- Search
|
|
Abstract
Purpose
The aim of this study was to assess oncological outcomes of postoperative radiotherapy plus chemotherapy (CRT) versus chemotherapy alone (CTx) in stage II or III upper rectal cancer patients who underwent curative surgery.
Methods
We retrospectively reviewed 263 consecutive patients with pathologic stage II or III upper rectal cancer who underwent primary curative resection with postoperative CRT or CTx from January 2008 to December 2014 at Chonnam National University Hwasun Hospital. Multivariate and propensity score matching analyses were used to reduce selection bias.
Results
Median follow-up was 48.1 months for the entire cohort and 53.5 months for the matched cohort. In subgroup analysis of the propensity score matched cohort, the 3-year local recurrence-free survival was 94.1% (95% confidence interval [CI], 87.8%–100%) in the CRT group and 90.1% (95% CI, 82.8%–97.9%) in the CTx group (P = 0.370). No significant difference in disease-free survival was observed according to treatment type. On multivariate analysis, circumferential resection margin involvement (hazard ratio [HR], 2.386; 95% CI, 1.190–7.599; P = 0.032), N stage (HR, 6.262; 95% CI, 1.843–21.278, P = 0.003), and T stage (HR, 5.896, 95% CI, 1.298–6.780, P = 0.021) were identified as independent risk factors for local recurrence of tumors of the upper rectum.
Rectal cancer accounts for about one-third of colorectal cancers and is a major cause of cancer death worldwide [1]. According to analysis of data in the Korea Central Cancer Registry and Korean Statistical Information Service, the incidence of rectal cancer in the Republic of Korea (hereafter Korea) is continuing to increase [2]. The treatment strategy for rectal cancer has changed dramatically over the past two decades, especially that for lower rectal cancer. Preoperative chemoradiotherapy (CRT) is currently used as a standard treatment for locally advanced rectal cancer [3, 4] because preoperative CRT is associated with higher sphincter saving and improved local control, despite a large amount of pelvic irradiation.
However, for treatment of upper rectal cancer, local control and preservation of anal sphincter are not as important as in lower rectal cancer. Furthermore, some authors have suggested that tumors of the upper rectum behave like sigmoid colon cancers and have similar oncological outcomes in terms of local recurrence and prognosis [5-7]. Recent advances in the accuracy of preoperative magnetic resonance imaging (MRI) allow more accurate assessment of the circumferential resection margin (CRM) and increase the potential for achieving a negative surgical margin [8]. As a result, routine administration of preoperative CRT has been questioned, and many surgeons prefer to perform surgery without radiotherapy when treating upper rectal cancers.
However, the role of adjuvant treatment after surgery for upper rectal cancer has not been evaluated. Therefore, we aimed to assess oncological outcomes according to adjuvant treatment (postoperative CRT versus chemotherapy alone [CTx]).
We retrospectively reviewed 263 patients with pathologic stage II or III upper rectal cancer who underwent low anterior resection from January 2008 to December 2014 at Chonnam National University Hwasun Hospital. This study received Institutional Review Board approval (IRB number: TMP-2018-119), which waived the requirement for informed consent in this retrospective study. Eligibility criteria were (1) pathologic stage II/III upper rectal cancer (lower margin of tumor between 10 and 15 cm from the anal verge by rigid sigmoidoscopy) and (2) underwent primary rectal cancer resection with curative intent. Exclusion criteria were (1) multiple primary cancers, (2) a previous history of pelvic radiotherapy, and (3) refusal of any adjuvant treatment. Patients without recurrence who did not complete at least 6 months of followup (n = 7) were also excluded. Ultimately, we included and analyzed a total of 263 patients.
All patients were assessed preoperatively, including detailed physical examination, laboratory testing, rigid sigmoidoscopy, colonoscopy, abdominopelvic and chest computed tomography, rectal MRI, and measurement of serum carcinoembryonic antigen (CEA) level. Positron emission tomography was selectively used when distant metastasis was suspected. Rectal cancer stage was determined in accordance with the seventh Union for International Cancer Control TNM staging system. CRM was considered positive if tumor was identified within 1 mm of the surgical resection margin.
Experienced surgeons performed radical oncological surgery, including tumor-specific mesorectal excision, high vascular ligation of the inferior mesenteric artery and vein, and en bloc resection of adjacent involved organs. As per institutional treatment policy, most upper rectal cancer patients underwent surgery if CRM involvement was strongly suspected on preoperative MRI. Postoperative CTx and CRT regimens have been described previously [9]. In short, CRT comprised 45–5,040 cGy in 25–28 fractions delivered to the pelvis, and concurrent CTx consisting of 5-fluorouracil (5-FU; 425 mg/m2/day) and leucovorin (LV) (20 mg/m2/day) was administered intravenously during the 1st and 5th weeks of radiotherapy. Four additional cycles of adjuvant CTx (5-FU; 425 mg/m2/day, 5 times weekly, every 4 weeks) were administered to the CRT group after completion of 4 weeks of CRT. Adjuvant CTx was recommended for all patients for 6 months and was initiated 4 weeks after surgery. CTx regimens were oral capecitabine (n = 20), 5-FU with LV (FL) (n = 119), and FL plus oxaliplatin (FOLFOX) (n = 22). Determination of whether to administer CTx or postoperative CRT as the adjuvant treatment was at the discretion of the surgeon.
Statistical significance of differences between groups was tested using the chi-square test or Fisher exact test for categorical variables and Student t-test for continuous variables. The Kaplan-Meier method was used to calculate cumulative recurrence rates, and the log-rank test was used to compare survival differences between treatment groups. To identify independent risk factors for local recurrence, multivariate analyses of survival were conducted using Cox proportional hazards models. Significant variables in univariate analysis (P < 0.1) were incorporated in the regression models. To reduce selection bias, propensity score matching was used. Case matching was performed using the propensity score generated by a logistic regression model based on age, sex, pT stage, pN stage, tumor differentiation, CEA level, and CRM involvement. Each patient was then matched one to one using a 0.2 caliper width. Patients who were found to be outside this caliper range were excluded, and unmatched patients were also excluded. All survival analyses were performed on all matchedpair patients. All P-values were 2-sided, with values less than 0.05 considered statistically significant. All analyses were performed using R software version 3.2.2 (R Foundation for Statistical Computing, Vienna, Austria).
A total of 263 patients who underwent curative intent surgery for advanced upper rectal cancer was included. Thirteen patients (10.3%) with pStage II and 89 patients (65.0%) with pStage III were treated by CRT. Clinicopathologic characteristics of the entire cohort (n = 263), as well as the propensity score-matched cohort (n = 122), are shown in Table 1. In the entire cohort, CRT group patients were younger (P = 0.032) and had higher N stage (P < 0.001) and more poorly differentiated tumors (P = 0.017). The proportion of tumors with perineural invasion, however, was higher in the CTx group than in the CRT group (P = 0.001).
After propensity score matching based on the 5 previously described specific criteria, 61 patients in the CRT group were matched with an equal number of subjects in the CTx group. After matching, there were no significant differences between the CRT group and CTx group in terms of clinicopathologic characteristics (Table 1).
Median follow-up was 48.1 months (interquartile range [IQR], 28.7–61.1 months) for the entire cohort, and 53.5 months (IQR, 53.5–69.9 months) for the matched cohort. During the follow-up period, 14 of the 263 patients (5.3%) developed local recurrence. Local recurrence rates in the CRT and CTx groups were 7.8% and 3.7%, respectively (P = 0.243). Upon diagnosis of local recurrence, 37.5% (3 of 8) of the CRT group and 50% (3 of 6) of the CTx group were found to also have distant metastasis. Table 2 lists the sites of local recurrence and distant metastases for each group. In the propensity score matched cohort, 3-year local recurrence-free survival was 94.1% (95% confidence interval [CI], 87.8%–100%) in the CRT group and 90.1% (95% CI, 82.8%–97.9%) in the CTx group (P = 0.370). No significant differences in disease-free survival (DFS) were observed according to treatment type. When patients were analyzed according to tumor stage, there were also no differences in local recurrence rate or distant metastasis (Table 3, Fig. 1).
Risk factors for local recurrence of upper rectal cancer in univariate analysis were T stage (P = 0.002), N stage (P < 0.001), CRM involvement (P = 0.028), and perineural invasion (P = 0.032). On multivariate analysis, CRM involvement (hazard ratio [HR], 2.386; 95% CI, 1.190–7.599; P = 0.032), N stage (HR, 6.262; 95% CI, 1.843–21.278, P = 0.003), and T stage (HR, 5.896, 95% CI, 1.298–6.780, P = 0.021) were identified as independent risk factors for local recurrence (Table 4).
Our data indicate that postoperative CRT did not improve local control or DFS compared with CTx after surgery for upper rectal cancer in patients with pathologic stage II or III disease. Although further studies are necessary to clarify the potential benefits of more intensive adjuvant CTx, we suggest that upper rectal cancer could be treated using similar treatment strategies as sigmoid colon cancer to avoid unnecessary postoperative irradiation, with a focus on controlling systemic disease.
Achieving an adequate resection margin, which is the most important factor related to local recurrence, is challenging in the upper rectum. Unlike the mid to lower rectum, which is constrained by a restrictive pelvis, the upper rectum does not have these limitations and is covered by the peritoneum anteriorly and laterally. Therefore, when tumors invade the bowel wall and eventually penetrate it, they are more likely to perforate the peritoneum. Tumors penetrating the peritoneum cause peritoneal carcinomatosis rather than specified regional local recurrence. In this situation, which is a more common pattern of disease progression in colon cancer, radiotherapy is not considered as a standard adjuvant treatment modality.
Few studies have described multidisciplinary treatment of upper rectal cancers. Rather, studies have shown that preoperative CRT improves local control with reduced toxicity compared with postoperative CRT [3, 4]. Even in this era, in which preoperative CRT is used as a standard treatment, the possibility of omission of radiotherapy is questioned and is being studied. In a Dutch mesorectal excision trial of the efficacy of preoperative CRT versus surgery alone, there was no significant improvement in local recurrence in upper rectal cancer (at 2 years of follow-up, 3.8% after surgery alone vs. 1.3% after preoperative CRT and surgery, P = 0.17) [4]. Similarly, a Swedish rectal cancer trial reported that the efficacy of preoperative CRT on local control was not significant in subgroup analysis [10]. Finally, in an Asian multi-institutional study, in which 2012 patients with stage II/III rectal and sigmoid cancer did not receive concurrent CRT, the 5-year local recurrence rates of sigmoid colon cancer, upper rectal cancer, and mid to lower rectal cancer were 2.5%, 3.5%, and 11.1%, respectively. The authors of that study concluded that routine use of preoperative CRT for locally advanced upper rectal cancer is unnecessary. Based on these results, if preoperative CRT is not useful for controlling local recurrence as expected, postoperative CRT may also be ineffective and lead only to toxicity [7].
In the current study, postoperative CRT was more frequently performed in patients with metastatic lymph nodes, suggesting that nodal status is an important consideration when determining treatment. Because lymph node metastasis is a strong independent risk factor for local recurrence and survival [11-16], we attempted to reduce the difference in nodal status by propensity score matching the groups. Before matching, the incidence of local recurrence of stage II and III was 0% and 10.2%, respectively. As shown in Table 1, nodal status was consistent even in the N subcategory. After matching, the 5-year local recurrence rates in the CRT group and CTx group were 4.9% and 9.8%, respectively (P = 0.489). Considering that about 80% of patients were node positive after matching, these results are comparable with previous studies of upper rectal cancer [5, 17, 18]. In 1998, Lopez-Kostner et al. [5] reported a 5-year local recurrence rate for upper rectal cancer of 4.7% compared to 12% for lower rectal cancer. Furthermore, Marinello et al. [17] reported that the 5-year actuarial local recurrence rate for upper rectal cancer was 4.9%, although only 5.6% of that patient group received preoperative CRT. Finally, it is highly desirable to select patients who will benefit from additional postoperative CRT. As stated above, we observed no local recurrence in stage II upper rectal cancer. Future research on this issue in the preoperative setting is required before it can be extrapolated to the postoperative environment.
There were several limitations to this study. First, because of the small cohort size and retrospective nature of the study, we could not address adequately the impact of postoperative CRT on local failure. Second, the chemotherapeutic agents used in the CTx group varied, and FOLFOX, which may be more effective at systemic disease control than the other chemotherapeutic agents used, was provided to a relatively small number of patients. FOLFOX CTx was not covered by the Korean national insurance during our study period; therefore, when there was concern about distant metastasis (increased number of positive lymph nodes, high CEA level, and apical lymph node metastasis), FOLFOX CTx was administered only if the patient agreed to pay for the treatment. This may account for our failure to observe better DFS in the CTx group. Finally, we were not able to compare treatment-related toxicity between the 2 groups.
In conclusion, this study provides further evidence that omission of postoperative CRT may not jeopardize oncologic outcomes in stage II and III upper rectal cancer patients. Furthermore, we observed that stage II upper rectal cancer patients are unlikely to experience local recurrence, regardless of whether they undergo postoperative CRT. Control of systemic disease and accurate pretreatment determination and assignment of patients at high risk for local failure to preoperative CRT need to occur to achieve major breakthroughs in the treatment of upper rectal cancer.
Fig. 1.
Oncologic outcomes in the entire cohort (A, local recurrence-free survival; B, disease-free survival; C, overall survival) and the propensity matched cohort (D, local recurrence-free survival; E, disease-free survival; F, overall survival). CRT, chemotherapy; F/U, follow-up.
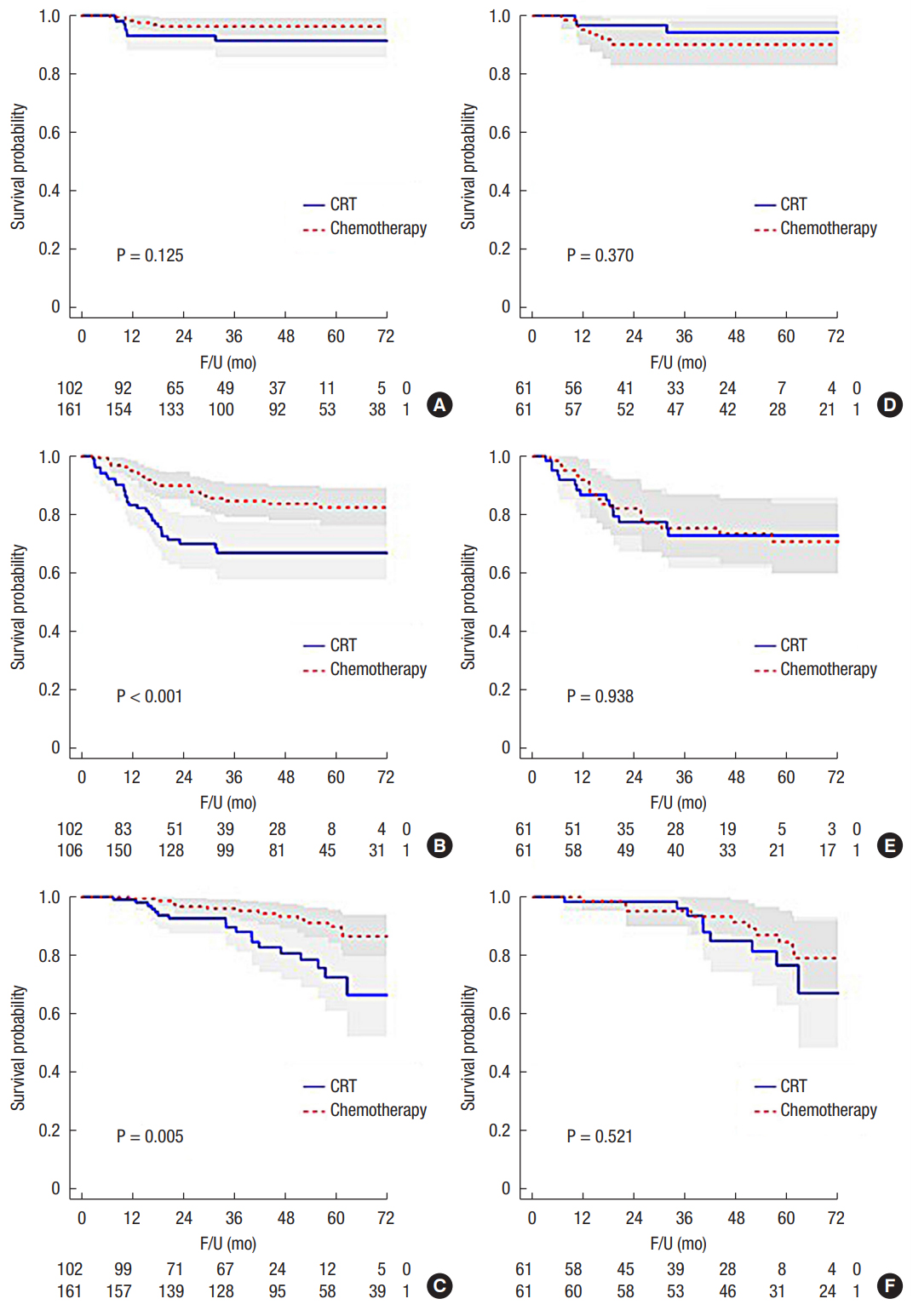
Table 1.
Patient characteristics
Table 2.
Sites of recurrence
Table 3.
Oncologic outcomes according to treatment group
Table 4.
Univariate and multivariate analyses of factors associated with local recurrence in upper rectal cancer
REFERENCES
2. Yoon M, Kim N, Nam B, Joo J, Ki M. Changing trends in colorectal cancer in the Republic of Korea: contrast with Japan. Epidemiol Health 2015;37:e2015038.



3. Sauer R, Becker H, Hohenberger W, Rödel C, Wittekind C, Fietkau R, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 2004;351:1731–40.


4. Kapiteijn E, Marijnen CA, Nagtegaal ID, Putter H, Steup WH, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med 2001;345:638–46.


5. Lopez-Kostner F, Lavery IC, Hool GR, Rybicki LA, Fazio VW. Total mesorectal excision is not necessary for cancers of the upper rectum. Surgery 1998;124:612–7.


6. Hainsworth PJ, Egan MJ, Cunliffe WJ. Evaluation of a policy of total mesorectal excision for rectal and rectosigmoid cancers. Br J Surg 1997;84:652–6.


7. Park JS, Sakai Y, Simon NS, Law WL, Kim HR, Oh JH, et al. Long-term survival and local relapse following surgery without radiotherapy for locally advanced upper rectal cancer: an international multi-institutional study. Medicine (Baltimore) 2016;95:e2990.



8. Tong T, Yao Z, Xu L, Cai S, Bi R, Xin C, et al. Extramural depth of tumor invasion at thin-section MR in rectal cancer: associating with prognostic factors and ADC value. J Magn Reson Imaging 2014;40:738–44.


9. Kim CH, Lee SY, Kim HR, Kim YJ. Pathologic stage following preoperative chemoradiotherapy underestimates the risk of developing distant metastasis in rectal cancer: a comparison to staging without preoperative chemoradiotherapy. J Surg Oncol 2016;113:692–9.


10. Swedish Rectal Cancer Trial; Cedermark B, Dahlberg M, Glimelius B, Påhlman L, Rutqvist LE, et al. Improved survival with preoperative radiotherapy in resectable rectal cancer. N Engl J Med 1997;336:980–7.


11. Valentini V, van Stiphout RG, Lammering G, Gambacorta MA, Barba MC, Bebenek M, et al. Nomograms for predicting local recurrence, distant metastases, and overall survival for patients with locally advanced rectal cancer on the basis of European randomized clinical trials. J Clin Oncol 2011;29:3163–72.


12. Gunderson LL, Sargent DJ, Tepper JE, Wolmark N, O’Connell MJ, Begovic M, et al. Impact of T and N stage and treatment on survival and relapse in adjuvant rectal cancer: a pooled analysis. J Clin Oncol 2004;22:1785–96.


13. Quirke P, Steele R, Monson J, Grieve R, Khanna S, Couture J, et al. Effect of the plane of surgery achieved on local recurrence in patients with operable rectal cancer: a prospective study using data from the MRC CR07 and NCIC-CTG CO16 randomised clinical trial. Lancet 2009;373:821–8.



14. Kim CH, Kim HJ, Huh JW, Kim YJ, Kim HR. Learning curve of laparoscopic low anterior resection in terms of local recurrence. J Surg Oncol 2014;110:989–96.


15. Attaallah W, Gunal O, Manukyan M, Ozden G, Yegen C. Prognostic impact of the metastatic lymph node ratio on survival in rectal cancer. Ann Coloproctol 2013;29:100–5.



16. Bernstein TE, Endreseth BH, Romundstad P, Wibe A, Norwegian Colorectal Cancer Group. Circumferential resection margin as a prognostic factor in rectal cancer. Br J Surg 2009;96:1348–57.










