INTRODUCTION
Surgical procedures for both benign and malignant disease of the anorectum are known to be technically demanding procedures. As far as the field of anorectal surgery is concerned, not only the short-term outcome but also the long-term functional outcome is a very important issue, probably due to the complexity of the anorectal anatomy. Both minor and major surgical procedures have real impacts on the quality of life of patients.
Because surgical techniques are based on an understanding of anatomy, accurate anatomical knowledge of the anorectum helps the surgeon to perform safe, high-quality surgery on patients with rectal diseases ranging from benign to malignant. Especially in rectal cancer surgery, an approach to the anus, such as the transanal total mesorectal excision, is attempted at a location different from that used in the conventional transabdominal approach, so a full understanding of the anatomy around the anus is now needed. Also, familiarity with the techniques used to treat patients with benign anorectal diseases, such as the hemorrhoidectomy or the fistulectomy, will be useful to surgeons performing coloanal or intersphincteric procedures for the surgical treatment of patients with distal rectal cancer. For those reason, we will discuss in this paper the basic anatomy of the anorectum and the latest knowledge on that subject. We will also describe the important structures and surgical strategies from the surgeonŌĆÖs point of view.
OVERALL ANATOMY OF THE ANAL CANAL
The anatomic anal canal is defined as beginning at the dentate line and ending at the anal verge. On the other hand, the surgical anal canal is defined as the area between the anorectal ring and the anal verge. The anorectal ring is usually palpable as a tight ring-like structure, and when patients are asked to squeeze the anal sphincter, it moves anteriorly. The anorectal ring consists of the external anal sphincter and the puborectalis muscles and is a good landmark for locating the tumor or the level of the colorectal anastomosis. The length of the surgical anal canal is 4.4 cm in men compared with 4.0 cm in women [1]. The internal anal sphincter (IAS), external anal sphincter and puborectalis muscle constitute the surgical anal canal. The anorectal ring is easily identified on digital examination and on image studies such as transrectal ultrasound or rectal magnetic resonance imaging (MRI).
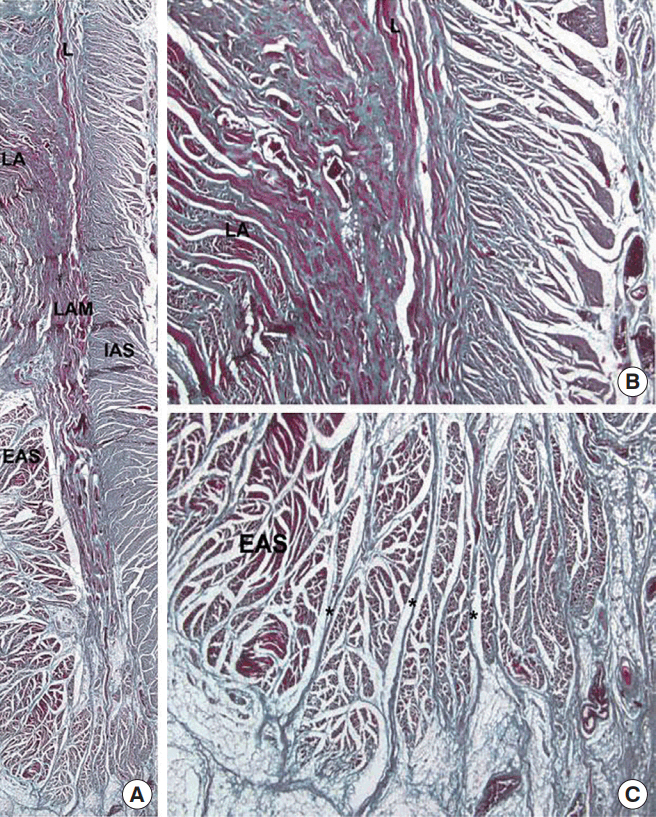
The anal canal has a cylindrical double-layered shape. The inner layer is composed of the IAS and the conjoined longitudinal muscle (CLM), which is innervated by the autonomic nerve system, whereas the outer layer is composed of the puborectalis muscle and the external sphincter muscle (EAS), which is stimulated by somatic nerves. The EAS finishes more distally than the internal sphincter. The EAS constitutes the end of the anal canal, just as it surrounds the IAS from the outside to the inside. As the orientation of the anal canal changes, the shape of the IAS, EAS, and CLM that compose anal canal also change. According to one study of the anal canal, which divided the anal canal into 9 different sagittal planes, the levator ani muscle in the anterolateral portion of anal canal is relatively thick and straight while the EAS is thin. In the lateral portion, the levator ani muscle is thick and curved whereas in the posterior portion, the levator ani muscle is thin and curved, and the EAS is thick. In contrast, the attachment length from the levator ani muscle to the CLM decreases as opposed to the gradual increase in the fraction of the levator ani muscle that overlaps the EAS from the anterolateral to the posterior position [2]. The anal canal ends at the anal verge, forming a transitional zone between the epithelium and the perianal skin. From a practical point of view, the grove between the internal and the external anal sphincter can be easily palpitated under anesthesia and should serve as a landmark for an internal sphincterotomy for anal fissure or an intersphincteric resection (ISR) for low-lying rectal cancer.
ANAL CANALŌĆÖS EPITHELIUM
The dentate line is the most important landmark for understanding the epithelium of the anal canal. The dentate line is located one third below the entire surgical anal canal. The area between the dentate line and the anal verge is called the ŌĆ£anoderm,ŌĆØ which is a nonkeratinized squamous epithelium and has no structures such as hair follicles or glands. It is also soft when palpated and sensitive to pain. During a hemorrhoidectomy, the excised portion is usually excessive or increased anoderm. When the anoderm is resected excessively, anal stricture may occur. Avoiding the underlying internal and external anal sphincter during excision of the anoderm and vascular cushions is also important. About 10 mm proximal to the dentate line, the ŌĆ£anal transitional zoneŌĆØ is found. It appears purple and represents an area of gradual transition from a squamous epithelium to a columnar epithelium. The transitional zone is known to be associated with the sampling reflex, which means that this area can discriminate feces from gas. Nevertheless, its functional role is still not clear.
The column of Morgagni begins at the dentate line, and the anal crypts are at its base. The anal crypts are connected to the underlying anal glands through the anal gland duct, and these are the presumed source of perianal abscesses and perianal fistulae. The anal gland ducts proceed from the anal canal and enter the submucosa, where two-thirds enter the IAS and half terminate in the intersphincteric plane [3]. The obstruction of these ducts is postulated to lead to anal fistulae and abscesses, which is the reason the internal opening of the cryptoglandular anal fistula is located at the dentate line. When a surgeon plans to perform a total proctectomy and coloanal anastomosis (CAA), the dividing line will be at the anorectal junction or at the dentate line (stapled CAA or hand-sewn CAA). The surgeon should consider the glandular ducts around there. With our initial CAA procedure, we experienced a couple of unexplained perianal fistulae.
Distal to the dentate line, the anoderm begins and continues for about 1.5 cm. At the anal verge, the squamous cells merge with the perianal skin. The perianal skin is also made up of squamous cells, but it has hair follicles and glands. The area below the dentate line is innervated with the inferior rectal nerve, which makes us sensitive to pain. However, the region proximal to the dentate line is innervated with the autonomic nerve system. This difference can be explained by the dentate line being a margin of different embryonic origins (endoderm vs. ectoderm).
INTERNAL ANAL SPHINCTER
When the rectum inserts into the pelvic diaphragm, the inner circular muscle of the rectum continues to the IAS and terminates approximately 1 cm proximal to the distal edge of the EAS. Overall, the IAS is found to be approximately 2ŌĆō3 mm in thickness [4] and to become thicker near the anal verge. Both the sympathetic fibers from the inferior pelvic plexus and the parasympathetic fiber through the inferior pelvic plexus and splanchnic nerves (S2ŌĆō4) innervate this muscle [5]. An anorectal manometric study showed that constant resting pressure may reflect the tone of the IAS. With solid stool in the rectum, the IAS begins to relax, and the external anal sphincter begins to contract; this is called the rectoanal inhibitory reflex.
CONJOINED LONGITUDINAL MUSCLE
The CLM, also called the longitudinal anal muscle, has been described as a vertical layer of muscular tissue interposed between the IAS and the EAS. This muscle lies on the intersphincteric space, which contains a thin layer of fatty tissue. The CLM begins at the anorectal ring as an extension of the longitudinal rectal muscle fibers and descends caudally [6]. Measuring approximately 2.5 mm in thickness, the CLM decreases in thickness with age [4], but there is no general agreement on its composition and attachments.
According to Macchi et al. [7], the CLM is mainly composed of outer striated muscle fibers and small numbers of inner smooth muscle fibers, which, based on immunohistochemical staining studies, originate from the levator ani muscle and the longitudinal muscle of the rectum. They also noted the presence of an oblique muscle bundle that went from the EAS to the IAS and vice versa. The spatial outcome was the spiral course of the striated and the smooth muscle fibers between the EAS and the IAS, which contributed to some shortening of the anal canal as well as to a narrowing of the anal canal during sphincter contraction (Fig. 1). Now, we can speculate that this muscle may contribute to maintaining continence. Moreover, Shafik [8] hypothesized that this muscle played a role in defecation. When the pubococcygeus contracts, the CLM also contracts, which makes the anal canal shorter and wider.
The anatomy of the CLM in relation to the IAS and the EAS is very important in implementing an ISR for low rectal cancer. The distal resection line may be at the intersphincteric groove (total ISR), or between the dentate line and the intersphincteric groove (subtotal ISR), or at the dentate line (partial ISR) [9]. According to Valad├Żo et al. [10], a resection of the proximal half of the internal sphincter should be considered when the incision is made on the dentate line or 1 to 2 mm distal to it. If the resection is initiated above the dentate line, but below the anorectal junction, a proximal third removal of the internal sphincter should be considered. Regardless of where the surgeon enters, the internal sphincter should be resected following the intersphincteric plane including the CLM. At the top of the EAS is the rectourethral muscle (RUM), which is on the link between the longitudinal muscle of the rectum coming from below and DenonvillersŌĆÖ fascia (DVF) coming from above. Because the RUM is known to be the tissue through which a vein and the cavernous nerve pass, complications can be prevented by dissecting into the back of this muscle [11]. In the lateral part of the ISR, the perineal plane tends to be directed dorsally to the levator ani muscle due to the overlap of levator ani muscle and the EAS. This causes a mismatch between the abdominal plane and the perineal plane. However, in this case, if the surgeon just cut the levator ani muscle between the 2 planes and that lead surgeon to complete the ISR plane [2]. The same concept can be applied for a hemilevator excision followed by an ISR for treating a tumor involving the puborectalis muscle [12]. When an ISR is planned, the surgeon should realize that the CLM may be completely broken, which can affect the patientŌĆÖs continence. K├Čhler et al. [13] reported a 29% reduction in resting anal pressure following an ISR; the squeeze pressure recovered to preoperative levels after 12 months.
At the most caudal part of the CLM, smooth muscles called the ŌĆ£corrugator cutis aniŌĆØ traverse the distal anal sphincter into the perianal skin and the ischiorectal fossa. Those smooth muscles also pass through the IAS to intersect within the subepithelial space. Any disruption of these muscles will bring about anal cushion prolapse, vascular outflow obstruction, and hemorrhoidal bleeding and thrombosis ŌĆō this is the well-known theory of symptomatic hemorrhoid disease [14]. The pathway of the corrugator cutis ani should be understood during a hemorrhoidectomy. Dissection should be done between the hemorrhoidal plexus and the IAS, not deep into IAS or even the CLM and the EAS. Therefore, lidocaine with epinephrine injection might be helpful in defining the anatomical dissection plane with adequate hemostasis.
EXTERNAL ANAL SPHINCTER
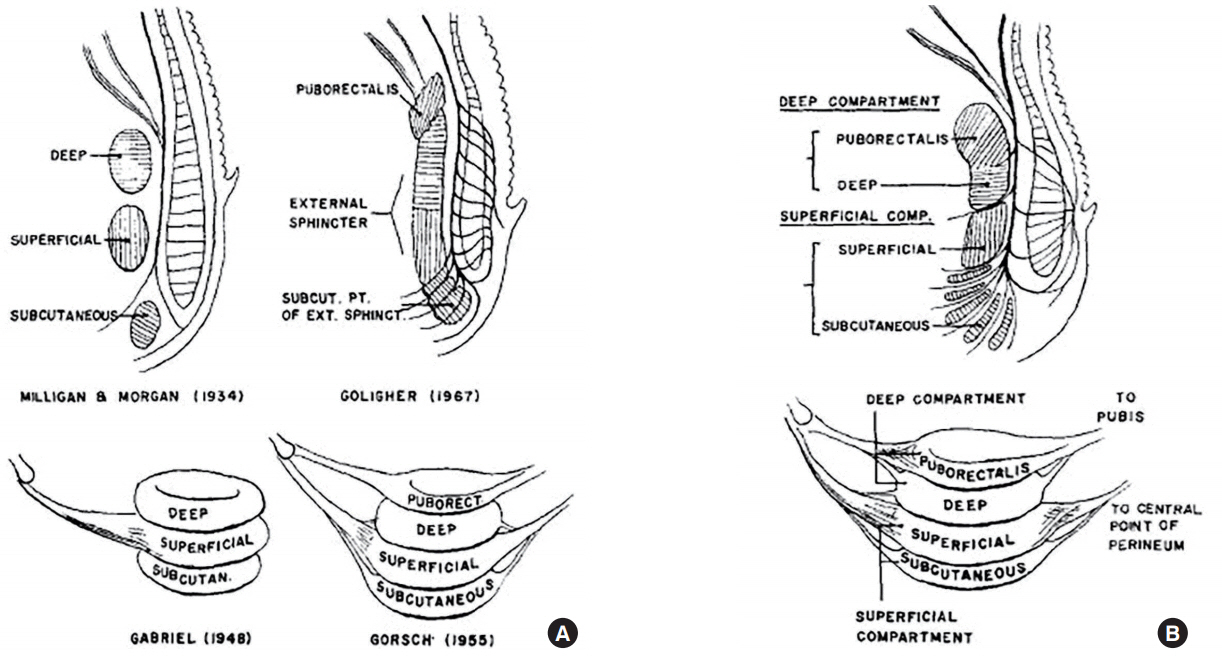
The EAS consists of a striated muscle that envelops the entire IAS and CLM. The EAS is approximately 2.7 cm high, but is anteriorly shorter in women, being approximately 1.5 cm [4]. The EAS is considered to have one or more subdivisions, although the precise composition is disputed. Milligan and Morgan [15] insisted the EAS has three distinct divisions, which are the Sphincter ani externus subcutaneous, superficialis, and profundus. However, Goligher et al. [16] failed to demonstrate these three parts of the EAS and described the EAS as one continuous sheet that is fused with the puborectalis. Oh and Kark [17] examined 48 cadaveric and surgical specimens and concluded that the arrangement and the distinction between the subdivisions are not the same at different planes. They also argued that describing the EAS as being composed of a deep compartment (deep sphincter and puborectalis) and a superficial compartment (subcutaneous and superficial sphincter) would be more appropriate (Fig. 2).
Shafik [18] suggested a triple loop system. The top loop (deep part of the EAS and puborectalis) wraps the upper part of the rectal neck and is attached to the symphysis pubis, which is innervated by the inferior hemorrhoidal nerve. The intermediate loop and the basal loop embrace the mid portion and the lower rectal neck, respectively. In addition, the perineal branch of the fourth sacral nerve and the inferior hemorrhoidal nerve innervated them, separately. Even though this concept and its functional interpretation are innovative, they are not generally accepted.
PERINEAL BODY
The perineal body (PB) is a fibromuscular mass between the upper end of the anterior anal canal and the posterior wall of the urethral membrane in males and between the posterior wall of the vagina and the anal canal in females. This area is crucial to the urologist, as well as to the proctologist who performs various types of perineal surgery: an abdominoperineal resection (APR), pull-through procedures, and repair of a rectovaginal fistula or a urethrorectal fistula. It represents the intersection of the EAS, the bulbospongiosus muscle, the external urethral sphincter, and the levator ani muscle, as striated muscle. Smooth muscles such as the RUM, CLM, and IAS are anchored to the PB [19]. Kraima et al. [19] described the PB as being both centrally located between the anal and urogenital triangle and almost completely formed by muscle fibers derived from the CLM. They also described the PB as filling the anterior gap of the cranial part of the EAS.
That superficial, deep, transverse perineal muscles contribute to the composition of the PB is generally accepted. Nevertheless, the detailed description of the PB differs with the researcher. One study using thin-slice MRI reported that the PB consisted of the bulbospongiosus, the superficial transverse perineal muscle, the IAS, the EAS, the puboperinealis muscle, and the pubo-analis [20].
RECTOURETHRALIS MUSCLE
Henle defined the muscular bundle interposed between the anterior perineal flexure of the rectum and the membranous urethra as ŌĆśprerectal.ŌĆÖ Roux was the first to use the term ŌĆśrectourethralis muscleŌĆÖ in reference to the prerectal muscle. It originates from the CLM, extends into the PB, and adheres anterolaterally to the levator ani muscle and posteriorly to the CLM [21]. Thus, it occupies the anterior portion of the rectum, which is not covered by the levator ani, and is located directly above the EAS [22].
According to Porzionato et al. [21], the RUM does not contact the posterior wall of the urethra. According to them, the mean distance between the RUM and the membranous urethra was 5.3 ┬▒ 1.25 mm in adults and 1.0 ┬▒ 0.41 mm in infants. In infant specimens, the RUM was clearly separated from the levator ani while in 8 of 10 adults (80%), the striated fibers of the levator ani and the smooth muscle fibers of the RUM intermingled. Those authors suggested that these structural associations might be related to functional cooperation between the 2 muscles.
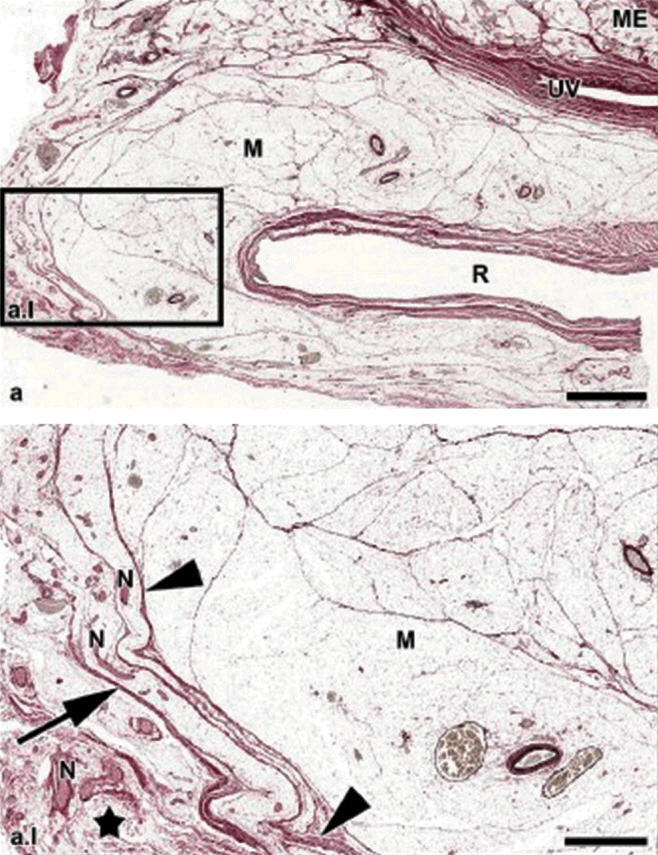
As the RUM is located superior to the PB, an understanding of this structure is essential to the surgeon performing a radical prostatectomy and an APR for prostate cancer and distal rectal cancer, respectively. Uchimoto et al. [22] showed that the cavernous nerve passed through the RUM and that the rectal vein drained into the venous plexus through the RUM. They recommended limited use of electrocauterization for hemostasis because those nerves and blood vessels might be damaged during an APR or an ISR. Furthermore, the abdominal surgical plane behind the DVF helps surgeons to guide their fingers along the external rectal muscularis propria. Palpating the external rectal muscularis propria should be helpful for adjusting the plane of the perineal approach for identifying the RUM. Moreover, Kraima et al. [19] emphasized that dissection should be started just anterior to the EAS and continue cranially, following the line between the RUM and the longitudinal muscle of the rectum. This not only preserves the autonomic nerves but also is oncologically safe when performing an APR (Fig. 3).
PELVIC FLOOR
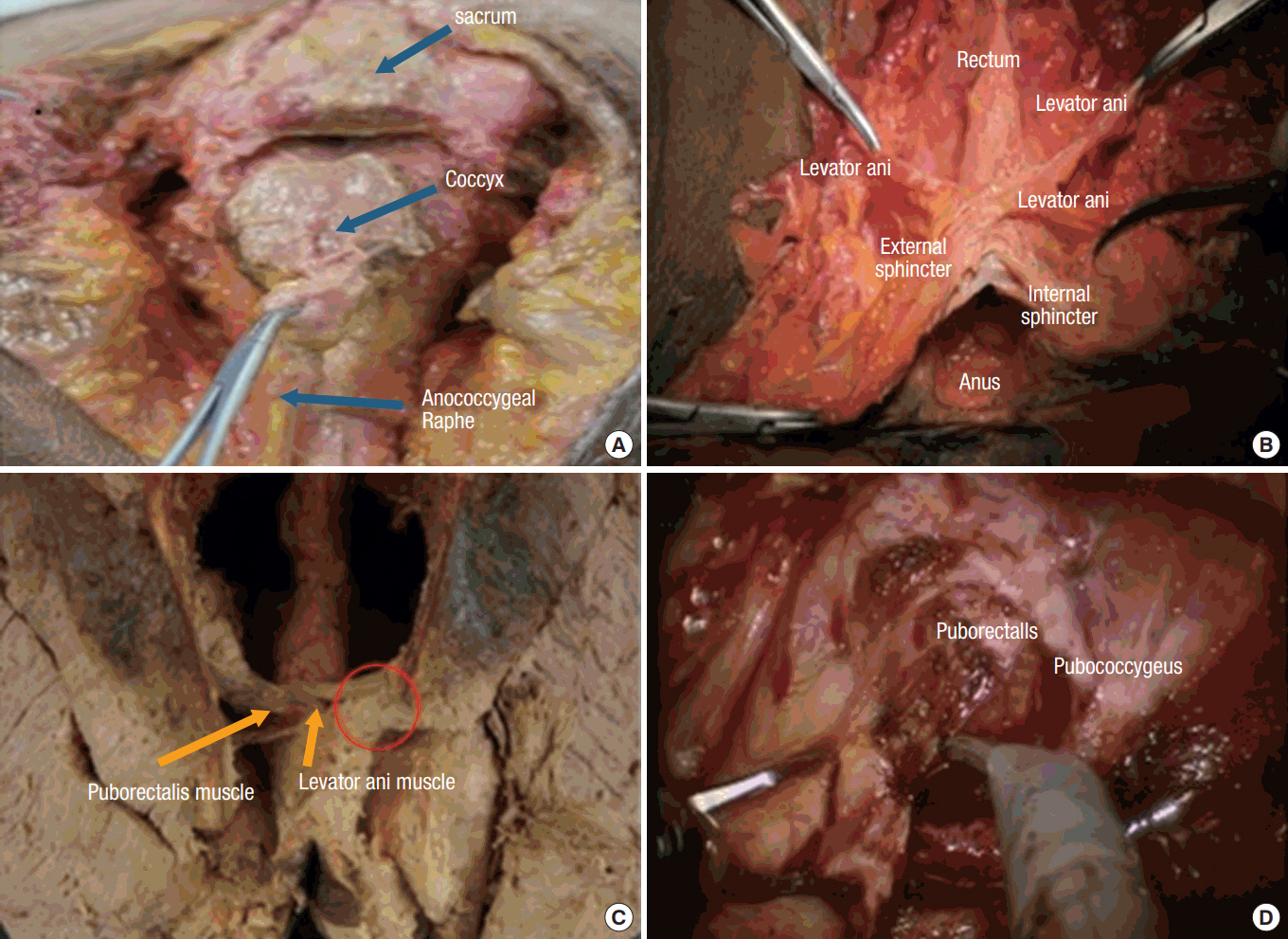
The pelvic floor is a complex structure of muscle, ligaments, and fascia with various roles. Although often thought to be a single muscular layer, the pelvic floor is actually more complex. From superior to inferior, it is composed of the endopelvic fascia, the muscular pelvic diaphragm, and the perineal membrane [5]. As for the pelvic diaphragm, it consists of the levator ani muscle (puborectalis, pubococcygeus, and iliococcygeus) and the coccygeus. Some had argued that the puborectalis was part of the deep portion of the EAS. However, imaging and embryological study showed that the levator ani muscle has a distinct muscle fascicle direction and shares the same primordium. The levator ani muscle is innervated by pudendal nerve branches and the S3 and 4 nerves. The pubococcygeus and the puborectalis muscles are primarily innervated by pudendal nerve branches such as the inferior rectal nerve while the iliococcygeus is primarily innervated by the sacral S3 and S4 nerves (Fig. 4) [5].
The levator ani muscle is thick anteriorly and mostly thin aponeurotic posteriorly. This funnel-shaped pelvic diaphragm suspends the rectum that is located just above the pelvic floor. Based on personal experience with rectal MRI, various shapes of pelvic floor, which may differ according to sex and BMI, may be observed. Its superior attachment is on the posterior surface of the pubic bone anteriorly and on the internal obturator muscle and the ischial spine laterally. Posteriorly, the levator ani muscle borders the coccygeus and the anococcygeal raphe (Fig. 5).
The pubovisceral muscle, which comprises the puborectalis and the pubococcygeus, is located medially and superiorly. The pubovisceral muscle was renamed because of its role in the suspension of the distal gut [23]. The pubovisceral muscleŌĆÖs origin is transparent and thin as it attaches tangentially to the pubic periosteum and runs laterally from the tendinous arch and ischial spine [24]. The medial part is continuous inferiorly with the PB and the CLM and is attached to the rectococcygeal fascia [23].
The clinical implication of the pelvic floorŌĆÖs anatomy in rectal cancer is the acuteness of the angle between the lateral wall of the levator ani muscle and the rectal wall, which may be related to the width of the pelvic floor, and that presents a working-space problem when dissecting the distal part during a TME. When an advanced low-lying rectal tumor is encountered, an APR or an extralevator APR should be considered so that a sufficient circumferential resection margin is obtained. In other words, if a need exists to excise the levator ani muscle at its origin, unless the anatomy of the pelvic floor is fully understood, an adequate extralevator APR cannot be performed. Actually, when the patient is in the jack-knife position, the levator ani muscle is more expanded and more visible. For an extralevator APR, some surgeons insist that the jack-knife position is necessary. Actually, circumferential margin positivity and tumor cell spillage from a perforated site are the main reasons for the poor oncologic outcomes following a standard APR for low rectal cancer. Therefore, an extralevator or a total levator excision approach has been developed to avoid coning down of the specimen. For an understanding of the correct surgical anatomical planes for these procedures, the pelvic floorŌĆÖs anatomy is essential. An en bloc resection of the pelvic floor with the mesorectal fascia (MRF) is important. That means the mesorectum should not be dissected off the levator ani muscles. Getting a cylindrically shaped specimen is important, which is the way to obtain a negative circumferential margin.
RECTUM, MESORECTUM, AND PELVIC FASCIA
Where the rectum starts is controversial because of the difference between the surgical and the anatomical anal canals. For surgeons, the rectum will be from the anorectal ring whereas for the anatomists, it will be from the dentate line. The rectum extends 12 to 15 cm proximally. The rectum can be distinguished from the sigmoid colon where the outer longitudinal muscle begins to form taenia coli. The rectum has 2 or 3 curves in the lumen, and those submucosal folds are called ŌĆśValves of Houston.ŌĆÖ From the anal verge, the rectum can be defined to have 3 parts: the lower rectum, 0ŌĆō6 cm; the middle rectum, 7ŌĆō11 cm; and the upper rectum, 12ŌĆō15 cm [25]. The lower third of the anterior portion is extraperitoneal while the posterior part is totally extraperitoneal. The mesorectum is a fatty tissue encircling the rectum and is a very important structure in a total mesorectal excision for rectal cancer. It contains the possible metastatic lymph nodes and blood vessels and is enveloped with clear distinct collagenous fiber, the so-called ŌĆśmesorectal fascia,ŌĆÖ in other words, the fascia propria. The mesorectum is thick on the posterior and lateral side of the rectum, but on the anterior side, it is thinner than it is on the other sides. The mesorectum is well known to be almost absent at approximately 2 cm above the puborectalis muscle [26]. The MRF corresponds to the visceral fascia and seems to be thicker on the posterior side of the rectum. The parietal fascia refers to the membrane that adheres to the periosteum over the sacrum (presacral fascia) (Fig. 6) [27]. Knowing the spatial relationship between the MRF and surrounding structures, such as the inferior hypogastric nerve and the pelvic plexus, during a TME cannot be emphasized enough. The ŌĆśholy planeŌĆÖ of a TME is an avascular area bordered by the MRF and the parietal fascia.
Kraima et al. [28] demonstrated that no extra constant fascial layer existed between the MRF and the parietal fascia. The MRF is a multilayered envelop that shows gaps at some points. The interruption of the MRF is more pronounced at the lower anterolateral mesorectum. The autonomic nerves are located directly lateral to the MRF, but within the parietal fascia (Fig. 7). Thus, if the mesorectum is not to be breached for oncologic reasons and at the same time, the nerves are not to be damaged to save the function, a TME should be carried out using a sharp dissection on the MRF. In other words, the closer the TME is performed to the MRF, the greater is the likelihood of autonomic nerve preservation.
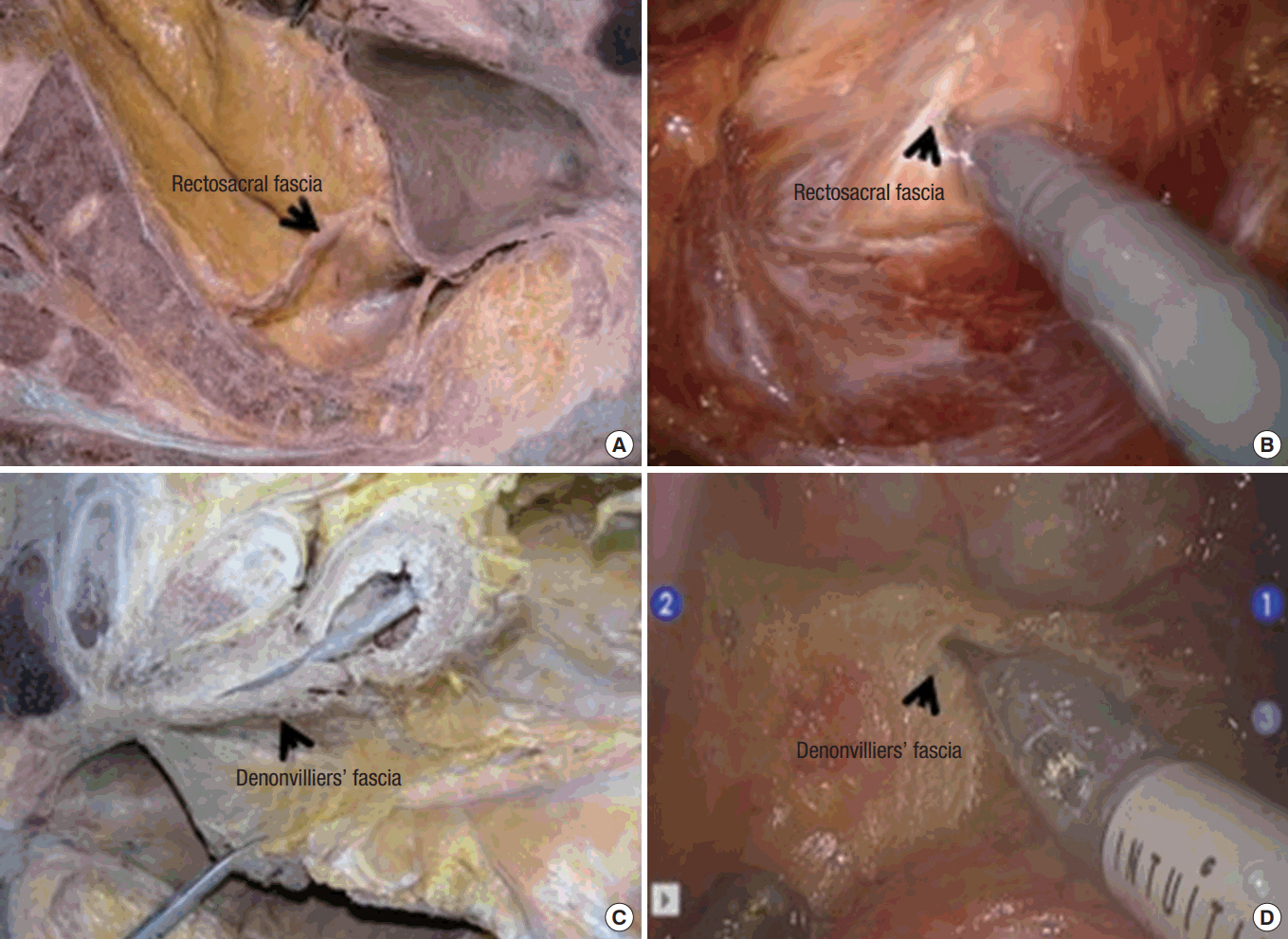
At the S4 level, relatively dense connective tissue links the presacral fascia and the MRF. This is called the rectosacral fascia (WaldeyerŌĆÖs fascia). Crapp and Cuthbertson [29] pointed out the clinical significance of this because it causes bleeding in the presacral venous plexus unless it is recognized and divided. When it is thick, a blunt dissection with the hand can cause avulsion injury to the presacral vein. A sharp division of the rectosacral fascia allows the surgeon to arrive at the level of the coccyx. If this fascia is sharply divided, dissection can enter the deep pelvic cavity; then, the gate to the pelvic floor will surely be met and the rectal wall (in this area, no mesorectum exists) will easily be separated from the pelvic floor. A better approach is to start the anterior pelvic dissection after doing the posterior dissection as much as possible. From a surgeonŌĆÖs practical point of view, such anatomical knowledge and concepts are very important.
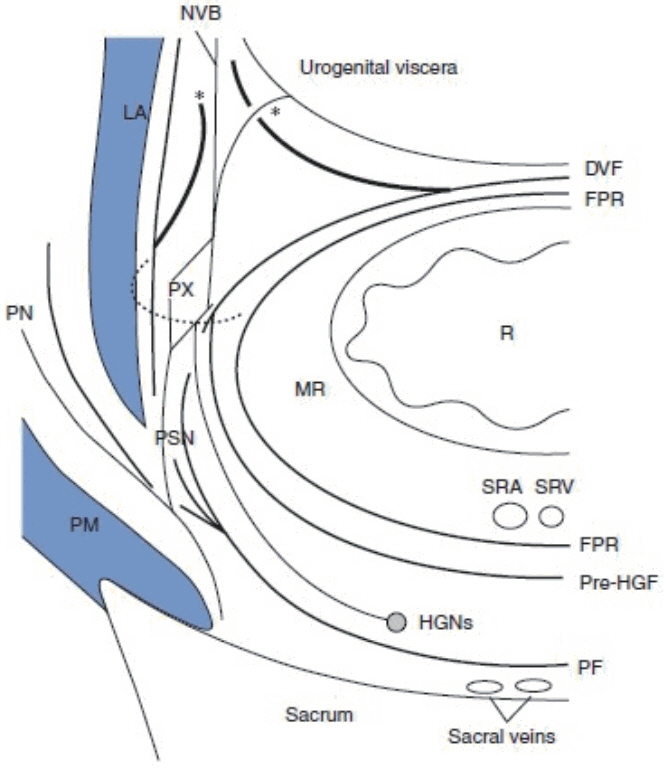
The DVF is present between the seminal vesicle and the rectum and forms a shiny, white surface. It is present as a rectovaginal septum in females. Debate exists about whether to enter anterior or posterior to the DVF. Lindsey and Mortensen [30] asserted that the mesorectal plane, immediately outside the MRF, is the anatomical plane that can minimize sexual dysfunction. They insisted the dissection should be performed between the mesorectal fascia and the DVF. However, Heald et al. [31] stated that a dissection behind the DVF was difficult and oncologically suboptimal while a dissection at the anterior plane to the DVF, called the extramesorectal plane, was natural and bloodless. He stated that a gentle backward pressure on the specimen helped the surgeon to enter the areolar tissue anterior to the DVF. According to Kraima et al. [32], the DVF begins at the top of the seminal vesicle and terminates in the PB. It consists of multiple fascial condensations of collagen and smooth muscle. Using whole mount microscopic sections of cadavers, they observed that the DVF was adhered to the posterior surface of the seminal vesicle and was indistinguishable from the MRF. At a more inferior level, some intervening space filled with adipose tissue existed between the prostatic fascia and the MRF at the posterolateral side of the prostate. However, the DVF was continuous with the MRF and the posterior prostatic fascia in the middle of the prostate, indicating that they were fused. Kinugasa et al. [33] described them as communicating branches of the bilateral pelvic plexus in front of the DVF, which compensated for each other. Lateral to the seminal vesicle, the DVF divided into multiple lamiae, and one layer of the DVF extended dorsolaterally, separating the MRF from the pelvic plexus. In a histologic evaluation using fresh cadavers, when the dissection plane was in front of the DVF, it extended laterally to the pelvic plexus. However, when the surgical plane started behind the DVF, the DVF and the pelvic plexus were clearly separated from the mesorectum.
Based on our experience, performing the anterior part of a TME following the mesorectal plane, as Lindsey proposed, is possible [30]. The extramesorectal plane is associated with a high risk of intraoperative hemorrhage and nerve injury. Unless the cancer is located at the anterior wall of the rectum and obtaining a sufficient margin is not at risk, we prefer to follow the mesorectal plane. However, in situations where a dissection is needed along the extramesorectal plane, we carefully use a U-shaped cut at the lateral border of the DVF to minimize damage to the neurovascular bundles (NVBs) (Fig. 8).
PELVIC PLEXUS AND NEUROVASCULAR BUNDLE
As the nerves from the preaortic plexus go down to the pelvis, they form the superior hypogastric nerve plexus at the level of the sacral promontory. The hypogastric nerve begins where the superior hypogastric nerve divides right and left. Each hypogastric nerve descends inferiorly and laterally and joins the pelvic splanchnic nerve (S2ŌĆō4) to form the pelvic plexus. The pelvic plexus is known to be located on the lateral side of the pelvic wall and away from the parietal fascia. One study that examined 12 autopsied cadavers showed that the pelvic plexus was located, on average, from 3.3 cm above to 2.3 cm below the peritoneal reflexion [34]. Numerous small NVBs running from the pelvic plexus to the genitalia cross the seminal vesicles in the 10 oŌĆÖclock and 2 oŌĆÖclock directions. Y-shaped pelvic autonomic nerve structures are clearly seen at the hemipelvis during cadaveric dissection (Fig. 9) [35].
Performing the TME so as to avoid injury to the pelvic plexus after initiating an anterolateral pelvic dissection is important. The MRF is directly adhered to the mesh like the pelvic plexus; this adhered portion used to be regarded as a ligament, the so-called lateral ligament. This ligamentous structure between the mesorectum and the inferior hypogastric nerve and the pelvic plexus varies in thickness. This lateral adhesion may contain a middle rectal artery. The middle rectal artery was found in only 22.2% of the cases, and in those cases, the diameter of the artery was 1.6 mm. Its origin was located 32.8 mm from the origin of the superior vesical artery, 19.9 mm from the upper border of the coccygeus muscle. The pelvic splanchnic nerves from the third and the fourth sacral nerves, which arise more posteromedially than the middle rectal artery, can be considered as components of the lateral ligament [36].
Regarding injury to the pelvic plexus, the surrounding areas should be handled carefully during mobilization of the rectum from the pelvic cavity. However, if the tumor is located near, or even invades, the pelvic wall, an en bloc resection with the pelvic plexus should be performed. Yamakoshi et al. [34] reported interesting data with the mean distances between the muscularis propria and the pelvic plexus in both autopsied and surgical specimens being 8.3 mm and 14.7 mm, respectively. The distance between the deepest point of the cancer and the pelvic plexus in pT3 patients was 5.4 mm, on average. This discovery led us to consider whether to resect the pelvic plexus concomitantly when middle or low rectal cancer has invaded deeply beyond the muscularis propria.
The NVBs run between the rectum and the posterolateral surface of the prostate. As described by Walsh and Schlegel [37, 38], they course in front of the rectogenital fascia or in the space occupied by the seminal vesicles and the prostate gland. The inferior branches of the pelvic plexus join with several vessels to form NVBs [39]. Based on WalshŌĆÖs report on a nerve-sparing radical prostatectomy, the seminal vesicles can be used as an intraoperative landmark to identify the pelvic plexus, which is punctured by branches of the inferior vesical artery and veins and is embedded in the thick fascia (Fig. 10).
During the pelvic dissection, the importance of avoiding damage to the pelvic plexus and the NVBs should be emphasized. If the lateral aspect of the rectum is to be successfully resected from the pelvic plexus, incising the rectosacral fascia for a meticulous dissection down to the coccyx and separating the MRF from the pelvic plexus are important. Proper traction of the rectum is important to prevent avulsion injury to the pelvic plexus. During the dissection of this area, the middle rectal artery is occasionally seen, and it should be ligated with surgical clips or electrocauterized. Mass ligation and too much traction can damage the third sacral nerve, which is important for erectile function. Cutting the rectosacral fascia and opening the retrorectal space usually reveals the largest nervi erigentes of the S3 component. A sharp dissection around this area is necessary, but is difficult to perform in the deep and narrow male pelvis. However, the use of gentle traction with the magnified view of a robotic instrument will make preserving these structures easier compared to open surgery for the treatment of rectal cancer.
ANORECTAL SPACES
Anorectal spaces usually refer to the spaces that border various myofascial structures such as the levator ani, obturator internus, IAS, and EAS. Anorectal spaces are important to the colorectal sutgeons for the treatment of anorectal abscesses rather than they contained other meaningful structures.
The largest anorectal space is the ischiorectal space. The ischiorectal space forms an ischiorectal fossa with the perianal space that is below the ischiorectal space. The fascia starts from the CLM and reaches out to the ischial tuberosity across the subcutaneous EAS and the rest of EAS, which is the boundary between the perianal space and the ischiorectal space [41]. This fascia is the so-called ŌĆśtransverse septum of ischiorectal fossa.ŌĆÖ The medial border of the ischiorectal space is formed by the puborectalis, pubococcygeus, and deep EAS. The lateral border consists of the obturator fascia and ischium. Anteriorly, this area is bordered by the urogenital diaphragm and the transverse perineal muscle [41]. Most of the ischiorectal space is filled with large lobules of fat. The pudendal nerve and vessels locate superolaterally in this space.
The perianal space means the lower part of the anal canal surrounded by the anoderm. It is in contact laterally with the fatty tissue of the buttock and medially with the intersphincteric space. This space is a common site for perianal abscesses or hematomas. The intersphincteric space lies between the IAS and the EAS. It is filled with areolar tissue and fat. This space is continuous cephalad to the supralevator space that is situated between the peritoneum and the levator ani muscle. As described earlier, the anal gland secretes into the anal canal through the anal crypt along the level of the dentate line. Because some anal glands are in the intersphincteric space, this space plays a major role in the development of perianal fistulae and accounts for about half of the total incidence.
The superficial postanal spaces are interposed between the skin and the anococcygeal ligament. The deep postanal spaces occupy the region between the levator ani and the anococcygeal ligament. Superficial and deep postanal spaces communicate with the ischiorectal fossa and become locations where the horseshoe abscesses can occur. The inflammatory process may communicate between the ischiorectal fossa via the deep postanal spaces (space of Courtney). The retrorectal space is located between the mesorectal fascia and the presacral fascia. This space is continuous with the retroperitoneum upwards.
CONCLUSION
We reviewed the basic anatomy of and recent research results on the anorectum and looked at the strategy of surgery from the perspective of the surgeon. Until recently, anatomists have been actively studying the anatomy of this area and have been able to confirm the detailed analysis and new interpretation of the ambiguous structure. In particular, the study of the complex structure of the RUM and the PB seems to have given surgeons a new perception of the dissection plane. When the APR is implemented, preservation of the RUM without damage to the carvernous nerve or veins pass through the RUM is important. In addition, when the TME is performed, autonomic nerves pass immediately outside the MRF, so a more elaborate dissection is necessary. Also, performing dissection on the mesorectal plane in terms of autonomic nerve preservation is reasonable because the connecting branches of the pelvic plexus pass anterior to the DVF. We recommend that incise along the extramesorectal plane when CRM positivity is expected. Having adequate knowledge of anorectal anatomy is the most important factor for improving the oncologic and the functional outcomes in patients. Oncologic surgical principles for cure and function preservation based on anatomical knowledge and an updated minimal invasive surgical technique are essential for surgeons, so this trinity of surgical science must be mastered. However, because anorectal anatomy is not yet fully understood, we hope that additional studies on its anatomy will enable anorectal surgery to be performed based on complete anatomical knowledge.