- Search
| Ann Coloproctol > Volume 39(1); 2023 > Article |
|
Abstract
Purpose
Methods
Results
ACKNOWLEDGMENTS
Fig. 2.
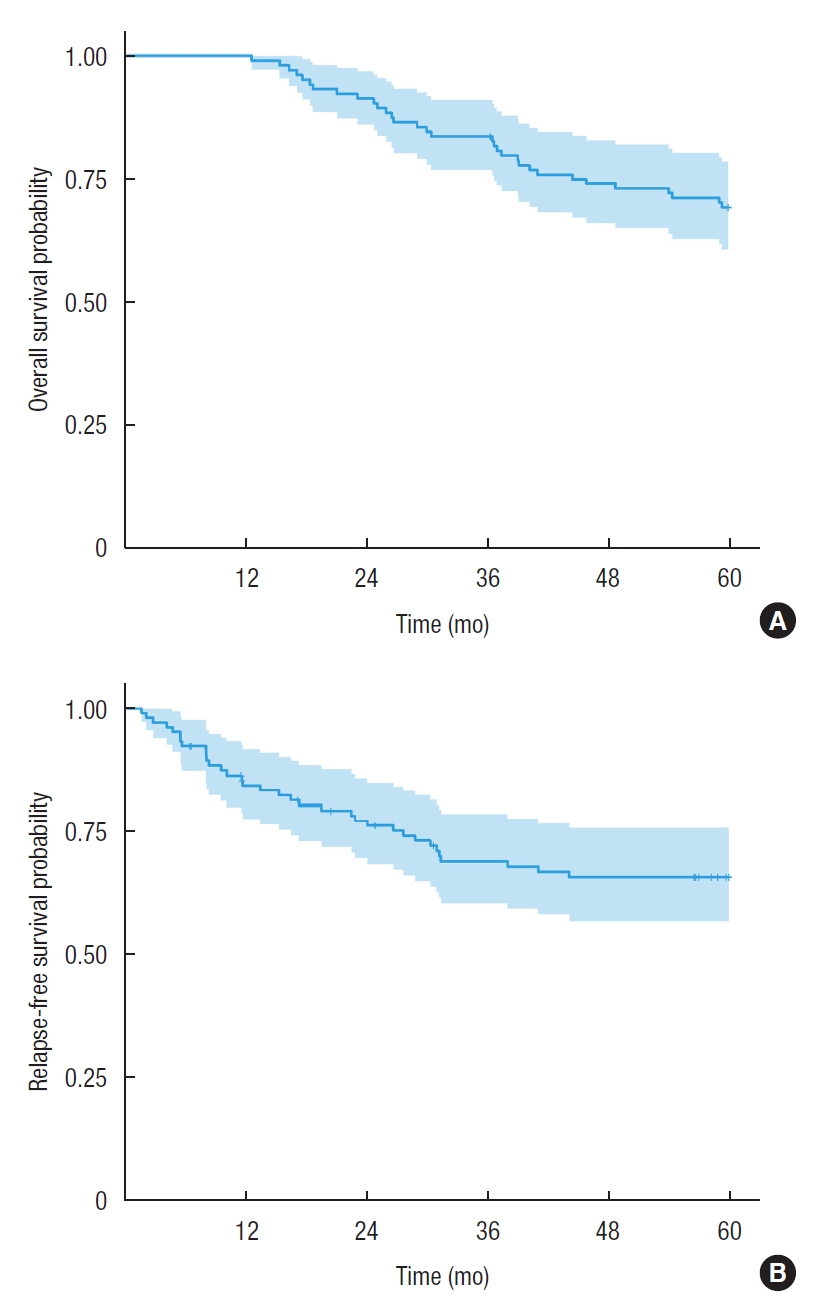
Fig. 3.
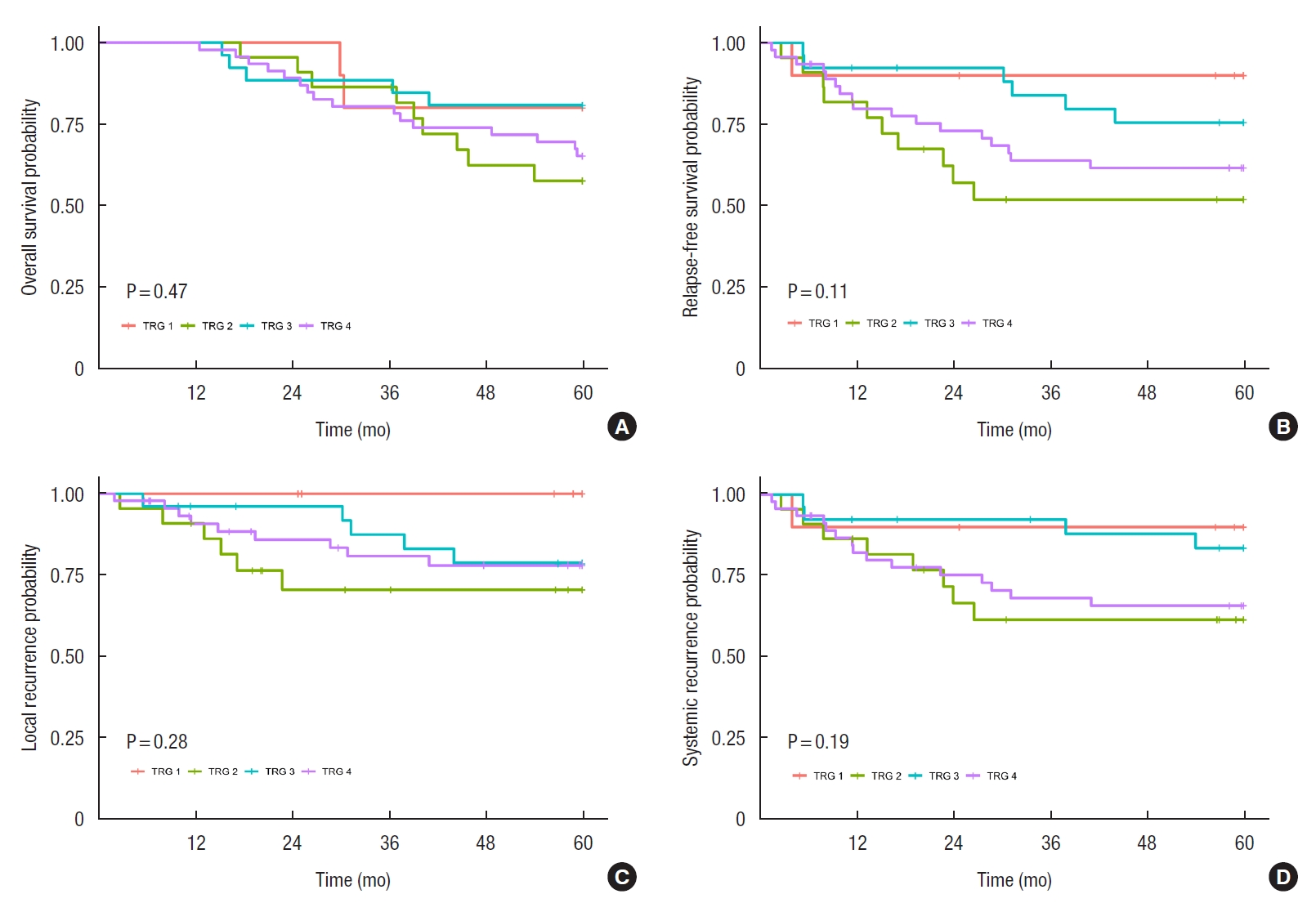
Fig. 4.
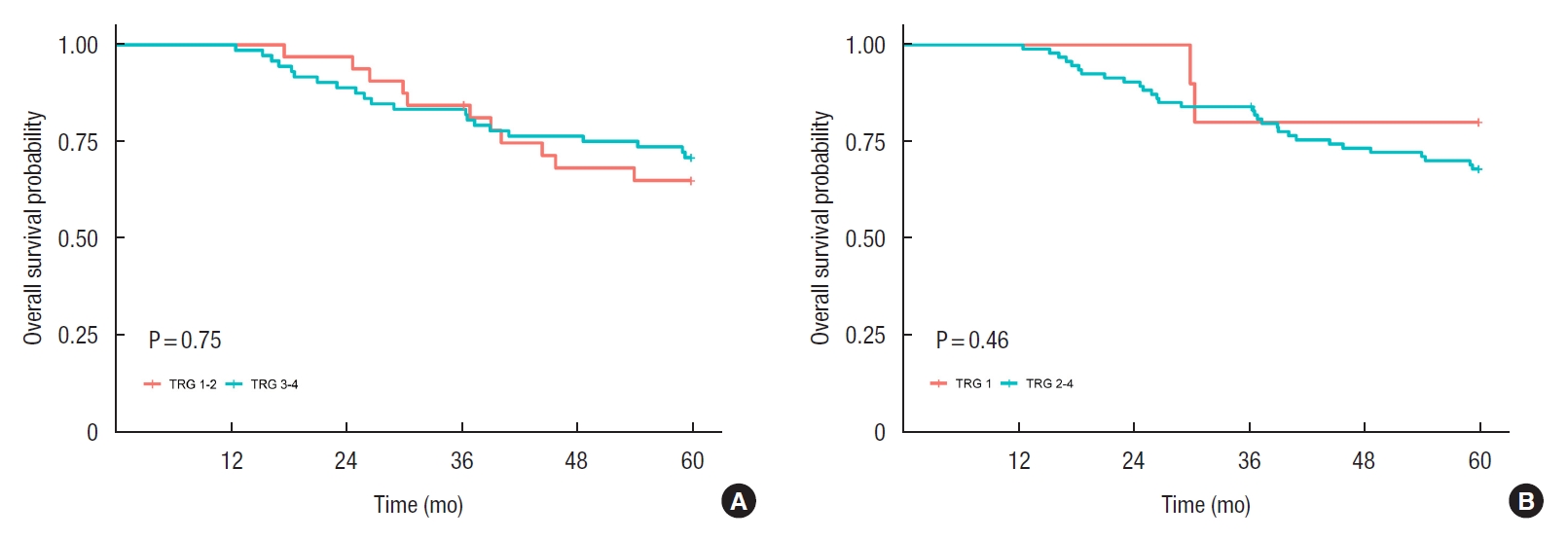
Fig. 5.

Table 1.
Table 2.
| Variable | TRG 1 (n = 10) | TRG 2 (n = 22) | TRG 3 (n = 26) | TRG 4 + 5a (n = 46) | P-value |
|---|---|---|---|---|---|
| Age (yr) | 56.0 ± 13.3 | 56.8 ± 14.3 | 58.6 ± 15.2 | 59.1 ± 11.6 | 0.86 |
| Male sex | 5 (50.0) | 10 (45.5) | 15 (57.7) | 31 (67.4) | 0.34 |
| cT stage | 0.22 | ||||
| cT3 | 7 (70.0) | 16 (72.7) | 18 (69.2) | 40 (87.0) | |
| cT4 | 3 (30.0) | 6 (27.3) | 8 (30.8) | 6 (13.0) | |
| cN stage | 0.83 | ||||
| cN0 | 3 (30.0) | 6 (27.3) | 11 (42.3) | 12 (26.1) | |
| cN1 | 7 (70.0) | 14 (63.6) | 13 (50.0) | 30 (65.2) | |
| cN2 | 0 (0) | 2 (9.1) | 2 (7.7) | 4 (8.7) | |
| Preoperative staging | 0.53 | ||||
| cT3–4N0M0 | 3 (30.0) | 6 (27.3) | 11 (42.3) | 12 (26.1) | |
| cT1–4N1–2M0 | 7 (70.0) | 16 (72.7) | 15 (57.7) | 34 (73.9) | |
| Tumor location | 0.65 | ||||
| Lower | 6 (60.0) | 14 (63.6) | 13 (50.0) | 22 (47.8) | |
| Middle | 3 (30.0) | 8 (36.4) | 11 (42.3) | 18 (39.1) | |
| Upper | 1 (10.0) | 0 (0) | 2 (7.7) | 6 (13.0) | |
| CRT CEA (ng/mL) | |||||
| Preoperative | 4.0 (2.9–6.7) | 5.1 (2.6–14.8) | 5.9 (2.9–18.5) | 6.1 (3.1–10.7) | 0.86 |
| Postoperative | 2.4 (1.6–3.4) | 3.6 (2.4–3.8) | 2.8 (1.6–3.8) | 2.8 (1.8–5.5) | 0.81 |
| CRT regimen | 0.61 | ||||
| 5FU/leucovorin | 8 (80.0) | 19 (86.4) | 20 (76.9) | 32 (69.6) | |
| Capecitabine | 2 (20.0) | 3 (13.6) | 6 (23.1) | 14 (30.4) | |
| CRM positivity | 0 (0) | 0 (0) | 1 (3.8) | 5 (10.9) | 0.27 |
| Tumor perforation | 0 (0) | 5 (22.7) | 1 (3.8) | 3 (6.5) | 0.17 |
| Tumor differentiation | 0.01* | ||||
| Well | 10 (100) | 10 (45.5) | 16 (61.5) | 36 (78.3) | |
| Moderate | 0 (0) | 7 (31.8) | 9 (34.6) | 8 (17.4) | |
| Poor | 0 (0) | 5 (22.7) | 1 (3.8) | 2 (4.3) | |
| LNs positive | 0 (0–0) | 0 (0–1.8) | 0 (0–1.8) | 0 (0–1.0) | 0.13 |
| LVI | 0 (0) | 2 (9.1) | 7 (26.9) | 20 (43.5) | 0.04* |
| PNI | 0 (0) | 2 (9.1) | 4 (15.4) | 8 (17.4) | 0.59 |
| Surgical interval (wk) | 16.0 (9.2–17.0) | 9.0 (8.0–11.5) | 10.5 (7.2–12.0) | 12.0 (8.2–15.0) | 0.03* |
| 5-Year OS | 8 (80.0) | 12 (54.5) | 21 (80.8) | 31 (67.4) | 0.22 |
| 5-Year RFS | 8 (80.0) | 9 (40.9) | 18 (69.2) | 28 (60.9) | 0.11 |
Values are presented as mean±standard deviation, number (%), or median (interquartile range).
TRG, tumor regression grade; CRT, chemoradiation; CEA, carcinoembryonic antigen; 5FU, 5-fluorouracil; CRM, circumferential resection margin; LN, lymph node; LVI, lymphovascular invasion; PNI, perineural invasion; OS, overall survival; RFS, relapse-free survival.
Table 3.
| Variable | Good response (n = 32) | Poor response (n = 72) | P-value |
|---|---|---|---|
| Age (yr) | 56.6 ± 13.8 | 58.9 ± 12.9 | 0.40 |
| Male sex | 15 (46.9) | 46 (63.9) | 0.16 |
| cT stage | 0.47 | ||
| cT3 | 23 (71.9) | 58 (80.6) | |
| cT4 | 9 (28.1) | 14 (19.4) | |
| cN stage | 0.84 | ||
| cN0 | 9 (28.1) | 23 (31.9) | |
| cN1 | 21 (65.6) | 43 (59.7) | |
| cN2 | 2 (6.2) | 6 (8.3) | |
| Preoperative staging | 0.87 | ||
| cT3–4N0M0 | 9 (28.1) | 23 (31.9) | |
| cT1–4N1–2M0 | 23 (71.9) | 49 (68.1) | |
| Tumor location | 0.27 | ||
| Lower | 20 (62.5) | 35 (48.6) | |
| Middle | 11 (34.4) | 29 (40.3) | |
| Upper | 1 (3.1) | 8 (11.1) | |
| CRT CEA (ng/mL) | |||
| Preoperative | 4.4 (2.6–14.4) | 6 (2.9–16.7) | 0.52 |
| Postoperative | 3.4 (1.8–3.8) | 2.8 (1.8–4.7) | 0.95 |
| Preoperative CRT regimen | 0.22 | ||
| 5FU/leucovorin | 27 (84.4) | 52 (72.2) | |
| Capecitabine | 5 (15.6) | 20 (27.8) | |
| CRM positive | 0 (0) | 6 (8.3) | 0.17 |
| Tumor perforation | 5 (15.6) | 4 (5.6) | 0.13 |
| Tumor differentiation | 0.13 | ||
| Well | 20 (62.5) | 52 (72.2) | |
| Moderate | 7 (21.9) | 17 (23.6) | |
| Poor | 5 (15.6) | 3 (4.2) | |
| LNs positive | 0 (0–1.0) | 0 (0–1.2) | 0.44 |
| LVI | 2 (6.3) | 27 (37.5) | 0.002* |
| PNI | 2 (6.3) | 12 (16.7) | 0.22 |
| Surgical interval (wk) | 9.0 (8.0–15.2) | 11.0 (8.0–14.0) | 0.67 |
| 5-Year OS | 20 (62.5) | 52 (72.2) | 0.45 |
| 5-Year RFS | 17 (53.1) | 46 (63.9) | 0.41 |
Values are presented as mean±standard deviation, number (%), or median (interquartile range).
TRG, tumor regression grade; CRT, chemoradiation; CEA, carcinoembryonic antigen; 5FU, 5-fluorouracil; CRM, circumferential resection margin; LN, lymph nodes; LVI, lymphovascular invasion; PNI, perineural invasion; OS, overall survival; RFS, relapse-free survival.
Table 4.
| Variable | 5-Year survival (n = 72) | 5-Year nonsurvival (n = 32) | Total (n = 104) | P-value |
|---|---|---|---|---|
| Sex | ||||
| Male | 44 (61.1) | 17 (53.1) | 61 (58.7) | 0.58 |
| Female | 28 (38.9) | 15 (46.9) | 43 (41.3) | |
| Age (yr) | 58.6 ± 13.4 | 57.2 ± 12.7 | 58.2 ± 13.2 | 0.61 |
| CCI | 4 (3–4.2) | 4 (3–4) | 4 (3–4) | 0.82 |
| Clinical staging | 0.45 | |||
| cT3–4N0M0 | 20 (27.8) | 12 (37.5) | 32 (30.8) | |
| cT1–4N1–2M0 | 52 (72.2) | 20 (62.5) | 72 (69.2) | |
| Tumor location | 0.70 | |||
| Lower | 39 (54.2) | 12 (37.5) | 55 (52.9) | |
| Middle | 26 (36.1) | 14 (43.8) | 40 (38.5) | |
| Upper | 7 (9.7) | 2 (6.3) | 9 (8.7) | |
| Preoperative CEA (µg/mL) | 5.5 (2.9–11) | 6.3 (2.6–20) | 5.7 (2.8–14.8) | 0.48 |
| Surgical interval after nCRT (wk) | 10 (8–13.2) | 11 (8–15) | 10 (8–15) | 0.69 |
| Operative time (min) | 384 (298–502) | 455 (349–540) | 405 (321–510) | 0.08 |
| Estimate blood loss (mL) | 200 (100–400) | 250 (150–680) | 200 (100–450) | 0.15 |
| Operation | 0.02* | |||
| APR | 19 (26.4) | 17 (53.1) | 36 (34.6) | |
| LAR | 53 (73.6) | 15 (46.9) | 68 (65.4) | |
| Intraoperative tumor perforation | < 0.001* | |||
| No | 71 (98.6) | 24 (75.0) | 95 (91.3) | |
| Yes | 1 (1.4) | 8 (25.0) | 9 (8.7) | |
| Circumferential margin | 0.07 | |||
| Positive | 2 (2.8) | 4 (12.5) | 6 (5.8) | |
| Negative | 70 (97.2) | 28 (87.5) | 98 (94.2) | |
| Tumor size (cm) | 3.0 (1.5–4.0) | 2.5 (0.7–4.3) | 3.0 (1.0–4.1) | 0.91 |
| Tumor differentiation | 0.01* | |||
| Well | 54 (75.0) | 18 (56.3) | 72 (69.2) | |
| Moderate | 16 (2.2) | 8 (25.0) | 24 (23.1) | |
| Poor | 2 (2.8) | 6 (18.8) | 8 (7.7) | |
| LVI | 0.79 | |||
| Absence | 19 (26.4) | 10 (31.2) | 29 (27.9) | |
| Presence | 53 (73.6) | 22 (68.8) | 75 (72.1) | |
| PNI | 0.03* | |||
| Absence | 66 (91.7) | 24 (75.0) | 90 (86.5) | |
| Presence | 6 (8.3) | 8 (25.0) | 14 (13.5) | |
| TRG | 0.22 | |||
| TRG 1 | 8 (11.1) | 2 (6.2) | 10 (9.6) | |
| TRG 2 | 12 (16.7) | 10 (31.2) | 22 (21.2) | |
| TRG 3 | 21 (29.2) | 5 (15.6) | 26 (25.0) | |
| TRG 4 | 31 (43.1) | 15 (46.9) | 46 (44.2) | |
| Pathological T stage | 0.61 | |||
| ypT0 | 8 (11.1) | 2 (6.2) | 10 (9.6) | |
| ypT1 | 4 (5.6) | 3 (9.4) | 7 (6.7) | |
| ypT2 | 14 (19.4) | 5 (15.6) | 19 (18.3) | |
| ypT3 | 38 (52.8) | 16 (50) | 54 (51.9) | |
| ypT4 | 8 (11.1) | 6 (18.8) | 14 (13.5) | |
| Pathological N stage | 0.19 | |||
| ypN0 | 51 (70.8) | 17 (53.1) | 68 (65.4) | |
| ypN1 | 14 (19.4) | 11 (34.4) | 25 (24.0) | |
| ypN2 | 7 (9.7) | 4 (12.5) | 11 (10.6) | |
| Pathological staging | 0.34 | |||
| ypT0N0M0 | 8 (11.1) | 2 (6.3) | 10 (9.6) | |
| ypT1–2N0M0 | 14 (19.4) | 4 (12.5) | 18 (17.3) | |
| ypT3–4N0M0 | 29 (40.3) | 11 (34.4) | 40 (38.5) | |
| ypT1–4N1–2M0 | 21 (29.2) | 15 (46.9) | 36 (34.6) | |
| Pathological complete response | 0.72 | |||
| Yes | 8 (11.1) | 2 (6.2) | 10 (9.6) | |
| No | 64 (88.9) | 30 (93.8) | 94 (90.4) | |
| Postoperative complication (CD grade) | 0.48 | |||
| I | 6 (35.3) | 3 (37.5) | 9 (36.0) | |
| II | 4 (23.5) | 4 (50.0) | 8 (32.0) | |
| IIIA | 3 (17.6) | 0 (0) | 3 (12.0) | |
| IIIB | 4 (23.5) | 1 (12.5) | 5 (20.0) | |
| Local recurrence | < 0.001* | |||
| Yes | 6 (8.3) | 14 (43.8) | 20 (19.2) | |
| No | 66 (91.7) | 18 (56.2) | 84 (80.8) | |
| Systemic recurrence | < 0.001* | |||
| Yes | 5 (6.9) | 23 (71.9) | 28 (26.9) | |
| No | 67 (93.1) | 9 (28.1) | 76 (73.1) |
Values are presented as number (%), mean±standard deviation, or median (interquartile range).
OS, overall survival; CCI, Charlson comorbidity index; CEA, carcinoembryonic antigen; nCRT, chemoradiation; APR, abdominoperineal resection; LAR, low anterior resection; LVI, lymphovascular invasion; PNI, perineural invasion; TRG, tumor regression grade; CD, Clavien-Dindo classification.









