nCRT. Intravenous 5-fluorouracil or oral capecitabine-based adjuvant chemotherapy was considered for either clinically or pathologically confirmed patients with locally advanced rectal cancer.
Postoperative surveillance was performed every 3 months for the first 2 years after surgery and then every 6 months for up to 5 years. Most patients were evaluated with a physical examination, serum CEA level, and chest X-ray at each visit. Abdominopelvic and chest computed tomographies were performed every 6 months. Colonoscopy was performed after the first year and then biennially.
Serum CEA level was measured preoperatively and postoperatively (within 3 months after surgery and before starting adjuvant treatments). The interval between CEA measurement and surgery were reviewed preoperatively (median, 4 days; interquartile range [IQR], 6 days) and postoperatively (median, 7 days; IQR, 2 days). The normal range of serum CEA level was defined as less than or equal to 5 ng/mL. The primary endpoint of this study was disease-free survival (DFS) based on CEA level according to nCRT.
Statistical analyses were performed using IBM SPSS Statistics for Windows (version 20.0; IBM Corp., Armonk, NY, USA). Survival rates were analyzed using the Kaplan-Meier method and the log-rank test. Multivariate analyses for prognostic factors were performed using a Cox proportional hazard model. Variables that were significant in univariate analysis were entered into the multivariate model. The P-values were derived from two-tailed tests, and P ≤ 0.05 was considered statistically significant.
RESULTS
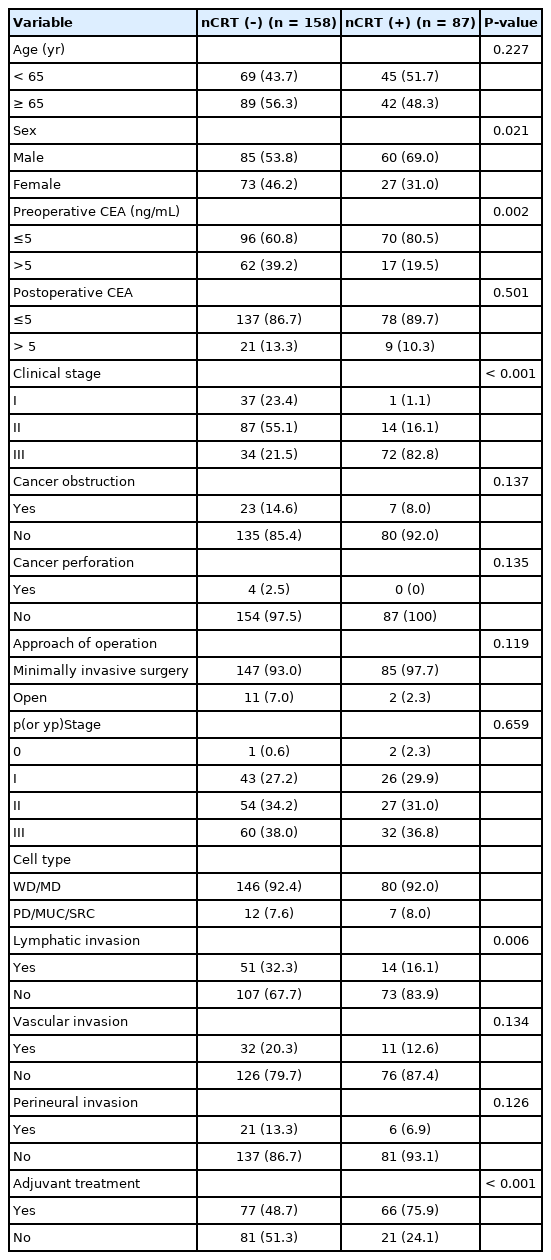
A total of 245 patients with rectal cancer was included in this study (Fig. 1). The clinicopathological features of the patients are described in Table 1. The patients included 145 males (59.2%) and 100 females (40.8%) with a median age of 66.0 years (range, 28–90 years). Of these, 87 (35.5%) received nCRT before surgery. Of the 245 patients, increased CEA level was observed preoperatively in 79 (32.2%) and postoperatively in 30 (12.2%). The median value for preoperative CEA level was 3.38 ng/mL (range, 0.01–383.0 ng/mL) and postoperative CEA was 2.0 ng/mL (range, 0.1–666.1 ng/mL).
To identify the impact of CEA on oncologic outcomes, we analyzed DFS according to CEA before and after surgery. The median follow-up duration was 47.3 months (range, 0.6–120.2 months). The 5-year DFS rates were significantly lower in patients with elevation of preoperative CEA level compared with patients with normal CEA level (62.7% vs. 84.9%, P < 0.001) (Fig. 2A). However, there was no significant difference in 5-year DFS rates according to postoperative CEA levels (75.1% vs. 78.6%, P = 0.424) (Fig. 2B).

Disease-free survival (DFS) according to serum carcinoembryonic antigen (CEA) levels (A) before surgery and (B) after surgery.
To evaluate the prognostic value of CEA according to nCRT, we analyzed DFS according to CEA before and after surgery in each group. In patients who had not received nCRT, elevated CEA level was a significant prognostic factor before surgery but was not significant after surgery (P = 0.013 and P = 0.710, respectively) (Fig. 3A). In patients who had received nCRT, elevated CEA level was a significant prognostic factor both before and after surgery (P = 0.002 and P = 0.024, respectively) (Fig. 3B). We performed additional analysis for DFS in each stage and confirmed that the results were similar to those of all patients (Supplementary Figs. 1–3).
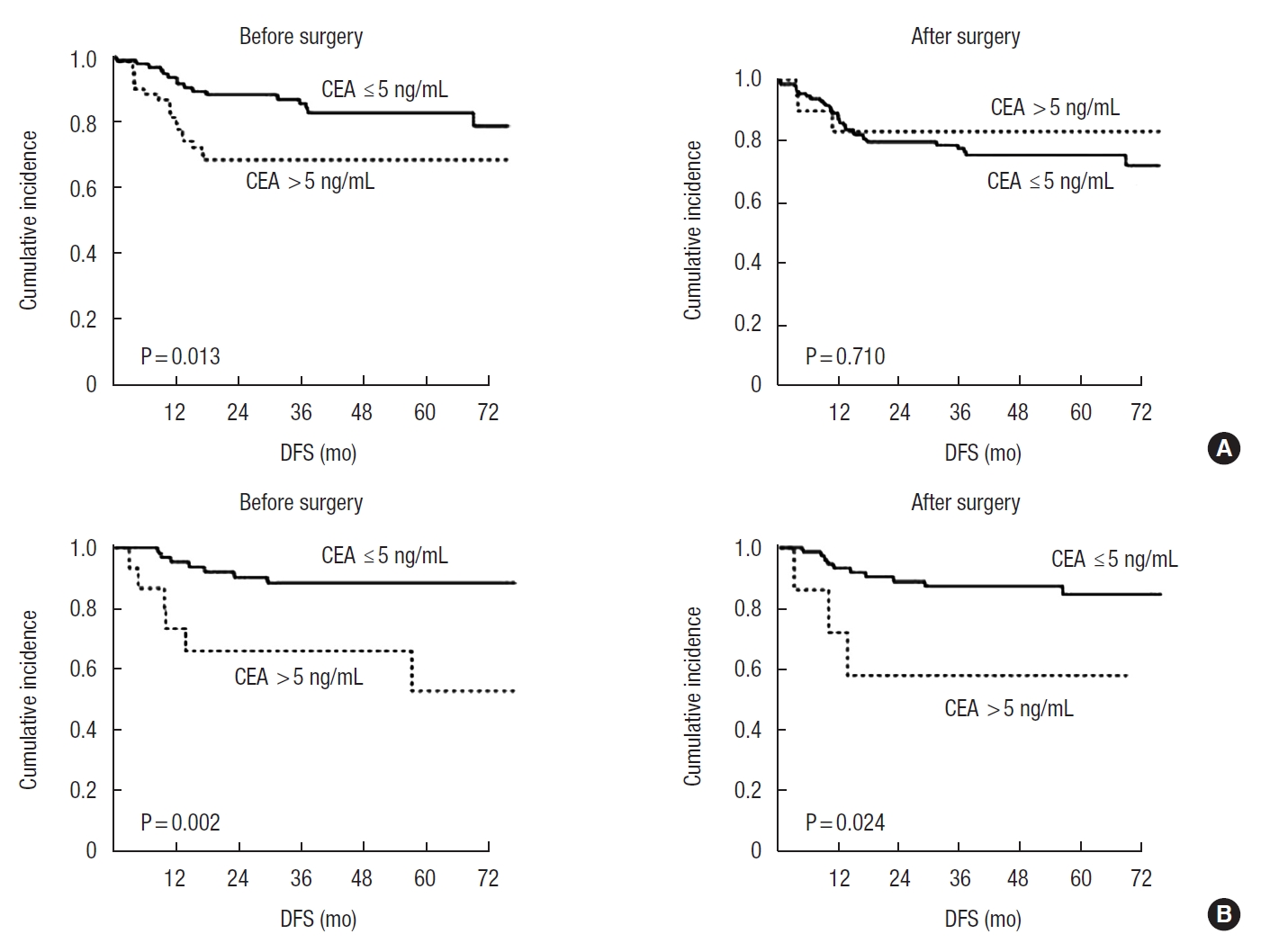
Disease-free survival (DFS) according to serum carcinoembryonic antigen (CEA) levels before and after surgery in patients who had (A) not received neoadjuvant chemoradiotherapy (nCRT) and (B) received nCRT.
In addition, we further analyzed DFS according to change in CEA level before and after surgery in patients who had received nCRT. The patients were divided into 3 groups according to the CEA levels as follows: group 1 (preoperative, ≤ 5 ng/mL and postoperative, ≤ 5 ng/mL); group 2 (preoperative, > 5 ng/mL and postoperative, ≤ 5 ng/mL); and group 3 (preoperative, > 5 ng/mL and postoperative, > 5 ng/mL). As shown in Fig. 4, the DFS was significantly worse in group 3 than in group 1 and group 2 (P = 0.008).

Disease-free survival (DFS) according to changes in carcinoembryonic antigen (CEA) levels in patients who had received neoadjuvant chemoradiotherapy. CEA level (ng/mL): group 1, preoperative ≤5 and postoperative ≤5; group 2, preoperative >5 and postoperative ≤5; and group 3, preoperative >5 and postoperative >5.
Cox proportional hazard modeling was performed to identify whether CEA levels before and after surgery, according to nCRT, were an independent prognostic factor for survival outcomes in rectal cancer. In patients who had not received nCRT, elevation of preoperative CEA level was an independent prognostic factor of DFS (P = 0.049; hazard ratio [HR], 2.017; 95% confidence interval [CI], 1.002–4.058), but postoperative CEA level was not. In patients who had received nCRT, elevation of both preoperative and postoperative CEA level was an independent prognostic factor of DFS (P = 0.017; HR, 5.443; 95% CI, 1.356–21.848; and P = 0.036; HR, 3.983; 95% CI, 1.093–14.515, respectively) (Table 2).
DISCUSSION
In this study, we investigated the prognostic value of CEA levels according to nCRT in rectal cancer patients who underwent radical surgery. We observed that preoperative CEA level was a prognostic factor regardless of nCRT, and postoperative CEA level was a significant prognostic factor only in patients receiving nCRT. In patients who had received nCRT, patients with continuing CEA level elevation after surgery showed significantly worse DFS compared to patients with lower CEA level postoperatively.
Many oncologic researchers have investigated the role of serum CEA as a prognostic indicator in CRC [13-18]. In locally advanced rectal cancer, the standard treatment is nCRT followed by radical surgery, and the relationship of CEA and response to nCRT has been studied [6, 7]. Some studies have reported that patients with elevated serum CEA levels before and after nCRT or surgery showed poor oncologic outcomes [19-22], whereas other studies have reported no relationship between serum CEA and oncologic outcomes [23, 24]. Thus, the association between serum CEA level and nCRT in rectal cancer is controversial.
CEA is a glycoprotein expressed on colonic epithelial cells and secreted into the bloodstream, leading to an increase in serum level [25]. CEA is reliable for detecting recurrence and is used to evaluate the biological activity of malignancies. Thus, it is recommended by the American Society of Clinical Oncology and the European Society for Medical Oncology as a prognostic biomarker during routine follow-up for CRC after surgical resection [5, 11]. In this study, CEA was an independent prognostic factor both preoperatively and postoperatively in patients who had received nCRT. Patients with persistent postoperative CEA level elevation showed the worst DFS. There may be tumor cells that have escaped systemic control while local control of tumor cells by nCRT. CEA reflects the burden of these circulating tumor cells and has been used as a marker for detecting micrometastases [26-30]. Our findings may be due to persistence of micrometastases during nCRT and continued production of CEA as a result. On the other hand, most of patients in non-nCRT group are clinically early staged rectal cancer compared to those in nCRT group. In non-nCRT group, the elevated CEA level after surgery is may be due to false positive. This finding has also been revealed in several existing literature [30-33]. Our results may be useful in predicting recurrence and establishing treatment strategies in rectal cancer patients who received nCRT, especially in patients with continuing CEA level elevation after surgery.
There were some limitations in our study. This study was performed retrospectively in a single institution. There was a lack of consideration of other factors contributing to false-positive CEA elevation. We have no data on CEA level prior to surgery after nCRT. Also, we did not analyze the association between CEA level and tumor response to nCRT.
In conclusion, serum CEA level was an independent prognostic factor both preoperatively and postoperatively in rectal cancer patients who had received nCRT. Close observation and intensive treatment are needed in patients with persistent CEA level elevation after surgery.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
SUPPLEMENTARY MATERIALS
Supplementary materials for this study are presented online (available at https://doi.org/10.3393/ac.2020.11.27).
Supplementary Fig. 1. Disease-free survival (DFS) according to serum carcinoembryonic antigen (CEA) levels in each stage (A) before surgery and (B) after surgery
Supplementary Fig. 2. Disease-free survival (DFS) according to serum carcinoembryonic antigen (CEA) levels in each stage of patients who had not received neoadjuvant chemoradiotherapy (A) before surgery and (B) after surgery.
Supplementary Fig. 3. Disease-free survival (DFS) according to serum carcinoembryonic antigen (CEA) levels in each stage of patients who had received neoadjuvant chemoradiotherapy (A) before surgery and (B) after surgery