- Search
|
|
Abstract
Carcinoembryonic antigen (CEA) is not normally produced in significant quantities after birth but is elevated in colorectal cancer. The aim of this review was to define the current role of CEA and how best to investigate patients with elevated CEA levels. A systematic review of CEA was performed, in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. Studies were identified from PubMed, Cochrane library, and controlled trials registers. We identified 2,712 papers of which 34 were relevant. Analysis of these papers found higher preoperative CEA levels were associated with advanced or metastatic disease and thus poorer prognosis. Postoperatively, failure of CEA to return to normal was found to be indicative of residual or recurrent disease. However, measurement of CEA levels alone was not sufficient to improve survival rates. Two algorithms are proposed to guide investigation of patients with elevated CEA: one for patients with elevated CEA after CRC resection, and another for patients with de novo elevated CEA. CEA measurement has an important role in the investigation, management and follow-up of patients with colorectal cancer.
Carcinoembryonic antigen (CEA) was first isolated from human colorectal cancer (CRC) tissue in 1965 by Gold and Freedman [1,2]. It is a foetal glycoprotein and is not usually produced in significant quantity after birth. CEA can become elevated in a number of pathologies. The most common clinical use is surveillance for recurrence of CRC. CEA levels are also sometimes measured in patients without a history of CRC. Elevated CEA in both situations presents a management dilemma to the colorectal surgeon, particularly in deciding the appropriate tests and the subsequent follow-up if those tests are inconclusive, and this led us to conduct this review. This review aims to evaluate the role of CEA in clinical colorectal practice, including the prognostic significance of CEA in patients with CRC, the role of CEA in follow-up after CRC resection, and the management of patients with raised CEA and no history of CRC.
A systematic search was performed in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. The MeSH headings “Carcinoembryonic antigen,” “CEA,” and “Colorectal Neoplasm” were used to identify relevant studies in the PubMed and Cochrane Library Databases.
Studies included had to be published in English from January 1, 1990, to April 30, 2017, be full text articles, and have comparison groups in which the CEA level was used to determine a component of management or outcome. Data were extracted from the included articles by 2 authors according to the aims of our review: prognostic significance of CEA in patients with CRC, role of CEA in follow-up after CRC resection, and management of patients with raised CEA and no history of CRC. Results were described qualitatively. No statistical analyses were performed.
Of the 2,712 papers identified, 271 were screened in abstract form by 2 authors independently. Thirty-two were deemed relevant, after which the full texts of the articles were obtained and reviewed by 2 authors (Fig. 1). A hand search of the reference lists found no additional studies.
Most trials were retrospective (Table 1). Elevated preoperative CEA predicted overall survival across stage I to stage III CRC, with elevated preoperative CEA carrying a 62% increase in the danger of death compared with a normal CEA level [3]. The 5-year disease-free survival was 84.6% vs. 69.8% [4], and the 5-year overall survival was 74.5% vs. 63.4% [5]. However, a study of 2 cohorts with Duke C cancer, 1990–2000 and 2001–2004, found that the predictive value of preoperative CEA was lost in the more recent cohort [6], and may be due to improved chemotherapy regimens. Higher preoperative CEA levels were associated with advanced disease stage.
Kim et al. [7] hypothesized that after curative surgery, and therefore after resection of the source of CEA, CEA levels would decrease exponentially. CEA was measured preoperatively and at day 7 and day 30 postoperatively. In the patient group where the CEA level had decreased exponentially, survival was significantly greater than it was in the group where CEA remained elevated, with a trend to increased disease-free survival. Similarly, in patients with stage 4 CRC who underwent a R0 resection, elevated postoperative CEA and CA 19-9 were associated with reduced disease-free survival [8]. Failure of the CEA to return to normal after surgery was indicative of residual or recurrent disease, with CEAs over 10 ng/mL being strongly associated with metastatic disease.
In rectal cancer, elevated pretreatment CEA levels are associated with a poor response to neoadjuvant treatment, with a retrospective analysis of patients with stage I–III rectal cancer showing significantly decreased pathological complete response, pathological tumor regression, tumor downstaging, and overall survival [9].
Two recent meta-analyses have shown no survival benefit with increased surveillance after resection for CRC [10, 11]. For CEA specifically, we identified 8 randomized controlled trials (RCT, Table 2). In a RCT looking at managing a rising CEA with a second-look laparotomy compared to conservative treatment [12], no survival advantage was noted, and the study was terminated. CEA began increasing 323 days prior to the onset of clinically evident recurrent disease. A subgroup analysis was not performed, so whether the R0 resection of recurrent or metastatic disease had improved disease-specific survival compared to those with unresectable disease was not clear. The results were also difficult to interpret in the modern setting, as today, a ‘second look’ laparotomy would only be performed on patients with proven resectable disease, and this selected group would be expected to have an improved survival if compared to patients with widespread disease.
More recent trials [13-21] compared a standard follow-up regimen to a higher intensity follow-up, though the regimens were heterogeneous (Table 3). In the FACs (Follow-up After Colorectal Surgery) trial [15], more intensive follow-up with CEA and computed tomography (CT) scanning increased the rate of surgical treatment for recurrent disease, but without survival advantage. The accuracy of a single CEA measurement was poor, with the conclusion that the CEA trend, rather than stand-alone measurements, should be used [22].
The Dutch trial ‘CEAwatch’ investigated the effect of the intensity of the CEA measurement on the detection of recurrent disease and the proportion of curable recurrences [16]. Here, an elevated CEA would trigger repeat CEA testing and, if maintained, chest/abdomen CT. Intensive surveillance (8 weekly CEA) detected more patients with recurrence who had undergone a curative treatment, defined as an R0 resection, compared to the control group (3 monthly CEA: 35% vs. 22%) [16]. No improvement in overall or disease-free survival was found. The only survival benefit was when recurrences were detected by using CEA or imaging as compared to those detected by using patients’ self-reported symptoms [23]. Intensive CEA testing was also found to be cost effective [17]. The GILDA (Gruppo Italiano di Lavoro per la Diagnosi Anticipata) trial investigated intensive surveillance by imaging for patients with Dukes B2-C CRC [18]. In the intensive surveillance arm, recurrences were detected 5.9 months earlier, but no difference in overall survival was reported.
The threshold of serum CEA used to trigger further investigations is controversial and has been addressed by a Cochrane review including 52 studies [24]. Sensitivity and specificity were 82% and 80% for a 2.5 ng/mL cutoff, 71% and 88% for 5 ng/mL, and 68% and 97% for 10 ng/mL, respectively. The review concluded that 10 ng/mL should be used as the threshold to trigger further investigations. Furthermore, the diagnostic accuracy of the postoperative CEA level in detecting recurrence is affected by the preoperative value. With a threshold of 5 ng/mL at 6 months, in patients with curatively resected colon cancer and normal preoperative CEA, the diagnostic accuracy for recurrence was 89.1%, in contrast to patients with an elevated preoperative CEA in whom the accuracy was 58.4%; an increased threshold of > 8 ng/mL in this latter group improved diagnostic accuracy to 75.6% [25].
The hypothesis that CEA level reflects tumor burden in patients with metastatic disease has also been studied by assessing treatment response to chemotherapy. Huang et al. [26] retrospectively analysed 447 patients with metastatic CRC who had undergone resection of the primary tumor and adjuvant chemotherapy. The ratio of posttreatment (after 6 courses of chemotherapy) to pretreatment CEA was strongly correlated with the radiological response and with overall survival [26]. Similarly, in patients with metastatic CRC receiving first-line chemotherapy, there were distinct CEA level slopes according to type of radiological response i.e., progression, partial response, and stable disease [27], suggesting that CEA levels and ratios have a role in the early assessment of treatment response in stage IV CRC. In the FIRE-3 trial, the CEA trend reflected response to targeted therapy [28] in patients with (K)RAS wild-type metastatic CRC. First-line FOLFIRI plus cetuximab gave better overall survival than FOLFIRI plus bevacizumab (28.7 months vs. 25.0 months) [29], with a corresponding faster and greater decrease in CEA level. In the cetuximab arm, CEA responders, defined as a decrease of at least 75%, had better disease-free and overall survivals than CEA nonresponders.
At the cellular level, a correlation between the immunohistochemical status of CEA staining and the serum CEA level was found in colorectal metastases, but not in the primary tumors [30], suggesting that serum CEA levels are influenced both by the production of CEA in tumor cells and by its release into the bloodstream. Positive staining for CEA in metastatic tissue also carried a worse survival. Overexpression of CEA has been reported to inhibit Natural Killer cells and cytotoxic lymphocytes, which may dampen the immune response to metastases [31].
Whilst no specific trials have addressed what to do for a patient with a raised CEA and no history of CRC, studies have evaluated CEA in screening and diagnosis (Table 4). In patients with elevated CEA, the chance of finding CRC is increased (4.6% vs. 1.3%) [32]. CEA is also a predictor of other diseases, including other cancers, diabetes, chronic lung disease and liver disease [33]. A combination of biomarkers, including CEA, CA 19-9, prostate-specific antigen, and CA125, increases the sensitivity of CEA as a screening modality [34, 35]. In a patient with raised CEA, a detailed history and clinical assessment are likely to be the most important first steps. No evidence was found to support CEA being used as a screening tool.
The available evidence supports the use of CEA in surveillance of CRC, particularly in postoperative follow-up. However, no evidence supports its use in screening and diagnosis of CRC, as CEA may be elevated in patients with other pathologies [2, 36-38] and many CRC patients will have a normal CEA. Serum levels > 3-5 ng/mL are deemed to be elevated [39, 40]. Higher baseline levels occur in males, smokers and the elderly [39, 40]. Elevated CEA levels have been associated with a number of benign and malignant conditions (Table 5). CEA is used most frequently in CRC, but it is also a tumor marker in mucinous adenocarcinomas of the endocervix and ovary, as well as in keratinising squamous cell carcinomas of the cervix [41]. CEA levels >10 ng/mL or trending upwards are more commonly associated with malignant conditions [40-43]. Levels > 20 ng/mL are suspicious for metastatic disease [42, 44-46]. As CEA is primarily metabolized in the liver, hepatic dysfunction and biliary obstruction can be associated with raised CEA levels.
In view of the high first-pass hepatic metabolism of CEA (≥90%), very high CEA levels tend to be due to CEA-producing tumors or metastases outside the portal venous drainage territory or to locally advanced tumors within the portal venous system’s drainage territory. Tumor differentiation also affects CEA level [45-47], with 80% of well-differentiated CRCs producing CEA and only 60% of poorly-differentiated tumors produce it [48], making surveillance less reliable. Following CRC resection in patients with normal hepatic function, a 95% reduction to the steadstate postoperative CEA level takes five half-lives, i.e., 35 days [39, 44, 47].
CEA levels correlate with prognosis [49-51]. In CRC, elevated preoperative CEA level (>5 ng/mL) is associated with a higher recurrence rate and disease-related mortality [50-54]. A postoperative CEA decrease is also a prognostic indicator [55, 56], predicting improved overall survival and disease-free survival [28]. This also applies to those having liver surgery for colorectal metastases [57]. CEA level has also been correlated with the presence of circulating cancer cells [54].
The role of pretreatment CEA level in predicting response to chemotherapy or radiotherapy is uncertain, with some studies showing no correlation [58] and with others showing that, compared to CEA <3 ng/mL, an elevated pretreatment CEA (>9 ng/mL) was associated with a poor response to long-course chemoradiotherapy [59]. Reduction of CEA following neoadjuvant chemoradiotherapy for rectal cancer has prognostic significance [60], with CEA ≤5 ng/mL being correlated with increased complete clinical and pathological response and better overall and disease-free survivals. High posttreatment CEA levels may, therefore, identify patients for adjuvant treatment and intensive surveillance, and may perhaps be a relative contraindication to ‘watch and wait’ in patients with apparently complete clinical response.
Surveillance recommendations for CEA in CRC patients vary. The American Society of Clinical Oncology (ASCO) recommends preoperative CEA and postoperative CEA every 3–6 months for at least 5 years [61, 62]. The UK National Institute of Clinical Excellence recommends CEA levels be measured every 6 months for at least 3 years [63]. In addition, ASCO [61] recommends CEA for monitoring the response of metastatic disease to systemic therapy. The CEA doubling time is affected by rate of growth of metastases and by site of recurrence [64, 65].
A patient with an elevated or progressively rising CEA level with previous CRC should be investigated for recurrent disease (Fig. 2), initially with cross-sectional imaging (CT scan or positron emission tomography-CT [PET-CT]) of the chest, abdomen and pelvis, and not colonoscopy because of CEA’s first-pass metabolism. Both the British Royal Society of Radiology [66] and the US oncology guidelines [67] suggest only performing a PET-CT when other imaging has been normal. If these investigations are negative, a repeat CEA level with clinical review at 3 months is suggested. If the CEA level remains stable without clinical evidence of recurrence, continued CEA measurements every 3 months and clinical review are encouraged. If the CEA level is increasing or >10 ng/mL or if clinical evidence of recurrence is found, repeat cross-sectional imaging is suggested.
While CEA should not be used as a diagnostic tool, it often is by the overexuberant health professional. This then raises the dilemma of determining if the elevated CEA is significant, in the absence of a previous history of CRC (Fig. 3). If the patient has a history of previous CEA-producing malignancy (Table 5), the patient should be evaluated in accordance with that particular malignancy.
In the absence of a history of a previous CEA-producing tumor, evaluation begins with a thorough history and clinical examination, looking for relevant symptoms and signs of CEA-producing benign and malignant conditions (Table 5), and smoking history. Further investigations would include repeat CEA, full blood count, and iron studies, liver and renal function tests, determinations of the CA 125 and calcitonin levels, and so on, as indicated from the clinical review.
If the clinical review does not raise any suspicion of a particular disease process and CEA is <10 ng/mL, no further investigations are recommended at this stage. The patient should be advised to stop smoking for cardiovascular and respiratory health. Determination of the CEA level and a clinical review should be repeated at 3 months. If the level falls, determination of the CEA level and a clinical review should be repeated at 6-month intervals until the CEA level returns to normal or until 2 consecutive decreases are noted. If, however, after 3–6 intervals, the CEA level remains above 5 ng/mL or if the level exceeds 10 ng/mL at any stage, further investigations (CT scan, PET-CT or organ-specific investigations) are indicated.
If the CEA level is greater than 10 ng/mL at the time of presentation, a PET-CT or whole-body CT can be used to look for primary and/or secondary malignancy, unless clinical review indicates a likely site of malignancy to guide a more specific investigations Depending on access and local costs, using a PET-CT scan before organ-specific testing might be more cost effective. If these preliminary tests are negative, a repeat CEA should be performed 3 months later with a clinical review. The clinical review is more important at this stage. A CEA level persistently >10 ng/mL at 1 year requires repeat investigation. Rising CEA levels >10 ng/mL or levels > 20 ng/mL require investigation every year until the underlying cause is detected.
In the clinical scenario of patients with de novo elevated CEA, our practice would be to investigate with whole-body PET-CT before organ-specific investigations, unless clinical evidence suggesting a specific site for the malignancy is found. If PET-CT scanning is not available, we suggest whole-body CT.
Fig. 1.
Identification, screening, and selection of papers for inclusion. CEA, carcinoembryonic antigen.
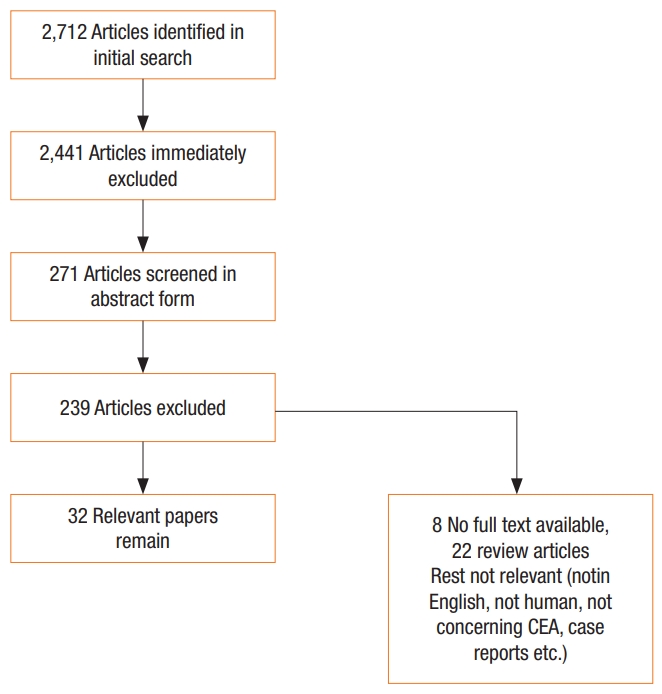
Fig. 2.
Algorithm 1: Investigation pathway for a patient with a raised carcinoembryonic antigen (CEA) with a previous history of a CEA-expressing cancer. aOrgan-specific investigations: tumor markers, CT scan, colonoscopy, gastroscopy, mammography, cystoscopy, Ultrasound Scan (US), bone scan, biopsy, other test as required. PET/CT, positron emission tomography-computed tomography.
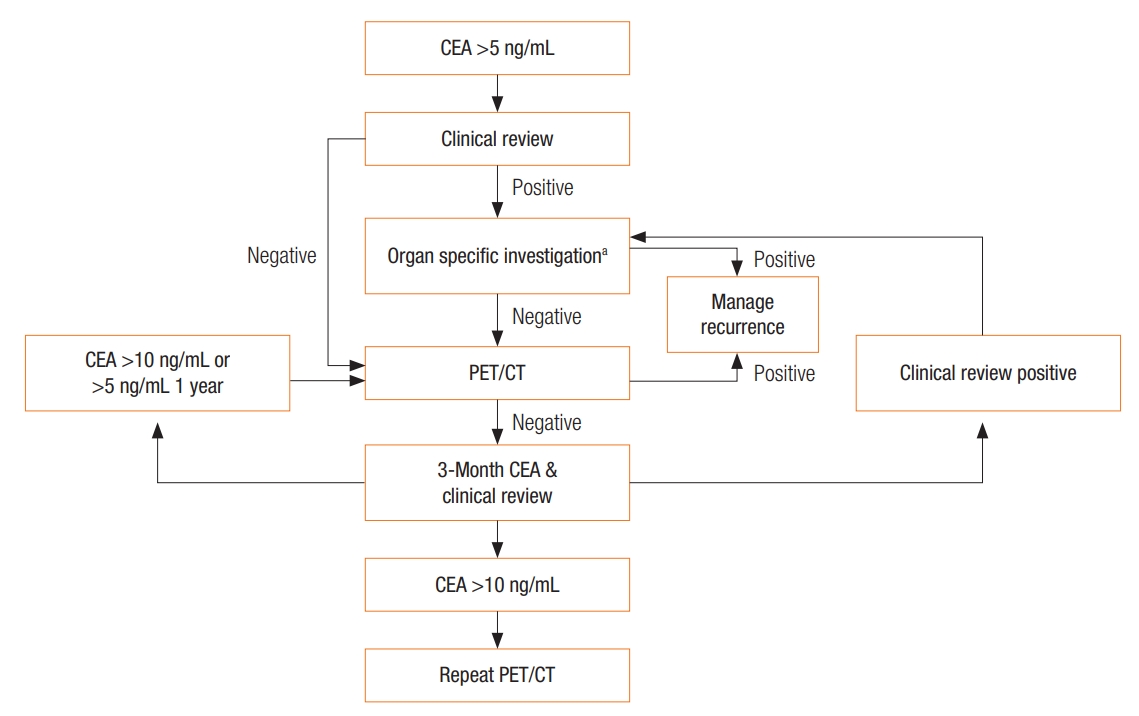
Fig. 3.
Algorithm 2: Investigation pathway for a patient with a de novo raised carcinoembryonic antigen (CEA). aOrgan-specific investigations: tumor markers, CT scan, colonoscopy, gastroscopy, mammography, cystoscopy, US, bone scan, biopsy, other test as required. Clinical review includes: a full history and examination of the thyroid, breast, thorax, abdomen and pelvis, visual field testing, fundoscopy, long bones examination. Look for melanoma. In females, cervical examination. In males, a prostate examination. PET/CT, positron emission tomography-computed tomography.

Table 1.
CEA and prognosis: selected studies
| Author | Year | Origin | Study type | Study summary | Endpoint | No. of patients | Age (yr)a | Sex ratio (%male) | Outcome summary |
|---|---|---|---|---|---|---|---|---|---|
| Stelzner [68] | 2005 | Germany | Cohort (retrospective database) | Database review for predictors of survival in colorectal cancer | Overall survival in stage IV colorectal cancer | 186 | 68.6 (range, 30–92) | 54.3 | Preop CEA ≥ 5 ng/mL associated with decreased overall survival in stage IV disease. |
| Katoh [6] | 2008 | Japan | Cohort (retrospective database) | Review of preop CEA levels in Dukes C patients | Survival | Retrospective: 237; prospective: 197 | 54.4% ≥ 60 in retrospective; 63.5% ≥ 60 in prospective | Retrospective: 59.1; prospective: 57.4 | CEA ≤ 2.5 ng/mL was a predictor of disease-free survival in Dukes C in earlier cohort. Association lost in later cohort, possibly due to improved chemotherapy. |
| Kim [7] | 2009 | Korea | Cohort (retrospective data base) | Postoperative analysis of CEA preop and day 7 and 30-day post op | Survival | 122 | 57.56 ± 12.24 | 62.3 | Significant decrease in survival if CEA levels remain elevated in postop period. |
| Sun [69] | 2009 | Taiwan | Cohort (retrospective data base) | Retrospective review of potential prognostic markers | Disease-free and overall survival | 1,367 | 66 (IQR, 19-25) | 55.4 | Preop CEA ≥ 5 ng/mL 2.38x more likely to die of cancer than those with CEA ≤5 ng/mL (P ≤ 0.001) |
| Thirunavukarasu [70] | 2015 | USA | Cohort (retrospective SEER database) | Review of preop CEA levels | 5-Year overall and disease-specific mortality | 16,619 | 67.4 ± 13.8 | 49.7 | Elevated CEA was associated with worse overall and disease-specific mortality |
| Becerra [3] | 2016 | USA | Cohort (retrospective data base) | Review of preop CEA levels in Stage I-III CRC | Overall survival | 69,512 | Normal CEA: 69.1 ± 13.0; elevated CEA: 70.3 ± 12.9 | Normal CEA: 49.0; elevated CEA: 43.7 | Preop CEA level is associated with overall survival |
| Ozawa [4] | 2017 | Japan | Cohort (retrospective National Cancer Database) | Review of preop CEA levels | 5-Year disease-free survival | 7,296 | 65.3 ± 11.2 | 54.4 | Preop CEA level is independently associated with 5-year disease-free survival |
| Spindler [5] | 2017 | USA | Cohort (retrospective National Cancer Database) | Review of preop CEA levels in Stage II CRC | 5-Year overall survival | 74,945 | Normal CEA: 70 (IQR, 59-79); elevated CEA 72 (IQR, 6081) | Normal CEA: 49.3 Elevated CEA: 43.4 | Preop elevated CEA is associated with reduced 5-year overall survival |
| Kim [71] | 2017 | Korea | Retrospective and prospective cohort | To determine cutoff values for preop CEA in Stage III CRC | 5-Year overall and disease-free survival | Retrospective cohort: 965; prospective cohort: 268 | Retrospective: 60 (range, 14-84); prospective: 60 (range, 27-80) | Retrospective: 53.9 Prospective: 48.9 | A cutoff value of 3 ng/mL is optimal. Preoperative CEA above this level is associated with inferior overall and disease-free survival. |
Table 2.
CEA and follow-up after CRC resection: selected studies
| Author | Year | Origin | Study type | Study summary | Endpoint | No. of patients | Age (yr), median (range) | Sex ratio (%male) |
|---|---|---|---|---|---|---|---|---|
| Treasure [12] | 1984 (published in full 2014) | UK | RCT | 티evated CEA prompted randomization to either continued monitoring (conventional arm) or laparotomy (aggressive) | Survival | 216 | Conventional: 62 (35-75); aggressive: 64 (33-75) | Conventional: 63; aggressive: 56 |
| Makela [72] | 1995 | Finland | RCT | Intensive vs. conventional follow-up | Recurrence | 106 | Conventional: 69 (33-85); intensive: 63 (33-81) | Conventional: 50; intensive: 48 |
| Ohlsson [20] | 1995 | Sweden | RCT | No follow-up vs. intensive follow-up | Recurrence | 107 | - | 47.7 |
| Pietra [21] | 1998 | Italy | RCT | Conventional or intense follow-up | Recurrence | 207 | - | - |
| Primrose [15] | 2014 | UK | RCT | Intensive vs. minimal follow-up 3 different intensive groups | Detection and curative treatment of recurrence | 1,202 | CT: 69 (62-76); CEA: 69 (6375); CT & CEA: 70 (64-76); minimal: 70 (63-75) | CT: 61.2; CEA: 61.3; CT & CEA: 61.3; minimal: 61.3 |
| Verberne [16] | 2015 | Netherlands | RCT (stepped wedge) | Standard vs. intensive follow-up | CRC recurrence | 3,223 | 70 (26-95) | 56 |
| Wille-Jørgensen [13] | On-going trial | Europe | Multicenter randomized controlled trial | Intensive vs. nonintensive follow-up | CRC recurrence and mortality | - | - | - |
| Lepage [14] | On-going trial | France | Multicenter randomized controlled trial | Standard vs. intensive follow-up | 5-year overall survival | - | - | - |
Table 3.
CEA and follow-up after CRC: outcome measures in selected studies
| Trial | Type of regimen | Time to first recurrence (mo) | Proportion of recurrences | Overall 5-year survival | Outcome summary |
|---|---|---|---|---|---|
| Treasure (CEASL) [12] | All patients had clinical review 3/12 for 2 years and 6/12 for the next 3 years. CEA was measured monthly for the first 3 years and 3/12 for the next 2 years. | Median time from primary surgery to CEA increases and randomization was 403 days (range, 103–1,754) | Conventional: 82%; aggressive: 77% | Not reported. End of trial figures below | Second look laparotomy in the event of CEA elevation did not improve survival. |
| Patients were randomized if the CEA became elevated (≥10 ng/mL). | Conventional: 82% died | ||||
| 216 Patients were randomized. | Aggressive: 84% died | ||||
| Conventional: Continued clinical monitoring with clinicians blinded to increased CEA. | |||||
| Aggressive: Clinician screen for widespread metastatic disease examinations and CXR. If not found, patient had a laparotomy to look for recurrence. | |||||
| Makela [72] | Clinic review for all 3/12 for 2 years, the 6/12 for 3 years with history, examination, complete blood cell counts, faecal occult bloods, CEA and CXR. | Conventional: 15 ± 10; intensive: 10 ± 5 | Conventional: 39%; intensive: 42% | Conventional: 54%; intensive: 59% | Intensive follow-up leads to earlier detection of recurrence. |
| USS liver every 6/12 and CT every year. | Most common sign indicating recurrence was CEA elevation in both groups. | ||||
| In addition: Conventional: rigid sigmoidoscopy at each visit for those who had undergone surgery for rectal and sigmoid cancers, and a barium enema was done for all patients at 12 months and once a year thereafter; Intensive: colonoscopy 3 months after the surgery to ensure a clean colon and once a year thereafter on allpatients | |||||
| Ohlsson [20] | None/Control: No FU | - | Control: 33%; intensive: 32% | Control: 67%; intensive: 75% | Intensive follow-up did not improve survival. |
| Intensive: clinical exam, rigid sig, colonoscopy, CT pelvis (in patients with APR), pulmonary X-ray, liver function tests, CEA and faecal hemoglobin at 3-, 6-, 9-, 12-, 15-, 18-, 21-, 24-, 30-, 36-, 42-, 48-, and 60-month intervals | |||||
| Pietra [21] | Conventional: clinical exam + CEA and USS at 6/12 intervals for 1 year then annually thereafter. | Conventional: 20.2 ± 6.1; iIntensive: 10.3 ± 2.7 | Conventional:19.4%; intensive: 25.2% | Conventional: 58.3%; intensive: 73.1% | Intense follow-up leads to earlier detection of recurrent disease and improved survival. |
| All patients received yearly CXR, colonoscopy and CT. | |||||
| Intensive: As above, but with intervals 3/12 for 2 years then 6/12 for 3 years | |||||
| Primrose (FACS) [15] | Control: CT at 12–18 months or if symptomatic | - | Cancer recurrence in 16.6% of patients, 5.9% of these surgically treated with curative intent | Intensive follow-up (any group) detected recurrence earlier and increased rate of curative surgical treatment. No advantage when using CT and CEA in combination. Could not demonstrate survival advantage. | |
| Intensive: CT: Scan of the chest, abdomen, and pelvis every 6 months for 2 years, then annually for 3. CEA: CEA every 3 months for 2 years, then every 6 months for 3 years, with a single CT scan at 12 to 18 months if requested at study entry by hospital clinician. CT and CEA: Both of the regimes combined. | |||||
| All had colonoscopy at 2 and 5 years | |||||
| Rosati (GILDA) [18] | Control: Clinical review and CEA every 3 months for 2 years, then every 6 months for 3 years. Colonoscopy at 1 year. Liver USS at 4 months and 16 months. | Intensive surveillance had earlier detection of 5.9 months (95% CI, 2.71–9.11) | Overall recurrence rate: 20.4%; control: 18.7%; intensive: 22% | Control: 52.7%; intensive: 47.8% | Intensive surveillance detected recurrences earlier, but there was no difference in overall survival. Quality of life was not affected by surveillance strategy. |
| Intensive: Clinical review and CEA as per control group. CBC and CA 19-9 included with CEA. Colonoscopy and CXR every 12 months. Liver USS every 4 months for 16 months, then yearly | |||||
| Verberne (CEAWatch) [23] | Control: 5-year follow-up. Clinic every 6/12 for 3 years, then annually thereafter. Liver USS and CXR at each visit. CEA every 3 to 6 months for 3 years and annually thereafter. | Specific time interval not given; however, the authors stated that the time to diagnosis of recurrent disease decreased with the intensive follow-up protocol as compared to the control protocol (HR, 1.45; 95% CI, 1.08–1.95; P = 0.013) | Overall recurrence rate: 7.5%; control: 3.6%; intensive: 4.4% | No difference in OS or DFS between 2 arms. Survival significantly worse when detected by patients self-report rather than CEA or imaging. | An intensified protocol with CEA monitoring and assessment of CEA rise rather than absolute value detected recurrences earlier than the standard protocol. This does not affect overall or disease-free survival. |
| Intensive: bimonthly CEA and yearly imaging for 3 years. CEA every 3/12 for next 2 years. Annual clinic review with imaging of chest and abdomen for 3 years. If 20% increase in CEA, another blood sample was drawn 4 weeks later. If a consecutive rise, CT scan of chest and abdomen was advised. Normal value was considered to be ≤2.5 ng/mL | |||||
| Wille-Jørgensen (COLOFOL) [13] | Nonintensive: CT scan of liver and lungs (or CT of liver + plain X-ray of lungs) + CEA after 12 and 36 months. | - | - | - | Recruitment ended 2015. Results awaited. |
| Intensive: CT scan of liver and lungs (or CT of liver + plain X-ray of lungs) + CEA after 6, 12, 18, 24, and 36 months | |||||
| Lepage (PRODIGE 13) [14] | Standard: Abdo USS every 3/12 for 3 years, 6/12 for 2 years, then annually. CXR ever 6/12 for 3 years, then annually. | - | - | - | Recruitment ended. Results awaited |
| Intensive: A CT thorax/abdominal/pelvis alternating with abdominal USS every 3/12 for 3 years, then every 6/12 for 2 years. CEA every 3/12 for 3 years, then 6/12 2 years |
FACS, Follow-up After Colorectal Surgery; GILDA, Gruppo Italiano di Lavoro per la Diagnosi Anticipata; CEAWatch, Carcino-Embryonic Antigen Watch; CEA, carcinoembryonic antigen; CRC, colorectal cancer; CT, computed tomography; FU, follow-up; CXR, chest X-Ray; USS, UltraSound Scan; CBC, complete blood count; CI, confidence interval; HR, hazard ratio; OS, overall survival; DFS, disease-free survival.
Table 4.
CEA and screening/diagnosis: selected studies
| Author | Year | Origin | Study type | Asymptomatic vs. symptomatic | Endpoint | No. of patients | Age (yr), median | Sex ratio (%male) | Outcome summary |
|---|---|---|---|---|---|---|---|---|---|
| Lee [32] | 2011 | Korea | Case control | Asymptomatic | Detection of colorectal cancer | 546 | (A) 56 | A) 77 | 4.6% of high CEA group had CRC compared to 1.3% of normal CEA group. |
| (A) CEA ≥5 ng/mL | (B) 54 | B) 73.4 | CEA more likely to be elevated in advanced colorectal cancer. | ||||||
| (B) CEA ≤5 ng/mL | |||||||||
| Nielsen [33] | 2011 | Denmark | Case control | Symptomatic | Detection of colorectal cancer | 4,509 | 61 | 45.9 | CEA more likely to be elevated in advanced colorectal cancer. Median of 8.1 ng/mL in stage 4 disease |
| Wild [35] | 2010 | Germany | Case control | Mixed | Comparison of serum-biomarker panel with Faecal occult blood testing | 1,027 | (A) 67 | (A) 52.2 | CEA more likely to be elevated in advanced colorectal cancer. 88.2% sensitivity in stage 4 disease. |
| (A) CRC 301 | (B) 62 | (B) 39.1 | |||||||
| (B) GI disease control 104 | (C) 66 | (C) 58.7 | |||||||
| (C) Adenoma 143 | (D) 62 | (D) 46.1 | CEA, seprase, CYFRA 21-1, ferritin and anti-p53 biomarker combination was comparable with faecal immunochemical testing with 82.4% versus 81.8% at 95% specificity, | ||||||
| (D) Other disease 141 | (E) 64 | (E) 42.6 | |||||||
| (E) Other malignancy 176 | |||||||||
| Wen [34] | 2015 | Taiwan | Case control | Mixed | Detection of cancer using panel of 8 markers | 41,516 | CEA sensitivity 53.8%. Increases, when used in combination as panel of 8 biomarkers, to 76.9% |
Table 5.
Conditions associated with elevated carcinoembryonic antigen (CEA) level
| Nonmalignant | Malignant |
|---|---|
| Smoking | Tumors associated with high CEA expression |
| Infections | Colorectala |
| Peptic ulcer disease | Ovariana |
| Inflammatory bowel disease | Cervicala |
| Pancreatitis | Lung |
| Hypothyroidism | Oesophageal |
| Liver cirrhosis, hepatitis | Gastric |
| Benign breast conditions | Small intestinal |
| Other benign tumors usually in organs where the cancers are associate with raised CEAd | Hepatobiliary |
| Pancreatic | |
| Breast | |
| Medullary | |
| Other CEA-expressing tumors | |
| Choriocarcinoma | |
| Osteosarcoma | |
| Retinoblastoma | |
| Hepatoma | |
| Melanoma | |
| Lymphoma | |
| Urinary bladder, prostate and renal cell carcinoma |
REFERENCES
1. Gold P, Freedman SO. Demonstration of tumor-specific antigens in human colonic carcinomata by immunological tolerance and absorption techniques. J Exp Med 1965;121:439–62.




2. Thomson DM, Krupey J, Freedman SO, Gold P. The radioimmunoassay of circulating carcinoembryonic antigen of the human digestive system. Proc Natl Acad Sci U S A 1969;64:161–7.



3. Becerra AZ, Probst CP, Tejani MA, Aquina CT, González MG, Hensley BJ, et al. Evaluating the prognostic role of elevated preoperative carcinoembryonic antigen levels in colon cancer patients: results from the national cancer database. Ann Surg Oncol 2016;23:1554–61.



4. Ozawa H, Kotake K, Hosaka M, Hirata A, Nakagawa Y, Fujita S, Sugihara K. Incorporation of serum carcinoembryonic antigen levels into the prognostic grouping system of colon cancer. Int J Colorectal Dis 2017;32:821–9.



5. Spindler BA, Bergquist JR, Thiels CA, Habermann EB, Kelley SR, Larson DW, et al. Incorporation of CEA improves risk stratification in stage II colon cancer. J Gastrointest Surg 2017;21:770–7.



6. Katoh H, Yamashita K, Kokuba Y, Satoh T, Ozawa H, Hatate K, et al. Diminishing impact of preoperative carcinoembryonic antigen (CEA) in prognosis of Dukes’ C colorectal cancer. Anticancer Res 2008;28(3B): 1933–41.

7. Kim JY, Kim NK, Sohn SK, Kim YW, Kim KJ, Hur H, et al. Prognostic value of postoperative CEA clearance in rectal cancer patients with high preoperative CEA levels. Ann Surg Oncol 2009;16:2771–8.




8. Abe S, Kawai K, Ishihara S, Nozawa H, Hata K, Kiyomatsu T, et al. Prognostic impact of carcinoembryonic antigen and carbohydrate antigen 19-9 in stage IV colorectal cancer patients after R0 resection. J Surg Res 2016;205:384–92.


9. Probst CP, Becerra AZ, Aquina CT, Tejani MA, Hensley BJ, González MG, et al. Watch and Wait?--elevated pretreatment CEA Is associated with decreased pathological complete response in rectal cancer. J Gastrointest Surg 2016;20:43–52.



10. Mokhles S, Macbeth F, Farewell V, Fiorentino F, Williams NR, Younes RN, et al. Meta-analysis of colorectal cancer follow-up after potentially curative resection. Br J Surg 2016;103:1259–68.



11. Jeffery M, Hickey BE, Hider PN, See AM. Follow-up strategies for patients treated for non-metastatic colorectal cancer. Cochrane Database Syst Rev 2016;Nov 24 11:CD002200.


12. Treasure T, Monson K, Fiorentino F, Russell C. The CEA Second-Look Trial: a randomised controlled trial of carcinoembryonic antigen prompted reoperation for recurrent colorectal cancer. BMJ Open 2014;4:e004385.



13. Wille-Jørgensen P, Laurberg S, Påhlman L, Carriquiry L, Lundqvist N, Smedh K, et al. An interim analysis of recruitment to the COLOFOL trial. Colorectal Dis 2009;11:756–8.


14. Lepage C, Phelip JM, Cany L, Faroux R, Manfredi S, Ain JF, et al. Effect of 5 years of imaging and CEA follow-up to detect recurrence of colorectal cancer: The FFCD PRODIGE 13 randomised phase III trial. Dig Liver Dis 2015;47:529–31.


15. Primrose JN, Perera R, Gray A, Rose P, Fuller A, Corkhill A, et al. Effect of 3 to 5 years of scheduled CEA and CT follow-up to detect recurrence of colorectal cancer: the FACS randomized clinical trial. JAMA 2014;311:263–70.


16. Verberne CJ, Zhan Z, van den Heuvel E, Grossmann I, Doornbos PM, Havenga K, et al. Intensified follow-up in colorectal cancer patients using frequent Carcino-Embryonic Antigen (CEA) measurements and CEA-triggered imaging: Results of the randomized “CEAwatch” trial. Eur J Surg Oncol 2015;41:1188–96.


17. Verberne CJ, Wiggers T, Grossmann I, de Bock GH, Vermeulen KM. Cost-effectiveness of a carcinoembryonic antigen (CEA) based follow-up programme for colorectal cancer (the CEA Watch trial). Colorectal Dis 2016;18:O91–6.


18. Rosati G, Ambrosini G, Barni S, Andreoni B, Corradini G, Luchena G, et al. A randomized trial of intensive versus minimal surveillance of patients with resected Dukes B2-C colorectal carcinoma. Ann Oncol 2016;27:274–80.



19. Makela JT, Laitinen SO, Kairaluoma MI. Five-year follow-up after radical surgery for colorectal cancer. Results of a prospective randomized trial. Arch Surg 1995;130:1062–7.


20. Ohlsson B, Breland U, Ekberg H, Graffner H, Tranberg KG. Follow-up after curative surgery for colorectal carcinoma. Randomized comparison with no follow-up. Dis Colon Rectum 1995;38:619–26.


21. Pietra N, Sarli L, Costi R, Ouchemi C, Grattarola M, Peracchia A. Role of follow-up in management of local recurrences of colorectal cancer: a prospective, randomized study. Dis Colon Rectum 1998;41:1127–33.


22. Shinkins B, Nicholson BD, Primrose J, Perera R, James T, Pugh S, et al. The diagnostic accuracy of a single CEA blood test in detecting colorectal cancer recurrence: Results from the FACS trial. PLoS One 2017;12:e0171810.



23. Verberne CJ, Zhan Z, van den Heuvel ER, Oppers F, de Jong AM, Grossmann I, et al. Survival analysis of the CEAwatch multicentre clustered randomized trial. Br J Surg 2017;104:1069–77.


24. Nicholson BD, Shinkins B, Pathiraja I, Roberts NW, James TJ, Mallett S, et al. Blood CEA levels for detecting recurrent colorectal cancer. In: Cochrane Collaboration. Cochrane Database of Systematic Reviews. Chichester (UK): John Wiley & Sons; 2015.
25. Saito G, Sadahiro S, Kamata H, Miyakita H, Okada K, Tanaka A, et al. Monitoring of serum carcinoembryonic antigen levels after curative resection of colon cancer: cutoff values determined according to preoperative levels enhance the diagnostic accuracy for recurrence. Oncology 2017;92:276–82.



26. Huang SC, Lin JK, Lin TC, Chen WS, Yang SH, Wang HS, et al. Concordance of carcinoembryonic antigen ratio and response evaluation criteria in solid tumors as prognostic surrogate indicators of metastatic colorectal cancer patients treated with chemotherapy. Ann Surg Oncol 2015;22:2262–8.



27. Michl M, Koch J, Laubender RP, Modest DP, Giessen C, Schulz Ch, et al. Tumor markers CEA and CA 19-9 correlate with radiological imaging in metastatic colorectal cancer patients receiving first-line chemotherapy. Tumour Biol 2014;35:10121–7.



28. Michl M, Stintzing S, Fischer von Weikersthal L, Decker T, Kiani A, Vehling-Kaiser U, et al. CEA response is associated with tumor response and survival in patients with KRAS exon 2 wild-type and extended RAS wild-type metastatic colorectal cancer receiving first-line FOLFIRI plus cetuximab or bevacizumab (FIRE-3 trial). Ann Oncol 2016;27:1565–72.



29. Heinemann V, von Weikersthal LF, Decker T, Kiani A, VehlingKaiser U, Al-Batran SE, et al. FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer (FIRE-3): a randomised, openlabel, phase 3 trial. Lancet Oncol 2014;15:1065–75.


30. Saito G, Sadahiro S, Okada K, Tanaka A, Suzuki T, Kamijo A. Relation between carcinoembryonic antigen levels in colon cancer tissue and serum carcinoembryonic antigen levels at initial surgery and recurrence. Oncology 2016;91:85–9.


31. Gray-Owen SD, Blumberg RS. CEACAM1: contact-dependent control of immunity. Nat Rev Immunol 2006;6:433–46.



32. Lee JH, Hong SP, Jeon TJ, Kang GH, Choi WC, Jeon SM, et al. Should a colonoscopy be recommended for healthy individuals with increased carcinoembryonic antigen levels? A case-control study. Dig Dis Sci 2011;56:2396–403.



33. Nielsen HJ, Brünner N, Jorgensen LN, Olsen J, Rahr HB, Thygesen K, et al. Plasma TIMP-1 and CEA in detection of primary colorectal cancer: a prospective, population based study of 4509 high-risk individuals. Scand J Gastroenterol 2011;46:60–9.


34. Wen YH, Chang PY, Hsu CM, Wang HY, Chiu CT, Lu JJ. Cancer screening through a multi-analyte serum biomarker panel during health check-up examinations: Results from a 12-year experience. Clin Chim Acta 2015;450:273–6.


35. Wild N, Andres H, Rollinger W, Krause F, Dilba P, Tacke M, et al. A combination of serum markers for the early detection of colorectal cancer. Clin Cancer Res 2010;16:6111–21.


36. Hammarström S. The carcinoembryonic antigen (CEA) family: structures, suggested functions and expression in normal and malignant tissues. Semin Cancer Biol 1999;9:67–81.


37. Benchimol S, Fuks A, Jothy S, Beauchemin N, Shirota K, Stanners CP. Carcinoembryonic antigen, a human tumor marker, functions as an intercellular adhesion molecule. Cell 1989;57:327–34.


38. Yap BS, Yap HY, Fritsche HA, Blumenschein G, Bodey GP. CSF carcinoembryonic antigen in meningeal carcinomatosis from breast cancer. JAMA 1980;244:1601–3.


39. Herbeth B, Bagrel A. A study of factors influencing plasma CEA levels in an unselected population. Oncodev Biol Med 1980;1:191–8.

40. Beaudonnet A, Gounon G, Pichot J, Revenant MC. Sex- and agerelated influences on carcinoembryonic antigen in blood. Clin Chem 1981;27:771.


41. van Nagell JR Jr, Donaldson ES, Hanson MB, Gay EC, Pavlik EJ. Biochemical markers in the plasma and tumors of patients with gynecologic malignancies. Cancer 1981;48(2 Suppl): 495–503.


42. Goldenberg DM, Neville AM, Carter AC, Go VL, Holyoke ED, Isselbacher KJ, et al. CEA (carcinoembryonic antigen): its role as a marker in the management of cancer. J Cancer Res Clin Oncol 1981;101:239–42.



43. Booth SN, Jamieson GC, King JP, Leonard J, Oates GD, Dykes PW. Carcinoembryonic antigen in management of colorectal carcinoma. Br Med J 1974;4:183–7.



44. Arnaud JP, Koehl C, Adloff M. Carcinoembryonic antigen (CEA) in diagnosis and prognosis of colorectal carcinoma. Dis Colon Rectum 1980;23:141–4.


45. Szymendera JJ, Nowacki MP, Szawłowski AW, Kamińska JA. Predictive value of plasma CEA levels: preoperative prognosis and postoperative monitoring of patients with colorectal carcinoma. Dis Colon Rectum 1982;25:46–52.


46. Wanebo HJ, Rao B, Pinsky CM, Hoffman RG, Stearns M, Schwartz MK, et al. Preoperative carcinoembryonic antigen level as a prognostic indicator in colorectal cancer. N Engl J Med 1978;299:448–51.


47. Armitage NC, Davidson A, Tsikos D, Wood CB. A study of the reliability of carcinoembryonic antigen blood levels in following the course of colorectal cancer. Clin Oncol 1984;10:141–7.

48. Midiri G, Amanti C, Benedetti M, Campisi C, Santeusanio G, Castagna G, et al. CEA tissue staining in colorectal cancer patients. A way to improve the usefulness of serial serum CEA evaluation. Cancer 1985;55:2624–9.


49. Duffy MJ. Carcinoembryonic antigen as a marker for colorectal cancer: is it clinically useful? Clin Chem 2001;47:624–30.


50. Reiter W, Stieber P, Reuter C, Nagel D, Lau-Werner U, Lamerz R. Multivariate analysis of the prognostic value of CEA and CA 19-9 serum levels in colorectal cancer. Anticancer Res 2000;20(2D): 5195–8.

51. Diez M, Pollan M, Müguerza JM, Gaspar MJ, Duce AM, Alvarez MJ, et al. Time-dependency of the prognostic effect of carcinoembryonic antigen and p53 protein in colorectal adenocarcinoma. Cancer 2000;88:35–41.


52. Nissan A, Stojadinovic A, Shia J, Hoos A, Guillem JG, Klimstra D, et al. Predictors of recurrence in patients with T2 and early T3, N0 adenocarcinoma of the rectum treated by surgery alone. J Clin Oncol 2006;24:4078–84.


53. Takagawa R, Fujii S, Ohta M, Nagano Y, Kunisaki C, Yamagishi S, et al. Preoperative serum carcinoembryonic antigen level as a predictive factor of recurrence after curative resection of colorectal cancer. Ann Surg Oncol 2008;15:3433–9.



54. Kanellos I, Zacharakis E, Kanellos D, Pramateftakis MG, Tsahalis T, Altsitsiadis E, et al. Prognostic significance of CEA levels and detection of CEA mRNA in draining venous blood in patients with colorectal cancer. J Surg Oncol 2006;94:3–8.


55. Lin JK, Lin CC, Yang SH, Wang HS, Jiang JK, Lan YT, et al. Early postoperative CEA level is a better prognostic indicator than is preoperative CEA level in predicting prognosis of patients with curable colorectal cancer. Int J Colorectal Dis 2011;26:1135–41.



56. Lee WS, Baek JH, Kim KK, Park YH. The prognostic significant of percentage drop in serum CEA post curative resection for colon cancer. Surg Oncol 2012;21:45–51.


57. Oussoultzoglou E, Rosso E, Fuchshuber P, Stefanescu V, Diop B, Giraudo G, et al. Perioperative carcinoembryonic antigen measurements to predict curability after liver resection for colorectal metastases: a prospective study. Arch Surg 2008;143:1150–8.


58. Suarez J, Vera R, Balen E, Gomez M, Arias F, Lera JM, et al. Pathologic response assessed by Mandard grade is a better prognostic factor than down staging for disease-free survival after preoperative radiochemotherapy for advanced rectal cancer. Colorectal Dis 2008;10:563–8.


59. Park YA, Sohn SK, Seong J, Baik SH, Lee KY, Kim NK, et al. Serum CEA as a predictor for the response to preoperative chemoradiation in rectal cancer. J Surg Oncol 2006;93:145–50.


60. Perez RO, São Julião GP, Habr-Gama A, Kiss D, Proscurshim I, Campos FG, et al. The role of carcinoembriogenic antigen in predicting response and survival to neoadjuvant chemoradiotherapy for distal rectal cancer. Dis Colon Rectum 2009;52:1137–43.


61. 1997 update of recommendations for the use of tumor markers in breast and colorectal cancer. Adopted on November 7, 1997 by the American Society of Clinical Oncology. J Clin Oncol 1998;16:793–5.


62. Meyerhardt JA, Mangu PB, Flynn PJ, Korde L, Loprinzi CL, Minsky BD, et al. Follow-up care, surveillance protocol, and secondary prevention measures for survivors of colorectal cancer: American Society of Clinical Oncology clinical practice guideline endorsement. J Clin Oncol 2013;31:4465–70.


63. National Institute of Clinical Excellence (NICE). Colorectal cancer: diagnosis and management [Internet]. London, National Institute of Clinical Excellence; 2011 [2018 Nov 1]. Available from: https://www.nice.org.uk/guidance/cg131
64. Yamamoto M, Maehara Y, Sakaguchi Y, Mine H, Yamanaka T, Korenaga D, et al. Distributions in CEA doubling time differ in patients with recurrent colorectal carcinomas. Hepatogastroenterology 2004;51:147–51.

65. Wood CB, Ratcliffe JG, Burt RW, Malcolm AJ, Blumgart LH. The clinical significance of the pattern of elevated serum carcinoembryonic antigen (CEA) levels in recurrent colorectal cancer. Br J Surg 1980;67:46–8.


66. The Royal College of Phycisians; The The Royal College of Radiologists. Evidence-based Indications for the use of PET-CT in the UK 2013. London: Royal College of Phycisians; 2013.
67. Fletcher JW, Djulbegovic B, Soares HP, Siegel BA, Lowe VJ, Lyman GH, et al. Recommendations on the use of 18F-FDG PET in oncology. J Nucl Med 2008;49:480–508.


68. Stelzner S, Hellmich G, Koch R, Ludwig K. Factors predicting survival in stage IV colorectal carcinoma patients after palliative treatment: a multivariate analysis. J Surg Oncol 2005;89:211–7.


69. Sun LC, Chu KS, Cheng SC, Lu CY, Kuo CH, Hsieh JS, et al. Preoperative serum carcinoembryonic antigen, albumin and age are supplementary to UICC staging systems in predicting survival for colorectal cancer patients undergoing surgical treatment. BMC Cancer 2009;9:288.




70. Thirunavukarasu P, Talati C, Munjal S, Attwood K, Edge SB, Francescutti V. Effect of Incorporation of pretreatment serum carcinoembryonic antigen levels into AJCC staging for colon cancer on 5-year survival. JAMA Surg 2015;150:747.


-
METRICS

- Related articles in ACP
-
Serum Carcinoembryonic Antigen for Recurrence in Colorectal Cancer Patients2013 August;29(4)







