Lifestyle Factors and Bowel Preparation for Screening Colonoscopy
Article information
Abstract
Purpose
The quality of bowel preparation is a major determinant of the quality of colonoscopy. This study evaluated lifestyle factors, including usual dietary style, associated with bowel preparation.
Methods
This retrospective study evaluated 1,079 consecutive subjects who underwent complete colonoscopy from December 2012 to April 2014 at National Cancer Center of Korea. Questionnaires on bowel preparation were completed by the subjects, with the quality of bowel preparation categorized as optimal (excellent or good) or suboptimal (fair, poor or inadequate). Lifestyle factors associated with bowel preparation were analyzed.
Results
The 1,079 subjects included 680 male (63.0%) and 399 female patietns (37.0%), with a mean age of 49.6 ± 8.32 years. Bowel preparation was categorized as optimal in 657 subjects (60.9%) and as suboptimal in 422 (39.1%). Univariate analyses showed no differences between groups in lifestyle factors, such as regular exercise, alcohol intake, smoking, and dietary factor. Body mass index (BMI) > 25 kg/m2 was the only factor associated with suboptimal bowel preparation on both the univariate (P = 0.007) and the multivariate (odds ratio, 1.437; 95% confidence interval, 1.104–1.871; P = 0.007) analyses.
Conclusion
Most lifestyle factors, including dietary patterns, exercise, alcohol intake and smoking, were not associated with suboptimal bowel preparation in Koreans. However, BMI > 25 kg/m2 was independently associated with suboptimal bowel preparation. More intense preparation regimens before colonoscopy can be helpful in subjects with BMI > 25 kg/m2.
INTRODUCTION
Colorectal cancer (CRC) is the third most common cancer and the fourth leading cause of cancer-related deaths in Korea [1]. Colonoscopy and the associated removal of precancerous lesions have been shown to reduce CRC-associated mortality rates [2]. Despite its invasiveness, colonoscopy is considered the standard diagnostic method for CRC and other colorectal diseases, as well as for the removal of precancerous polyps. The efficacy of colonoscopy depends on several factors. The presence of adenomatous polyps affects the risk of CRC after screening colonoscopy [3]. Detection of adenomas, however, depends on the ability to visualize the colonic mucosa, emphasizing the importance of bowel preparation [4-6]. However, bowel preparation at the time of colonoscopy is found to be suboptimal in about 20% of subjects [5].
Suboptimal bowel preparation has been associated with longer intubation time, decreased cecal intubation rate, a higher rate of missed lesions, increased patient discomfort and a higher risk of complications [7]. Factors associated with suboptimal bowel preparation have been found to include in inpatient status, constipation, use of tricyclic antidepressants, later colonoscopy starting time, male sex, and low socioeconomic status [8]. Other factors associated with suboptimal bowel preparation include increased body mass index (BMI) [9] and a low residue diet [10, 11]. The normal Korean diet includes high amounts of fiber and sodium. However, dietary patterns in Korea have become more westernized with industrialization. This study was designed to evaluate the relationship between lifestyle factors, including dietary patterns, and quality of bowel preparation for screening colonoscopy in Korean subjects.
METHODS
Subjects and data collection
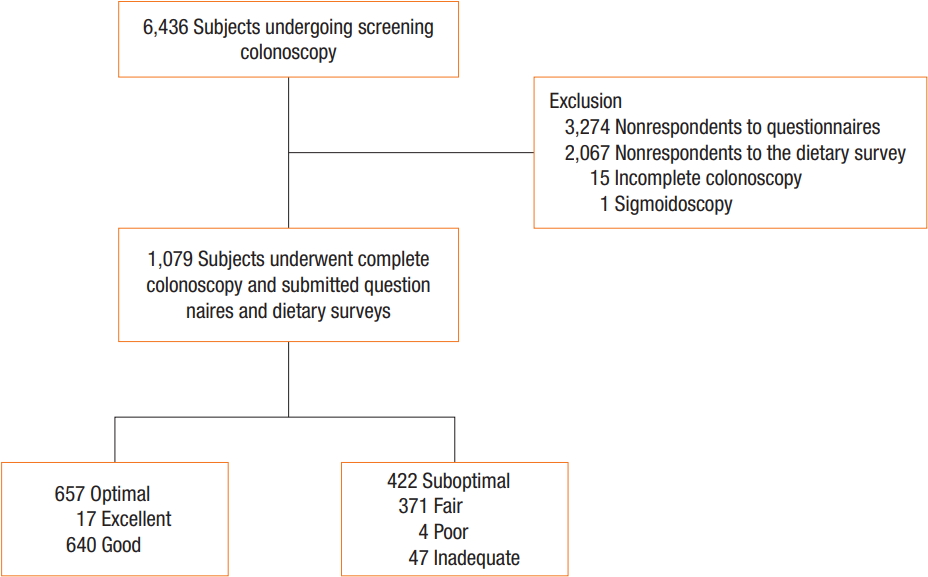
Individuals enrolled in this study underwent self-motivated comprehensive cancer screening, including colonoscopy at the Center for Cancer Prevention and Detection of the National Cancer Center (NCC, Goyang, Korea) between December 2012 and April 2014. This study was approved by the Institutional Review Board of the NCC (NCC2014-0218). All participating subjects provided signed informed consent before enrollment. Of the 6,436 consecutive subjects who had undergone screening colonoscopy at our institution, 5,344 did not respond to the questionnaires and dietary surveys. Fifteen subjects could not undergo complete colonoscopy, and one underwent sigmoidoscopy with Yal enema (Fig. 1).
Subjects who had undergone screening colonoscopy were prospectively given questionnaires on environmental, lifestyle and host factors associated with the risk of cancer. Questionnaires were mailed to the subjects one week before colonoscopy. Questionnaires included a 3-day food record (1 weekend day and 2-week-days) and questions on family history, surgical or medical history, lifestyle, and education level. The questionnaires were returned to the hospital at the time of colonoscopy. During the scheduled visit, subjects’ food records were reviewed by trained nutritionists who used aids such as food models to improve reporting accuracy. Daily energy and macro- and micronutrient intakes were calculated using the Korean Nutrition Society’s nutrient database (Can-Pro 3.0, The Korean Nutrition Society, Seoul, Korea) [12]. Nutrient density was calculated by standardizing nutrient intake per 1,000 kcal based on everyone’s intake. Personal data (age, sex, alcohol consumption, smoking status, personal medical history, and medication history) were collected prior to colonoscopy. Smoking status was classified as current smoker (daily or occasionally), former smoker (quit at least 1 year before), or nonsmoker (lifetime abstainer). Alcohol consumption status was similarly classified. Weight and height measurements were automated (InBody, Biospace Co., Ltd, Seoul, Korea). BMI was calculated as weight in kg divided by height in m2 and categorized as normal or abnormal, as described previously and according to World Health Organization expert consultation [13].
Before colonoscopy, all subjects were advised on proper methods of bowel preparation, including (1) a low fiber diet for the 72 hours before colonoscopy, (2) a liquid diet on the day before colonoscopy, (3) 2 tablets of 5-mg bisacodyl at 7 PM on the day before colonoscopy, (4) 2 L of polyethylene glycol plus ascorbic acid, one liter at 6 and the other at 9 PM on the day before colonoscopy. All subjects underwent colonoscopy during the morning using video colonoscopes (Olympus CF-Q240, CF-Q260 or CF-H260, Olympus, Tokyo, Japan).
The adequacy of bowel preparation was assessed by the endoscopists on withdrawal of the colonoscope, by using the Aronchick score as excellent, good, fair or poor [14]. Colonoscopy findings included the presence of polyps, their numbers and sizes, Paris classification and methods of polypectomy. Subjects were categorized into 2 group, those with optimal (excellent or good based on the Aronchick score) and suboptimal (fair, poor, or inadequate) bowel preparation.
Statistical analyses
The study was designed to assess the association between lifestyle factors and bowel preparation. The characteristics of subjects with optimal and suboptimal bowel preparation were compared using Student t-tests for continuous variables and chi-square tests for categorical variables. Daily nutrition and food intake were categorized by quartiles. Regression models were used to assess associations of daily nutrition with food intake and bowel preparation quality. Odds ratios (ORs) and 95% confidence intervals (CI) were estimated using logistic regression, both in the crude model and in the multivariate model. The multivariate model was adjusted for age, sex, smoking status, and alcohol consumption. Tests for trends were derived from logistic regression with a single term representing the medians of each quartile. A 2-sided P-value less than 0.05 was regarded as statistically significant. All statistical analyses were performed using SAS 9.3 (SAS Institute Inc., Cary, NC, USA) and SPSS ver. 14.0 (SPSS Inc., Chicago, IL, USA).
RESULTS
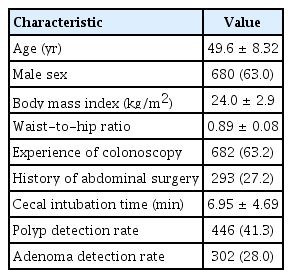
A total of 1,079 subjects underwent complete colonoscopy and completed questionnaires and dietary surveys. These 1,079 subjects included 680 male (63.0%) and 399 female patients (37.0%), with a mean age 49.6 ± 8.3 years. Bowel preparation was categorized as optimal for 657 subjects (60.9%) and as suboptimal for 422 subjects (39.1%). Preparation quality was characterized as excellent for 17 (1.6%), good for 640 (59.3%), fair for 371 (34.4%), poor for four (0.4%) and inadequate for 47 subjects (4.3%) (Fig. 1). The 1,079 included subjects had a mean BMI of 24.1 ± 2.9 and a mean waist-to-hip ratio of 0.89 ± 0.08. All subjects underwent colonoscopy for surveillance; of these, 682 (63.2%) had previously undergone surveillance colonoscopy, and 293 (27.2%) had a history of abdominal surgery (Table 1). Polyps were detected in 41.4% of the subjects and adenomatous polyps in 28.0%.
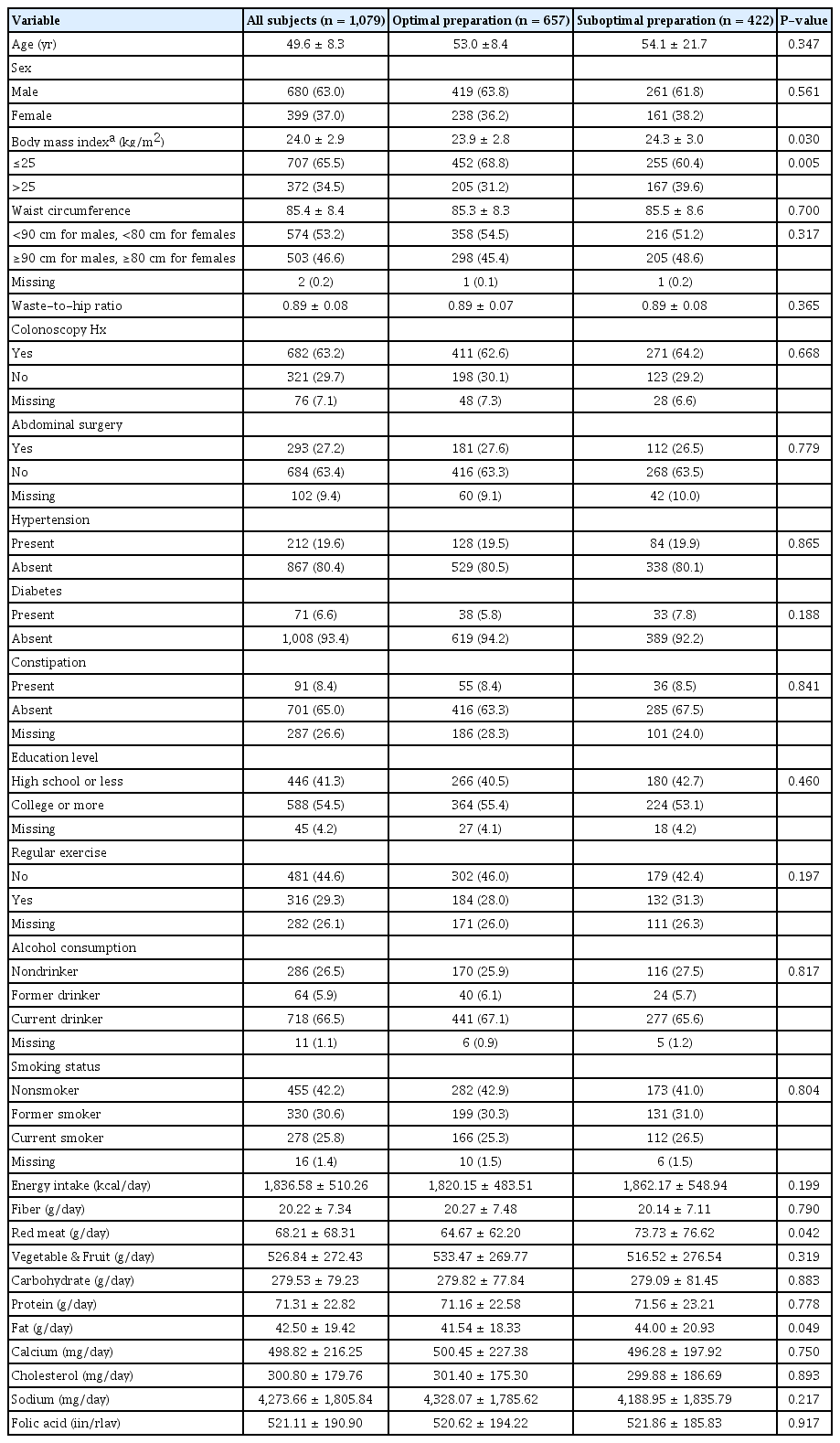
Table 2 shows the characteristics of the optimal and the suboptimal bowel preparation groups. The percentage of subjects with BMI > 25 kg/m2 was significantly higher in subjects with suboptimal bowel preparation than in subjects with optimal bowel preparation (P = 0.005). In contrast, the mean waist-to-hip ratios were similar (0.89 ± 0.07 vs. 0.89 ± 0.08, P = 0.365). Other variables, such as history of abdominal surgery, comorbidities (e.g., hypertension, diabetes, constipation), and social habits (e.g., alcohol use, smoking) did not differ significantly between these two groups. Mean red meat intake was significantly higher in subjects with suboptimal bowel preparation than in those with optimal bowel preparation (73.73 ± 76.62 g/day vs. 64.67 ± 62.20 g/day, P = 0.042). Other dietary factors did not differ significantly between these 2 groups.
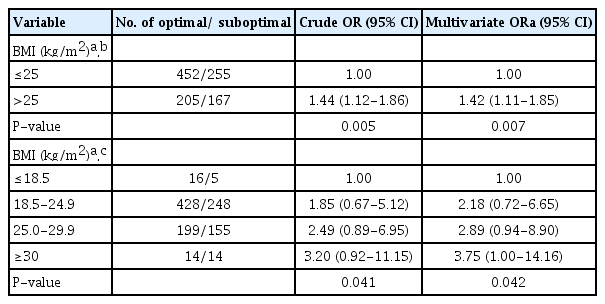
Table 3 shows the association between BMI and suboptimal bowel preparation. Subjects with higher BMI were at significantly higher risk for suboptimal bowel preparation. BMI > 30 kg/m2 was significantly associated with suboptimal bowel preparation on both the univariate (OR, 3.20; 95% CI, 0.92–11.15; P = 0.041) and the multivariate (OR, 3.75; 95% CI, 1.00–14.16; P = 0.042) logistic regression analyses. In contrast, no factor associated with the level of nutrition of food intake was significantly associated with adequacy of bowel preparation on either the univariate or the multivariate logistic regression analyses (Table 4).
DISCUSSION
This study found that obesity was an independent risk factor for suboptimal bowel preparation quality in subjects undergoing surveillance colonoscopy. Although dietary factors associated with bowel preparation were also analyzed because we expected suboptimal bowel preparation to be associated with ingestion of a higher amount of dietary fiber, we found no association between dietary patterns and the adequacy of bowel preparation.
In Korea, traditional diets contain higher amounts of fiber and rice and lower amounts of red meat than Western diets. Industrialization, however, has caused Korean diets to become more westernized. Nevertheless, vegetable intake remains higher in Korean than in Western diets. We hypothesized that dietary patterns and lifestyle factors would have an impact on bowel preparation. However, we found that dietary patterns and lifestyle factors were not associated with the quality of bowel preparation.
Factors shown to be important in the quality of bowel preparation include dietary restrictions and timing of colonoscopy [15-17]. For example, a low residue diet for 2 days before colonoscopy was found to improve the quality of bowel preparation [18], suggesting that a low residue diet before colonoscopy is essential for optimizing the quality of bowel preparation. However, a clear liquid diet was not mandatory for bowel preparation [19, 20].
The dietary survey in this study included questions about usual dietary patterns, not those 2 or 3 days before colonoscopy. We found no association between subjects’ usual dietary patterns and the quality of bowel preparation. However, dietary patterns for several days immediately before colonoscopy were found to be important for optimizing the quality of bowel preparation.
Our results are consistent with those of previous studies, showing that obesity was a risk factor for suboptimal preparation quality. For example, 2 studies reported that BMI ≥ 30 kg/m2 was an independent predictors of inadequate bowel preparation at colonoscopy (OR, 1.35; 95% CI, 1.09–1.68; P = 0.006) [9, 21]. However, the reason for the association between obesity and suboptimal bowel preparation is not clear. Adequate bowel preparation has been found to be particularly important in obese subjects due to their increased incidence of colonic adenomas [22, 23]. Thus, suboptimal preparation in obese subjects may result in a higher rate of missed adenomas than in subjects whose BMI ≤ 25 kg/m2. A more intensive preparation regimen may therefore benefit subjects with higher BMI. Preparation quality in obese subjects may be improved by a higher dose of current preparation regimens, the addition of another cathartic, a longer period of dietary restriction (low-residue diet and clear-liquid diet), and education to optimize understanding and compliance.
Inadequate bowel preparation has also been associated with delayed colonoscopy starting time; failure to follow preparation instructions; inpatient status; procedural indications of constipation, use of tricyclic antidepressants, male sex, a history of cirrhosis, stroke, or dementia, and a history of abdominal surgery, such as, an appendectomy, colorectal resection, or hysterectomy [24, 25]. This study found that other factors tested, including a history of abdominal surgery, older age, history of underlying disease, and usual dietary pattern, were not associated with quality of bowel preparation.
Subjects in this study population underwent colonoscopy as part of their personal health checkups and paid its costs personally, suggesting that these subjects were more interested in healthcare and more likely to pay attention to bowel preparation than other subjects. Surprisingly, however, the overall rate of suboptimal bowel preparation was higher in this study than in previous studies. Recent results, and the guideline of the European Society of Gastrointestinal Endoscopy recommend a split-dose regimen for bowel preparation, with time between the last dose of preparation and colonoscopy being no longer than four hours [26]. This protocol was difficult to apply to our study population, as these subjects underwent colonoscopy early in the morning. Moreover, many individuals underwent colonoscopy more than four hours after bowel preparation because they also underwent abdominal ultrasound and/or esophagogastroduodenoscopy on the same day. This is very common in Korea and Japan, where the incidence rates of stomach and liver cancer are higher than in Western countries.
Our study had several limitations, including its retrospective design. However, the data were collected prospectively. Another limitation was its performance at a single institution, the Center for Cancer Prevention and Detection of the National Cancer Center in Korea, which may limit generalization of its results to other populations. Moreover, the determination of preparation quality can vary significantly among endoscopists. The average scores of bowel preparation quality were assessed in each segment of the colon by using the Aronchick scale and were not routinely documented based on evaluation before and after lavage. Thus, the assessment of bowel preparation quality likely showed high interobserver variability.
In conclusion, this study found that lifestyle factors, especially dietary patterns, were not associated with the quality of bowel preparation whereas obesity was associated with suboptimal bowel preparation. A more intensive preparation regimen and more intensive dietary restrictions before colonoscopy may result in better bowel preparation in subjects with a BMI ≥ 25 kg/m2.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This work was supported by a National Cancer Center Grant (NCC-1410250 & NCC-1510150).