- Search
|
|
Abstract
Purpose
Anemia is associated with poor treatment results for a variety of cancers. The effect of low hemoglobin levels on long-term outcomes after the treatment of patients with an anal squamous cell carcinoma (SCC) remains unclear. For that reason, this study aimed to investigate the effect of anemia on treatment outcomes following chemoradiation for an anal SCC.
Methods
This was a retrospective study of all patients who underwent curative treatment for an anal SCC between 2009 and 2015 at 2 trusts in the United Kingdom. Data were collated from prospectively collected cancer databases and were cross-checked with operating-room records and records in the hospitalsŌĆÖ patient management systems.
Results
We identified 103 patients with a median age of 63 years (range, 36ŌĆō84 years). The median overall survival was 39 months (range, 9ŌĆō90 months), and the disease-free survival was 36 months (range, 2ŌĆō90 months). During the follow-up period, 16.5% patients died and 13.6% patients developed recurrence. Twenty-two people were anemic prior to treatment, with a female preponderance (20 of 22). No differences in disease-free survival (P = 0.74) and overall survival (P = 0.12) were noted between patients with anemia and those with normal hemoglobin levels. On regression the analysis, the combination of anemia, the presence of a defunctioning colostomy, lymph-node involvement and higher tumor stage correlated with poor overall survival.
Although an anal squamous cell carcinoma (SCC) is an uncommon malignancy of the anal canal, its incidence has been steadily increasing. In 2014, 1,307 new cases were reported in the United Kingdom (UK) [1]. Since 1974, chemoradiotherapy has been the mainstay of treatment, with salvage surgery reserved for patients whose response to chemoradiotherapy is incomplete or who develop recurrence [2].
The efficacy of chemoradiation is partially mediated by the production of reactive oxygen species (free radicals) caused by oxygen-accepting electrons within cancer cells. Free radicals can damage DNA, which can initiate apoptosis within target cells. The efficacy of radiotherapy and chemotherapy is, therefore, improved in well-oxygenated tissues, but reduced in anemic tissues [3, 4]. Bone marrow suppression and subsequent anemia are common side effects of chemoradiotherapy and may compound low pretreatment hemoglobin levels to affect response to treatment and overall survival adversely [5, 6]. For that reason, the aim of this study was to determine the correlation between the response to chemoradiotherapy in patients with anal squamous cell cancer and their prechemoradiotherapy hemoglobin level.
We conducted a retrospective review of all cases of anal SCC between 2009 and 2015 at 2 trusts in the UK (Plymouth Hospitals NHS Trust and Abertawe Bro Morgannwg University Health Board). Data were obtained from prospectively updated cancer databases and crosschecked with patient notes, blood results, and other laboratory results. IRB approval and informed consent were waived according to the institutional rule.
All patients underwent examination under anesthesia rectum and radiological staging in the form of a computed tomography scan of the abdomen and pelvis or magnetic resonance imaging. Patients with inguinal lymphadenopathy additionally underwent an ultrasound-guided biopsy. When clinically indicated, some of the patients underwent a positron emission tomography scan, and all cases were discussed by the colorectal or anal multidisciplinary team. The TNM version 5 staging system developed and maintained by the American Joint Committee on Cancer and the Union for International Cancer Control was used to standardize staging [6].
The majority of patients received chemotherapy in the form of 5-fluorouracil plus mitomycin. A minority of patients, instead, received capecitabine plus mitomycin at the treating oncologistŌĆÖs discretion. All radiotherapy was performed as per standard protocol. Laboratory reference ranges for hemoglobin were acquired, along with variations in the normal range due to age and gender. The hematology laboratories defined these as 120ŌĆō155 g/L for females aged 18 and above and 130ŌĆō175 g/L for their male counterparts. Prechemoradiotherapy hemoglobin levels were compared between patients who developed recurrence and those who remained disease free.
Statistical analyses were performed using the StatsDirect version 3.0 for Windows (StatsDirect Ltd., Cambridge, UK). Kaplan-Meier survival curves were calculated and compared using the log rank test. The results were presented as a hazard ratio (HR) and the 95% confidence interval (CI). For multivariate analyses, we used a Cox regression model. For other comparisons between treatments, the Mann-Whitney test was used for continuous or ordered categorical data; the chi-square test was used when appropriate. P = 0.05 (2-sided) was considered the limit of significance.
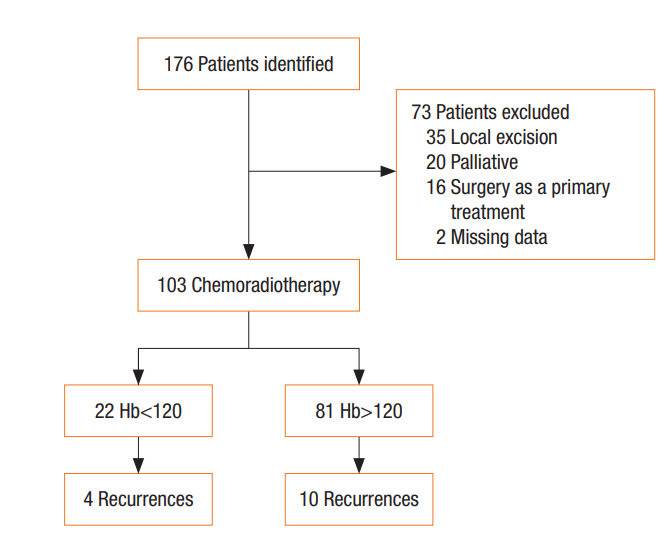
Across the 2 trusts, 176 patients were diagnosed with anal SCC during the study period. Patients who were treated with local resection or who were deemed palliative were excluded from further analysis. Patients were also excluded if they opted for no treatment. After applying the exclusion criteria, we identified 103 patients who received chemoradiotherapy with curative intent and were, therefore, suitable for further analysis (Fig. 1).
Of the 103 patients, 80 were female and 23 were male. The median age was 63 years (range, 36ŌĆō84 years). The median overall survival was 39 months (range, 9ŌĆō90 months), and the median disease-free survival was 36 months (range, 2ŌĆō90 months). During the follow-up period, 16.5% (n = 17 of 103) of the patients died, and 13.6% (n = 14 of 103) experienced recurrence. The median time from treatment to recurrence was 16.5 months (range, 2ŌĆō22 months). The median survival time from the time of recurrence to death was 7 months (range, 0ŌĆō36 months). PatientsŌĆÖ demographic details are displayed in Table 1.
Twenty-two patients in this study were anemic prior to treatment, with a female preponderance (18 of 22). In the anemic population, 5 patients experienced recurrence. No difference in the development of recurrence was noted between patients with anemia (low hemoglobin level, LHB) and those with normal hemoglobin levels (NHB) (P = 0.74; HR, 1.23; 95% CI, 0.36ŌĆō4.23). In addition, no difference in overall survival was noted between patients with LHB and those with NHB (P = 0.12) (Fig. 2).
Of the 103 patients included in the study, 18.4% (n = 19 of 103) had a defunctioning colostomy prior to their chemoradiotherapy treatment. No difference in recurrence was noted between patients with or without a defunctioning colostomy (P = 0.33; HR, 1.79; 95% CI, 0.56ŌĆō5.76). Patients with stomas had reduced overall survival time (P = 0.008) (Fig. 3). A trend towards shorter overall survival for men was noted (P = 0.056; HR, 0.39; 95% CI, 0.11ŌĆō1.3), with no difference in disease-free survival (P = 0.29; HR, 2.87; 95% CI, 0.74ŌĆō11.19).
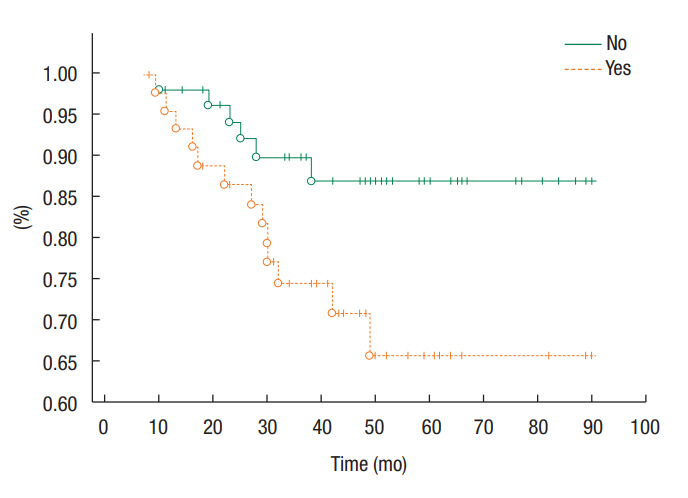
Amongst the patients included in the study, 10, 28, 31, and 27 were staged as having a T1, T2, T3, and T4 tumor, respectively. The staging status for 7 patients was not recorded, and 4 patients had tumors classified as T3/T4. The distribution of anemia by stage of disease is detailed in Table 2. No difference in overall survival or disease-free survival was found in regard to stage of tumor (P = 0.32 and P = 0.16, respectively). The presence of nodal disease was associated with lower overall survival (P = 0.013), but not reduced disease-free survival (Fig. 4). Thirteen patients had stage I disease, 34 stage II, 26 stage IIIA, and 29 stage IIIB. No difference in overall survival or disease-free survival was found between patients with clinical stage I/II or III tumors (P = 0.66; HR, 1.25; 95% CI, 0.47ŌĆō3.28; and P = 0.68; HR, 1.25; 95% CI, 0.43ŌĆō3.65, respectively).
We also performed regression analyses for factors such as anemia, tumor stage, lymph-node involvement, and presence of a defunctioning colostomy. In this model, none of the individual factors statistically influenced survival; however, the simultaneous presence of all of them reduced overall survival (P = 0.05) (Table 3).
Within the literature, the correlation between prechemoradiotherapy treatment anemia and poor treatment outcome has been widely discussed for other cancers, including lung, breast, cervix, and hematological malignancies [7]. Oblak et al. [8] specifically examined the association between patientsŌĆÖ hemoglobin levels prior to commencement of chemoradiotherapy for squamous cell cancer of the anal canal and outcome of treatment. In that study of 100 patients, an hemoglobin >120 g/L was an independent prognostic indicator for survival, in keeping with established literature for other cancers. However, interestingly, the multivariate analysis in the Oblak study revealed no association between anemia and disease-free survival. Our study confirms that finding of no association between anemia and disease-free survival, but in contrast to the Oblak study, we additionally found no association between pretreatment anemia and survival. Instead, the regression analysis demonstrated that a combination of multiple factors, such as anemia, the presence of a defunctioning stoma, a higher tumor stage and lymph-node involvement, correlated with poor survival post treatment. No individual factor displayed independent significance. Such interplay of factors may suggest a more complex or even a cumulative mechanism conferring poorer survival on these patients. Banerjee et al. [9] explored the interaction of hematological parameters and subsequent response to chemoradiotherapy in patients with an anal SCC. They demonstrated significantly poorer prognosis in patients with pretreatment leukocytosis, an association strengthened by coexisting anemia. This suggests a cumulative effect, like the one observed in our data.
The importance of treating anemia is recognized for a variety of solid and hematological malignancies. Iron deficiency is thought to be the major contributing factor to anemia in this setting [10]. Supplementation is advocated even in the context of normal ferritin levels, when low transferrin levels still represent reduced bioavailability of iron, despite normal reserves [11]. Whilst treatment with iron +/ŌĆō erythropoiesis stimulating agents is correlated with normalizing hemoglobin levels [12], clear evidence for improved performance status and better treatment outcomes has not been demonstrated. We await the published long-term follow-up of these patients [13], but a lack of clear improvement in survival post treatment despite normalizing hemoglobin again hints at a more complex interplay of factors conferring poor outcome in patients with an anal SCC.
This study has limitations. Due to the relative rarity of anal SCCs, we analyzed small subpopulation groups, which can make drawing clear conclusions statistically fraught. In particular, no association between stage of disease and overall survival or recurrence of disease was found in this study. This is most likely due to a type-1 error resulting from the small number of patients. Moreover, the recruitment of patients from only 2 tertiary referral centers means a risk of selection bias is possible. Additionally, we did not examine the effect of potentially confounding comorbidities.
In this study, the presence of anemia influenced neither disease-free survival nor overall survival. This is in contrast to the findings published in the current literature. Regression analysis revealed poorer survival in patients with a combination of anemia, lymph-node involvement and defunctioning stoma. We suggest that the interaction between anemia and survival is more complex than previously demonstrated and is potentially reliant on other coexisting factors. This may be the reason simply reversing anemia has not, to date, been shown to improve survival.
Fig.┬Ā2.
Kaplan-Mayer curve showing the percent of patients surviving at a given time (overall survival) in relation to the presence or absence of anemia. NHB, normal hemoglobin level; LHB, low hemoglobin level.
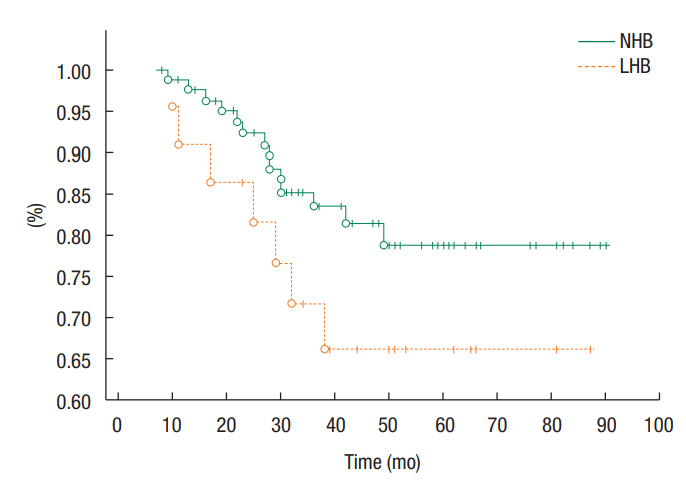
Fig.┬Ā3.
Percent of patients surviving at a given time (overall survival) in relation to the presence or absence of a defunctioning stoma.
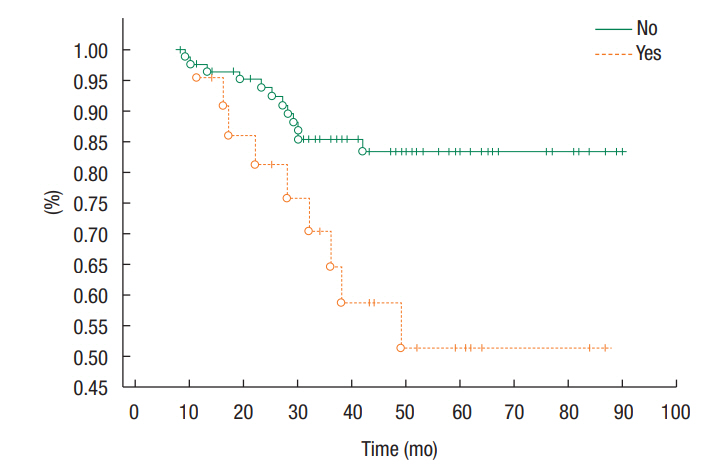
Fig.┬Ā4.
Percent of patients surviving at a given time (overall survival) in relation to the presence or absence of lymph-node involvement.
Table┬Ā1.
Demographics and survival data of patients with squamous cell anal cancer undergoing curative radiotherapy
Table┬Ā2.
Tumor staging and anemia status of patients with squamous cell anal cancer undergoing curative radiotherapy [6]
| Stage of disease | Anemic | Normal hemoglobin |
|---|---|---|
| I | 3 (14) | 13 (15) |
| II | 6 (27) | 28 (33) |
| III | 10 (45) | 40 (48) |
| IV | 3 (14) | 3 (4) |
| Total | 22 | 84 |
Table┬Ā3.
Cox regression model for tumor stage, presence of a stoma, involvement of lymph nodes, and presence of anemia
| Variable | Beta | Standard error | t-value | P-value |
|---|---|---|---|---|
| Tumor stage | ŌĆō0.691 | 0.864 | ŌĆō0.799 | 0.42 |
| Stoma | 0.595 | 0.519 | 1.146 | 0.25 |
| Lymph-node involvement | 1.520 | 0.855 | 1.757 | 0.08 |
| Anemia | 0.829 | 0.505 | 1.642 | 0.10 |
REFERENCES
1. Cancer Research UK. Anal cancer statistics [Internet]. London, Cancer Research UK; [cited 2017 Jul 3]. Available from: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/anal-cancer
2. Nigro ND, Vaitkevicius VK, Considine B Jr. Combined therapy for cancer of the anal canal: a preliminary report. Dis Colon Rectum 1974;17:354ŌĆō6.


3. Harrison LB, Chadha M, Hill RJ, Hu K, Shasha D. Impact of tumor hypoxia and anemia on radiation therapy outcomes. Oncologist 2002;7:492ŌĆō508.


4. Kitano T, Tada H, Nishimura T, Teramukai S, Kanai M, Nishimura T, et al. Prevalence and incidence of anemia in Japanese cancer patients receiving outpatient chemotherapy. Int J Hematol 2007;86:37ŌĆō41.


5. Barrett-Lee PJ, Ludwig H, Birgeg├źrd G, Bokemeyer C, Gasc├│n P, Kosmidis PA, et al. Independent risk factors for anemia in cancer patients receiving chemotherapy: results from the European Cancer Anaemia Survey. Oncology 2006;70:34ŌĆō48.


6. American Joint Committee on Cancer. Implementation of AJCC 8th Edition Cancer Staging System [Internet]. Chicago (IL), American Joint Committee on Cancer; c2018 [cited 2017 Jul 3]. Available from: https://cancerstaging.org/About/news/Pages/Implementation-of-AJCC-8th-Edition-Cancer-Staging-System.aspx
7. Caro JJ, Salas M, Ward A, Goss G. Anemia as an independent prognostic factor for survival in patients with cancer: a systemic, quantitative review. Cancer 2001;91:2214ŌĆō21.


8. Oblak I, Cesnjevar M, Anzic M, Hadzic JB, Ermenc AS, Anderluh F, et al. The impact of anaemia on treatment outcome in patients with squamous cell carcinoma of anal canal and anal margin. Radiol Oncol 2016;50:113ŌĆō20.



9. Banerjee R, Roxin G, Eliasziw M, Joseph K, Maclean A, Buie WD, et al. The prognostic significance of pretreatment leukocytosis in patients with anal cancer treated with radical chemoradiotherapy or radiotherapy. Dis Colon Rectum 2013;56:1036ŌĆō42.


10. Ludwig H, Van Belle S, Barrett-Lee P, Birgeg├źrd G, Bokemeyer C, Gasc├│n P, et al. The European Cancer Anaemia Survey (ECAS): a large, multinational, prospective survey defining the prevalence, incidence, and treatment of anaemia in cancer patients. Eur J Cancer 2004;40:2293ŌĆō306.