Characteristics and outcomes of colorectal cancer surgery by age in a tertiary center in Korea: a retrospective review
Article information
Abstract
Purpose
Colorectal cancer (CRC) occurs in all age groups, and the application of treatment may vary according to age. The study was designed to identify the characteristics of CRC by age.
Methods
A total of 4,326 patients undergoing primary resection for CRC from September 2006 to July 2019 were reviewed. Patient and tumor characteristics, operative and postoperative data, and oncologic outcome were compared
Results
Patients aged 60 to 69 years comprised the largest age group (29.7%), followed by those aged 50 to 59 and 70 to 79 (24.5% and 23.9%, respectively). Rectal cancer was common in all age groups, but right-sided colon cancer tended to be more frequent in older patients. In very elderly patients, there were significant numbers of emergency surgeries, and the frequencies of open surgery and permanent stoma were greater. In contrast, total abdominal colectomy or total proctocolectomy was performed frequently in patients in their teens and twenties. The elderly patients showed more advanced tumor stages and postoperative ileus. The incidence of adjuvant treatment was low in elderly patients, who also had shorter follow-up periods. Overall survival was reduced in older patients with stages 0 to 3 CRC (P<0.001), but disease-free survival did not differ by age (P=0.391).
Conclusion
CRC screening at an earlier age than is currently undertaken may be necessary in Korea. In addition, improved surgical and oncological outcomes can be achieved through active treatment of the growing number of elderly CRC patients.
Graphical Abstract
INTRODUCTION
Colorectal cancer (CRC) is the third most common cancer in the world and the second most cancer in Korea [1, 2]. Although the incidence rate has decreased slightly in recent years, the incidence and prevalence of CRC in Korea have increased with westernization [3-6]. In the United States, a statistical analysis based on SEER (Surveillance, Epidemiology and End Results) data reported that the 5-year overall survival rate for CRC patients from 2006 to 2012 was 66.2%, while 76.3% from 2010 to 2014 in Korea [2, 3]. This difference is believed to be the result of high level of medical treatment, active national cancer screening program, and national health insurance in Korea. However, the 5-year survival rates of CRC patients have been found to differ in different age groups [7-11]. According to Anele et al. [12], patients over the age of 60 years had lower survival rates compared to younger patients in even same stage. It is still unclear whether it was due to differences in tumor biology or because relatively passive treatments were selected for elderly patients.
Another issue related to the age of CRC patients is the use of screening programs. Currently, there are no guidelines that specifically address screening in the National Comprehensive Cancer Network (NCCN) or European Society for Medical Oncology guidelines. In Korea, the National Cancer Screening Program guidelines recommend a fecal occult blood test (FOBT) every year for those over 50 years of age. In case of abnormal results on FOBT, colonoscopy or double-contrast barium enema are recommended. Nonetheless, the FOBT is considered to have low accuracy and the initiation of screening at 50 years of age has been questioned.
In several studies, the characteristics of CRC patients in Eastern countries, especially in Korea, have been noted to differ from those in Western countries [3, 6, 13-15]. In particular, the necessity of starting screening earlier has been raised, as many cases of CRC occur at an earlier age than in the West [16-19]. In this study, we aimed to confirm the different characteristics in CRC by age group, thereby providing evidence to support future modifications of the therapeutic strategy and screening program in Korea.
METHODS
A total of 4,326 patients who underwent primary resection of CRC from September 2006 to July 2019 were included. Only patients with primary cancer and pathologically confirmed adenocarcinoma located from the appendix to the rectum were included in the study. Recurrent cancer, metastasis of other cancers, and patients who underwent diversion without resection were excluded. This study was approved by the Institutional Review Board of Korea University Anam Hospital (No. 2020AN0528) and all participants provided informed consent.
Tumors located in the appendix, cecum, ascending, hepatic flexure, and transverse colon were classified as right-sided colon cancer, while splenic flexure, descending, and sigmoid colon cancer were classified as left-sided colon cancer. Rectosigmoid colon cancers were classified as rectal cancer. Since 2006, our hospital has been performing most surgeries using a minimally invasive approach. In addition, D3 lymphadenectomy is routinely performed with curative intent for all CRC. Because the types of surgery were so diverse in this study, they were classified into lesional radical operations (including complete mesocolic excision [CME] or total mesorectal excision [TME], total surgery [total abdominal colectomy or total proctocolectomy], and limited surgery).
Perioperative evaluation and treatment were conducted based on the NCCN guidelines. All patients were evaluated preoperatively by physical examination, total colonoscopy, abdominopelvic computed tomography (CT), chest CT, and routine laboratory tests, which included tumor markers. If necessary, additional tests, such as rectal magnetic resonance imaging (MRI), liver MRI, and positron emission tomography-CT were performed. Stent insertion, neoadjuvant chemotherapy, or radiotherapy were performed before surgery if needed. Routinely, the patients were allowed water the day after surgery and a soft diet on the second day after surgery. Adjuvant treatment after surgery was performed in consideration of cancer stage, age, general condition, and socioeconomic status. Follow-up examinations were carried out at 3-month intervals during the first 2 years postoperatively, at 6-month intervals until 5 years after surgery, and then annually if there was no evidence of recurrence.
The 4,326 patients were divided by age in order to compare their clinical and tumor characteristics, operative and postoperative data, and oncologic outcomes. Descriptive results were presented as a mean or median for continuous outcomes, and as frequency and percentage for categorical outcomes. Student t-test or analysis of variance were used to compare continuous variables, and the chi-square test or Fisher exact test were applied for categorical variables. Five-year overall survival (OS) and disease-free survival (DFS) were estimated using the Kaplan-Meier method and differences in survival were analyzed with the log-rank test. All statistical analyses were performed using IBM SPSS Statistics ver. 22.0 (IBM Corp., Armonk, NY, USA). A P-value less than 0.05 was considered statistically significant.
RESULTS
CRC patients aged 60 to 69 years comprised the largest group, followed by those aged 50 to 59 years and 70 to 79 years (10−19 years, 0.1%; 20−29 years, 0.7%; 30−39 years, 2.9%; 40−49 years, 10.6%; 50−59 years, 24.5%; 60−69 years, 29.7%; 70−79 years, 23.9%; 80−89 years, 7.1%; and 90−99 years, 0.4%). Males comprised the highest ratio in the 50s and 60s, and the younger or older ages had a higher proportion of females. The presence of comorbidities, in particular cardiovascular disease, increased with patient age; however, it was present in a lower percentage of patients aged 90 to 99 years than in those aged 70 to 79 years or 80 to 89 years. The American Society of Anesthesiologists (ASA) physical status (PS) classification tended to increase with age, but the ratio of ASA PS grade I was relatively high among patients aged 90 to 99 years. The body mass index was low in very young and very elderly patients.
Although rectal cancer was most common in all age groups, the proportion of right-sided colon cancer tended to increase with age. Right-sided colon cancer was most frequently reported among patients aged 90 to 99 years. In young ages, 40% of patients represented multiple lesions. The preoperative tumor marker levels did not differ significantly between groups. Preoperative complication rate was high in very young and very elderly patients, and the rate of neoadjuvant treatment was also low in these groups (Table 1).
In patients aged 90 to 99 years, the rate of emergency surgery was significantly high. Most of the surgery was performed by laparoscopy in all age groups, and the proportion of robotic surgery was relatively high in young patients (20−29 years, 35.5%; 30−39 years, 34.4%; and 40−49 years, 30.9%), while the rate of open surgery was relatively high in the elderly (80−89 years, 7.4% and 90−99 years, 11.8%) (Table 2, Fig. 1).
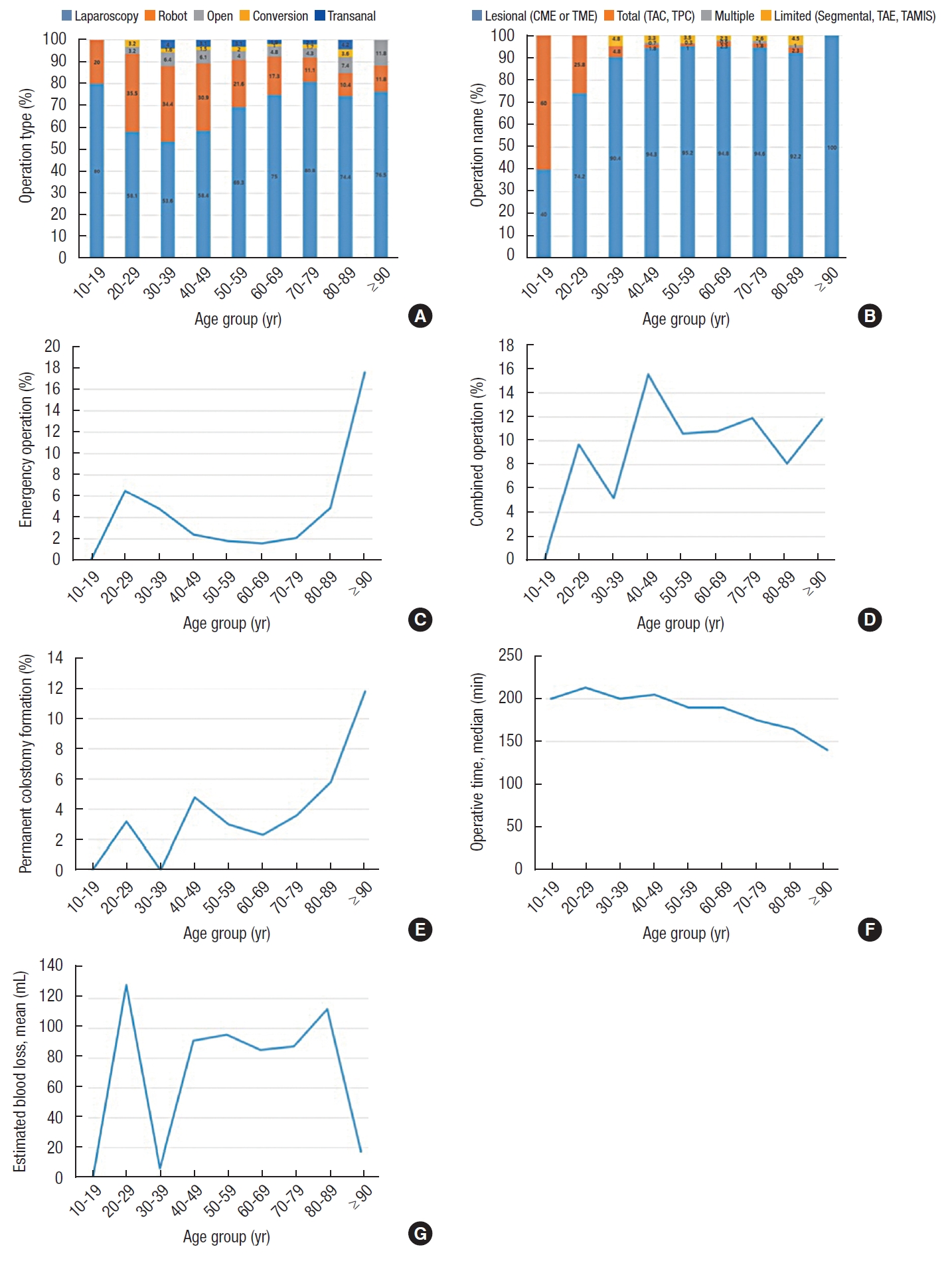
Operative data by age for the study population. (A) Operation type. (B) Operation name. (C) Emergency operation. (D) Combined operation. (E) Permanent colostomy formation. (F) Operative time. (G) Estimated blood loss. CME, complete mesocolic excision; TME, total mesorectal excision; TAC, total abdominal colectomy; TPC, total proctocolectomy; TAE, transanal excision; TAMIS, transanal minimally invasive surgery.
The majority of patients underwent radical operations, including CME or TME, while the rate of total abdominal colectomy or total proctocolectomy was high in young patients (10−19 years, 60.0% and 20−29 years, 25.8%). The proportion of patients who underwent combined operations did not differ between groups, but the rate of permanent stoma was high in elderly patients (90−99 years, 11.8%). The operation time tended to be shorter as the patient’s age increased.
In pathology evaluations, older patients had more advanced T and TNM stages, and larger tumor sizes (Table 3). However, the N stage, M stage, number of positive lymph nodes (LNs), proximal resection margin, and circumferential resection margin did not differ significantly between groups. In young patients, the number of retrieved LNs was significantly greater, and distal resection margin was longer. In elderly patients, gas passing, defecation, and soft diet intake were delayed after surgery. In contrast, the duration of hospital stay after surgery did not differ between groups. The overall complication rate was 26.4% and the leakage rate was 5.5%. In older patients, the postoperative complication rate was high: ileus occurred in a greater percentage of patients aged 90 to 99 years (35.3%). Reoperation did not differ between the groups, but postoperative mortality was significantly higher in elderly patients (Table 4).
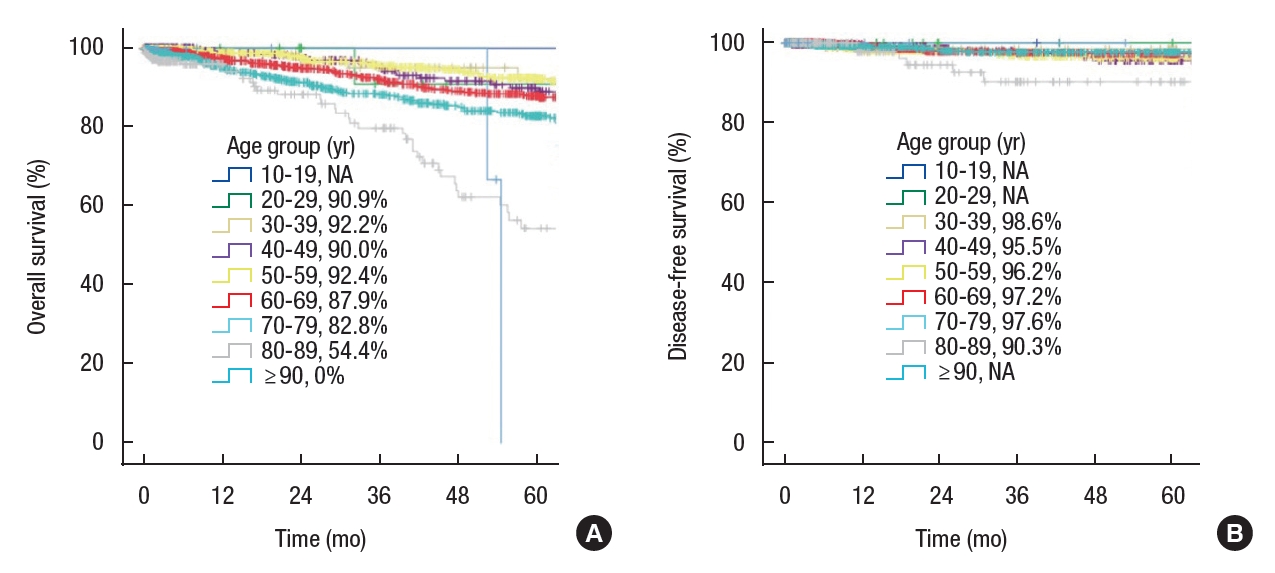
In elderly patients, postoperative treatment, especially chemotherapy, was rarely performed, and the follow-up period was shorter (Table 4). When survival analysis was performed in those with stage 1 to 3 disease, 5-year OS was significantly different between age groups (10−19 years, not applicable [NA]; 20−29 years, 90.9%; 30−39 years, 92.2%; 40−49 years, 90.0%; 50–59 years, 92.4%; 60–69 years, 87.9%; 70−79 years, 82.8%; 80−89 years, 54.4%; and 90−99 years, 0%; P<0.001); however, there was no difference in 5-year DFS (10−19 years, NA; 20−29 years, NA; 30−39 years, 98.6%; 40−49 years, 95.5%; 50−59 years, 96.2%; 60−69 years, 97.2%; 70−79 years, 97.6%; 80−89 years, 90.3%; and 90−99 years, NA; P=0.391) (Fig. 2).
DISCUSSION
In our study, we found various surgical techniques were performed for CRC in all ages, including elderly patients. Patients aged 50 to 79 years accounted for the majority of those who underwent surgery; however, even compared to the national cancer statistics in Korea, there was a greater proportion of young patients in our study population. According to Korean national cancer statistics, out of a total of 28,127 new CRC patients in 2016, 17 patients (0.06%) were 10 to 19 years of age, 98 (0.34%) were 20 to 29, 609 (2.16%) were 30 to 39, 2,202 (7.83%) were 40 to 49, 5,969 (21.2%) were 50 to 59, 7,453 (26.5%) were 60 to 69, 7,492 (26.6%) were 70 to 79, and 4,287 (15.2%) were 80 to 89. Compared to our data, the age distribution was shifted to the right. This difference is likely the result of including only patients who underwent surgery in our study.
In elderly patients, active treatments such as surgery may be selected less frequently. Although age is not an independent factor, surgical treatment for elderly patients is sometimes not recommended due to comorbidities and poor overall health [20-22]. In our study, the presence of comorbidities tended to be low among elderly patients. Perhaps this is because only patients in good general condition were selected for elective surgery. In our study, the 90s group had shorter operative times and less estimated blood loss (EBL) than patients of other age groups. The high rate of open surgery was thought to contribute to this. In addition, the absence of elective cases requiring massive dissection and the small sample size were also likely causes of low EBL deviation.
Even when considering that our study included only patients who underwent surgery, the larger proportion of younger patients compared to other countries remains valid. According to a study conducted in the United Kingdom [12], about 2.1% of CRC patients are less than 40 years old. In contrast, in Korea, the proportion of CRC patients under 40 years of age is reported to be 6.5% [4]. As a result, changes to the recommended CRC screening age have been suggested. Currently, the screening program in Korea is an FOBT once every year after the age of 50 years. If a potential problem is detected on the FOBT, colonoscopy is recommended. However, these recommendations were made based on foreign statistics, and may not be appropriate for the Korean population. In both the national cancer statistics and our study, CRC patients aged 30 to 49 years account for about 10% to 13.5% of reported cases. We, therefore, propose that screening should start earlier than is currently recommended. Even in the West, the age at which CRC is diagnosed has decreased in recent years, and efforts are being made to lower the screening age to 45 years [7, 16, 19]. Also, it is necessary to pay attention to genetic counseling [23].
In our study, the proportion of rectal cancer was particularly high (mean, 46.4%). This is thought to be because our hospital is specialized in robotic rectal cancer surgery. The distribution of cancer lesions also differed significantly between age groups, but the proportion of right-sided colon cancer was relatively high in the elderly patients [24-27]. It is consistent with other studies comparing different age groups. Yang et al. [24] reported that in patients over 70 years of age, the right-sided colon cancer rate was 2 to 3 times higher than that of young patients (right/left colon cancer ratio: 1.00 in patients < 40 years vs. 2.40 in patients > 70 years). On the other hand, young patients often had multiple lesions likely because most of them were hereditary CRCs, such as familial adenomatous polyposis or hereditary nonpolyposis CRC [28, 29]. For this reason, the proportion of young patients who underwent total abdominal colectomy or total proctocolectomy was high in our study.
At our institution, most of the elderly patients underwent minimally invasive surgery, and radical resection with curative intent was performed rather than limited surgery. Despite the relatively high rate of emergency surgery, the rate of open surgery was limited to around 10% among patients aged 80 to 99 years. The preferential use of minimally invasive surgery is expected to result in less pain, and more rapid postoperative recovery. In our previous study, we reported that it was possible to apply fast-track treatment after laparoscopic surgery in old patients [30]. In this study, although mortality and postoperative complications such as ileus occurred frequently among the elderly patients, these outcomes appear to have been the result of emergency surgery. Even in the scope of surgery, active oncologic treatment was performed for all ages. As a result, the 5-year DFS was over 90% even among patients aged 80 to 89 years. In elderly patients, it is often difficult to implement adjuvant chemotherapy, so we postulate that providing active surgical treatment will help improve the quality of life for the remainder of the patient’s life.
There are several limitations to our study. First, due to the nature of the disease, there were very few patients in their teens, 20s, and 90s, and the follow-up duration for these patients was short. In addition, analysis of cancer-specific survival was not performed. Second, data from patients who did not undergo surgery for various reasons or who received palliative surgery were not included in this study; this study design may have led to bias with regard to the age-specific composition of the disease, as only patients in good condition were selected for surgery, and survival may have been evaluated as overly good, especially among the elderly. Third, our institution may not be representative of most institutions that perform CRC surgeries. Given our hospital is one of the few institutions that specialize in minimally invasive surgery in Korea, our surgical population and surgical approaches may differ from those at other institutions.
Despite these shortcomings, this study, based on data from more than 4,000 patients, provides valuable information on the age-specific characteristics of CRC in the East, and particularly in Korea. Recently, many studies using data from the Health Insurance Corporation in Korea have been reported, and our study has the advantage that it contains clinical information that such studies cannot provide. In addition, our findings are particularly relevant in light of the aging of the Korean population, given surgery is being performed very actively even in the elderly, especially using minimally invasive techniques that provide good surgical and oncological outcomes.
In our study, we found young CRC patients were relatively common in Korea compared to Western countries, and many surgeries that used minimally invasive techniques were performed in elderly patients. When considering the age distribution of CRC patients in Korea, it is necessary to consider a protocol that starts screening from an earlier age than is currently recommended. In addition, in consideration of the ongoing aging of the Korean population, surgical and oncological outcomes may be improved by actively treating elderly CRC patients based upon their general condition rather than their chronological age.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
None.