Spotlight on laparoscopy in the surgical resection of locally advanced rectal cancer: multicenter propensity score match study
Article information
Abstract
Purpose
This study was aimed to assess the feasibility of laparoscopic rectal surgery, comparing quality of surgical specimen, morbidity, and mortality.
Methods
Prospectively acquired data from consecutive patients undergoing laparoscopic surgery for rectal cancer, at 2 minimally invasive colorectal units, operated by the same team was included. Locally advanced rectal tumors were identified as T3B or T4 with preoperative magnetic resonance imaging scans. All the patients were operated on by the same team. The 1:1 propensity score matching was performed to create a perfect match in terms of tumor height.
Results
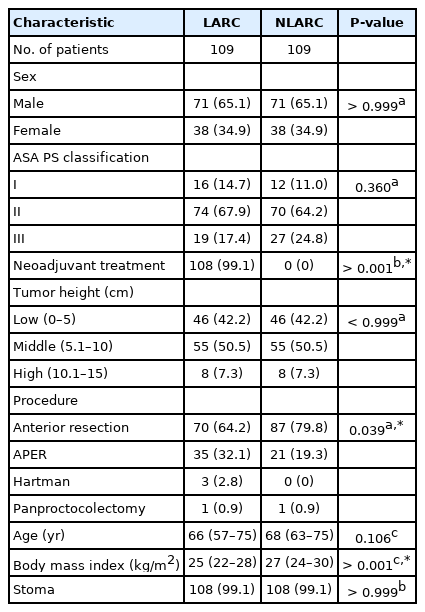
Total of 418 laparoscopic resections were performed, out of which 109 patients had locally advanced rectal cancer (LARC) and were propensity score matched with non-LARC (NLARC) patients. Median operation time was higher for the LARC group (270 minutes vs. 250 minutes, P=0.011). However, conversion to open surgery was done in 5 vs. 2 patients (P=0.445), reoperation in 8 vs. 7 (P=0.789), clinical anastomotic leak was found in 3 vs. 2 (P=0.670), and 30-day mortality rates was 2 vs. 1 (P>0.999) between LARC and NLARC, respectively. Readmission rate was higher in the NLARC group (33 patients vs. 19 patients, P=0.026), due to stoma-related issues. There was no statistically significant difference in the R0 resection between the 2 groups (99 patients in LARC vs. 104 patients in NLARC, P=0.284).
Conclusion
This study demonstrates that standardized approach to laparoscopy is safe and feasible in LARC. Comparable postoperative short-term clinical and pathological outcomes were seen between LARC and NLARC groups.
Graphical Abstract
INTRODUCTION
Rectal cancer incidence has increased worldwide and the treatment for rectal cancer, especially low rectal cancer has seen a paradigm shift in the last couple of decades from open to laparoscopic, and lately to robotic surgery [1, 2]. In locally advanced rectal cancer (LARC), straightforward surgical resection or multimodality treatment like the rectal resection after neoadjuvant chemoradiotherapy (CRT) is still the “standard of care” [3]. These decisions are now dependent on the staging pelvic magnetic resonance imaging (MRI) and dedicated multidisciplinary team (MDT) recommendations. In the last decade, image-guided surgery and MRI-guided decision making have played a key role to decrease local-recurrence rates to historical levels [4, 5].
Minimally invasive approach to rectal cancer has been put into question, as 2 recent randomized controlled trials failed to show noninferiority of laparoscopic surgery compared to open resections in terms of pathological outcomes [6, 7]. Although at long-term follow-up, these findings were not confirmed and the locoregional and distant recurrence rates proved to be similar, a discussion on safety of performing laparoscopy for rectal cancer has started again [8].
As this debate resurfaced, the oncologic safety of laparoscopy, especially for LARCs, is still controversial. Despite the increased patient-related benefits of the minimally invasive approach, most of the data in literature come from single institutions or is not focused on LARC patients.
We present this multicenter propensity score match study to assess the feasibility of laparoscopic surgery in LARC patients, comparing the short-term surgical and clinical outcomes to those with non-LARC (NLARC).
To our knowledge, it is the only study to compare the feasibility of laparoscopic surgery in LARC patients.
METHODS
All the consecutive patients who were subjected to potentially curative elective laparoscopic resections for rectal carcinoma from December 2006 to December 2013, at National Health Services Hospital Portsmouth, United Kingdom and from Champalimaud Foundation (CF), Lisbon, Portugal between 2013 and 2018 were identified from a prospectively maintained database. All the patients who presented to these institutes with the histological diagnosis of rectal carcinoma, confirmed by MRI, and operated laparoscopically through a standardized approach as explained by Bullock et al. [9] irrespective of their T stage, were included in the study. Exclusion criteria included all other tumors, open and robotically performed rectal cancer surgery and synchronous rectal tumors.
All included patients signed an informed consent allowing their data to be used for retrospective analysis and research. The requirements for anonymization of personal dataset by the Data Protection Act 1998 were satisfied. According to the Health Research Authority, this study did not require their approval due to its status as a clinical audit.
Perioperative care
Both surgeons had vast experience in laparoscopic colorectal surgery. For oncological staging colonoscopy, computed tomography chest and abdomen was used along with MRI pelvis to stage rectal tumor. All the patients were discussed in MDT meetings before the surgical plan was formulated for each patient considering the current standards. The presence of carcinoma within 15 cm from the anal verge and confirmed by staging MRI was defined as rectal carcinoma. LARC was defined as all the T3 tumor with threatened or involved circumferential resection margins (CRM), positive extramural venous invasion and T4 tumors. These patients were subjected to CRT due to their high risk of local recurrence. NLARC were all the rest of T1 to T3 tumors and who were offered surgery first. As described previously, total mesorectal excision was performed in a standardized way through a minimally invasive laparoscopic approach. All the patients were managed postoperatively through an enhanced recovery program as described by Kehlet and Wilmore [10]. The patients were sent home as per the criteria set for discharge.
Patient selection
All the patients who received CRT and surgery were allocated to LARC group, as these were the patients who were at high risk of local recurrence. Surgery was performed at 8 to 12 weeks following completion of their treatment. The NLARC group consisted of all the rest of the patients who were not fulfilling the criteria of LARC, and included T1 to T3 tumors with low risk of local recurrence, and were propensity score matched as 1:1, to the patients of LARC group. The procedure and timing of surgery was a shared decision, keeping patient preferences and MDT recommendations in mind.
Outcome assessment
Outcomes were assessed for both the groups in the perioperative period and were analyzed. Data were analyzed retrospectively from a prospectively maintained database. Baseline characteristics included sex, age, body mass index (BMI), American Society of Anesthesiologists (ASA) physical status (PS) grade, and T stage; and LARC patients were closely matched for tumor height. Perioperative data analyzed included operation time, type of procedure performed, and conversion to open from laparoscopic surgery (defined as any incision needed to either mobilize the rectum or ligate the vessels). Postoperative clinical and pathological data examined included length of stay, anastomotic leak, lymph node yield, 30-day postoperative complications (Clavien-Dindo classification), 30-day readmission and reoperation, 30-day mortality, and microscopic clear resection margins (R0).
Statistical analysis
Once collected and checked, data were analyzed using IBM SPSS Statistics ver. 24 (IBM Corp., Armonk, NY, USA). The LARC cases were propensity score matched to NLARC cases. Tumor height (low vs. middle vs. high) was the only variable used to calculate the propensity score matching. This is because tumor height greatly differed between the 2 groups before propensity score matching and when other variables, such as BMI or procedure performed, were included in the matching variables it was not possible to create a good match between the 2 groups in terms of tumor height. Propensity scores were calculated via logistic regression analysis by applying the propensity score matching function on IBM SPSS Statistics ver. 24 with the match tolerance set to 0.1.
Nonparametric data was expressed as median with interquartile range and parametric data as mean with standard deviation. Cohort demographic and clinical characteristics were compared using the chi-square test or Fisher exact test for categorical variables, Mann-Whitney U-test for nonparametric continuous variables, and t-test for parametric continuous variables. P-values of < 0.05 were considered statistically significant.
Univariate binary logistic regression analysis was performed on all patients, receiving laparoscopic rectal cancer resections (n=418) to assess, whether tumor advancement (LARC vs. NLARC) affected readmission. Following this, a multivariate model was applied where tumor advancement was adjusted for all clinically relevant variables including age, sex, BMI, ASA PS grade, and tumor height. The constant was included in the analysis model and data are presented as odds ratio, 95% confidence interval, and P-value.
RESULTS
A total of 418 patients received laparoscopic rectal cancer resections (109 in LARC, 309 in NLARC). The 109 LARC cases were propensity score matched (1:1) with 109 NLARC cases to reduce the effect of tumor height, as a significant confounding factor in the analysis.
Cohort characteristics
Propensity score matching created a perfect match in terms of tumor height. There were more abdominoperineal resections in the LARC group (32.1% vs. 19.3%, P= 0.039) and more anterior resections in the NLARC group (64.2% vs. 79.8%, P= 0.039). Median BMI was also higher by 2 kg/m2 in the NLARC group (27 kg/m2 vs. 25 kg/m2 for LARC). As discussed above, only patients in the LARC group received neoadjuvant radiotherapy except for 1 patient. There were no other differences in the remaining cohort characteristics examined (sex, ASA PS grade, age, and stoma). Table 1 summarizes the cohort characteristics of the 2 groups.
Clinical and pathological short-term outcomes
Readmission rate was higher in the NLARC group (17.4% vs. 30.3%, P= 0.026) and median operation time was longer in the LARC group (270 minutes vs. 250 minutes, P= 0.011). There was no difference in any of the remaining short-term outcomes examined, including overall complications and complications graded as III to V on the Clavien-Dindo classification. Table 2 summarizes the short-term outcomes of the 2 cohorts.
Logistic regression analysis
Univariate logistic regression analysis of all 418 cases showed that readmission rate was not affected by stage of the tumor (LARC vs. NLARC). This was still the case in multivariate analysis when other clinically relevant factors were adjusted for (BMI, ASA PS grade, sex, age, and tumor height). Table 3 summarizes the regression analysis results for readmission.
DISCUSSION
Laparoscopic surgery is now considered as one of the valid options for the operative management of rectal cancer and is reconfirmed by one of the studies long-term results, showing no difference in local recurrence and disease-free survival [8]. Our study was demonstrated and confirmed the safety and feasibility of laparoscopic approach for LARC. Propensity score matching (1:1) was performed for tumor height between the 2 groups. As a protocol all those patients with threatened or involved CRM were subjected to CRT and others to upfront surgery. There were more patients who received the abdominoperineal excision of the rectum (APER) in LARC (32.0%) when compared to NLARC (19.3%). It is largely due to the fact that more patients who underwent APER in the LARC group had sphincter involvement before neoadjuvant CRT and were planned for this procedure following down-staging treatment. Conversion to open surgery, anastomotic leak rate, R0 resection, reoperation, morbidity, and mortality were nonsignificant between the groups. An operation time of 20 minutes more was found in the LARC; this is due to the technical challenge that LARC would present and larger numbers undergoing APER procedure in this group. A statistically significant readmission rate (17.4% vs. 30.3%) was seen in the NLARC group. However, the complication rates and reoperation rates were similar in both groups. Most of the patients requiring readmission were due to protective loop ileostomy problems requiring nonoperative management following their readmission.
Although in most studies laparoscopic surgery is compared with open surgery, laparoscopy is now becoming standardized approach with more patients and surgeons opting for laparoscopic surgery [3, 11, 12]. It’s a unique study, as no one has ever compared the utility of laparoscopic surgery in the case of LARC, considering most of the surgeons will either operate them through open technique or mark them inoperable. At the same time, certain studies including the COREAN trial [13] have clearly demonstrated not only the safety and feasibility of laparoscopic approach in LARC but also shown that long-term survival and local recurrence is comparable to open group. In another study by Miyajima et al. [14] reported more than 1,000 laparoscopically operated cases and showed its feasibility for selected cases of rectal cancer patients. Our study aims to address the very issue of the laparoscopic approach, for rectal cancer with direct comparison using propensity score, for LARC treated by laparoscopy and showed similar postoperative complications rate in both the groups including the anastomotic leak rates, 30-day morbidity, and mortality.
In another study by Nasir et al. [15] showed an overall CRM negativity of 93% in their regrowth patients following complete clinical response, postneoadjuvant CRT for LARC patients in their watch and wait cohort of patients, defining the role of minimally invasive surgery. In the American College of Surgeons Oncology Group (ACOSOG) Z6051 trial, CRM negative rates were found in 88% for laparoscopic resections and 92% for open resections with overall CRM negativity in 90% of the patients [7]. Similarly, ALaCart trial showed slightly higher CRM negative rates of 93% and 97% for laparoscopic and open arms respectively, and failed to prove the noninferiority of laparoscopic surgery [6]. These trials mainly focused on the comparison of pathological specimens between laparoscopic and open resections and also excluded T4 tumors from their study population. However, as far as our study is concerned, the rate of R0 resection was found in 91% of LARC and 95% in NLARC patients, all operated laparoscopically, which is similar to the results shown by these 2 noninferiority trails. Whereas, the COLOR II trial for low rectal cancers, excluding T3 with threatened CRM and T4 tumors, showed CRM was involved in 9% of the laparoscopic arm, similar to our results of LARC patients, and 22% in open arm with higher 3 years locoregional recurrence rates in open arm [16].
A United Kingdom-based group, Panteleimonitis et al. [17] showed the feasibility of laparoscopic surgery in high-risk rectal cancer patients (BMI ≥ 30 kg/m2, neoadjuvant CRT, tumor < 8 cm from the anal verge, and previous abdominal surgery), and presented similar short- and long-term outcomes between high-risk and low-risk patients. The only variables of difference were higher blood loss and length of stay in high-risk groups. Similar short-term results were seen in our study.
One of the surrogate markers of surgical technique is its completeness of resection with good early postoperative outcomes, the CLASSIC trial had a small number of rectal cancer patients, which showed an increase in CRM positivity in low anterior resections operated laparoscopically, but following the long-term follow-up results, it showed no difference in the overall survival and diseasefree survival of patient whether, operated openly or laparoscopically [18, 19]. Similar short- and long-term results were also seen in COREAN trial, with no difference between open and laparoscopic surgery [13]. Similarly in our study, short-term results including conversion to open surgery, anastomotic leak rate, R0 resection, reoperation, and morbidity, and mortality were nonsignificant between the 2 groups of patients, except for the readmission rate, which was higher on the NLARC patients, due to more stoma related problems. Even major Clavien-Dindo grade III to V, requiring interventions were comparable in both arms.
Conversion from laparoscopic to open surgery in ACOSOG Z6051, ALaCart, and COLOR II was 11.3%, 9%, and 16% respectively, which is quite high with respect to our study, as 5 patients (4.6%) were converted in the LARC arm and 2 patients (1.8%) in NLARC arm with a nonsignificant P-value [6, 7, 16]. However, in COREAN trial the conversion rate was quite impressive with only 1% in the laparoscopic arm [13]. To our understanding of the trials mentioned earlier, the learning curve for laparoscopic surgery was not achieved globally, that might be one reason for having a high conversion rate for laparoscopic surgery.
A study conducted by Park et al. [20], showed that open surgery is associated with lower rates of sphincter preservation with respect to laparoscopic and robotic surgery. In our study, as comparison was between patients who were propensity score matched for tumor height, all operated laparoscopically, organ preservation was still possible in 64% of LARC and 80% of NLARC patients with an overall 72% organ preservation in both the arms.
At least 2 recent randomized control trials and meta-analysis have raised questions about the quality of specimen in patients operated by laparoscopic approach, when compared with open surgery [6, 7]. The technical difficulty of operating in a narrow pelvis, confounded by radiotherapy effects and advanced nature of tumor adds to the challenge for laparoscopic approach [3, 9, 21]. Similarly, our study showed tumor height and male sex as the risk factors for readmission on univariate and multivariate analysis.
A limitation of this study is that it had been suffered from the effect of retrospective review of prospectively collected data. Also, in a group with LARC, most patients had undergone neoadjuvant CRT, which might contribute to operative challenges and affect postoperative clinical outcomes. However, we have tried to address this issue by performing the propensity score match between the groups. Secondly, all procedures performed or supervised were by highly trained specialize surgeons with vast experience in both laparoscopy and cancer care; this may be difficult to generalize widely.
In conclusion, this study demonstrates that standardized approach to laparoscopy is safe and feasible in LARC. Contrary to the laparoscopic approach being challenged by the 2 recent noninferiority studies, laparoscopic rectal resection can still be executed in a safe and feasible fashion, even for LARC patients after neoadjuvant CRT. Although, there is still a long way to go for laparoscopic rectal cancer surgery, to be unanimously performed worldwide.
The proxy indicators of a surgical technique are dependent on its short- and long-term outcomes, the practicability of laparoscopic approach for LARC, is evident by the comparable perioperative results of the same technique for LARC and NLARC patients.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.